Globe Newswire09.05.18
WishBone Medical Inc. announces the official launch of the single-use, sterile packed AlloMate Bone Pin System for the maintenance of alignment and fixation of bone fractures, osteotomies, arthrodesis or bone grafts and may also be used to fill a void left after screw removal.
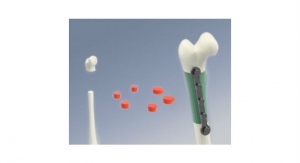
The AlloMate Bone Pin System consists of allograft bone pins and accessory instrumentation for quick, reliable and reproducible bone pin insertion. The complete system will be available in five different individual sizes (2 mm, 2.5 mm, 3 mm, 3.5 mm, and 4 mm) of sterilized human cortical bone pins and corresponding single-use instruments for convenient insertion. The company will launch these various sizes in stages. The 3 mm is currently available and the 2.5 mm will be available soon followed by the 2 mm and the additional sizes. Initial market feedback with the 3 mm system has been extremely positive.
Daniel S. Schulman, D.P.M., who helped develop this system, said, “I have performed well over four hundred foot reconstructive procedures without the use of a metal implant. I can validate that this system will definitely simplify the surgical technique and minimize intra-operative and post-operative surgical complications. Patients are elated when they are told there will be no need for a second surgery to remove potentially painful, broken or even infected hardware.”
These bone pins are sturdy, easy to handle and can integrate into the patient’s bone to help with the healing process. These sterile packed implants and instruments may potentially reduce the risk of infection and can be accurately trimmed intraoperatively to an appropriate length.
WishBone Medical Board Chairmand and CEO Nick Deeter said, “The AlloMate Bone Pin System does help minimize the need for removal in a secondary operation to remove hardware like stainless steel wires. Minimizing the number or surgeries for kids is very important to us.”
WishBone Medical is a global pediatric orthopedic company committed to providing anatomically appropriate innovative implants and instruments in sterile packed, single use, disposable kits to prevent infection, reduce overall costs for customers and achieve the best outcomes for children around the world who are still growing. The company is based in Warsaw, Ind.
The AlloMate Bone Pin System consists of allograft bone pins and accessory instrumentation for quick, reliable and reproducible bone pin insertion. The complete system will be available in five different individual sizes (2 mm, 2.5 mm, 3 mm, 3.5 mm, and 4 mm) of sterilized human cortical bone pins and corresponding single-use instruments for convenient insertion. The company will launch these various sizes in stages. The 3 mm is currently available and the 2.5 mm will be available soon followed by the 2 mm and the additional sizes. Initial market feedback with the 3 mm system has been extremely positive.
Daniel S. Schulman, D.P.M., who helped develop this system, said, “I have performed well over four hundred foot reconstructive procedures without the use of a metal implant. I can validate that this system will definitely simplify the surgical technique and minimize intra-operative and post-operative surgical complications. Patients are elated when they are told there will be no need for a second surgery to remove potentially painful, broken or even infected hardware.”
These bone pins are sturdy, easy to handle and can integrate into the patient’s bone to help with the healing process. These sterile packed implants and instruments may potentially reduce the risk of infection and can be accurately trimmed intraoperatively to an appropriate length.
WishBone Medical Board Chairmand and CEO Nick Deeter said, “The AlloMate Bone Pin System does help minimize the need for removal in a secondary operation to remove hardware like stainless steel wires. Minimizing the number or surgeries for kids is very important to us.”
WishBone Medical is a global pediatric orthopedic company committed to providing anatomically appropriate innovative implants and instruments in sterile packed, single use, disposable kits to prevent infection, reduce overall costs for customers and achieve the best outcomes for children around the world who are still growing. The company is based in Warsaw, Ind.