PR Newswire09.19.18
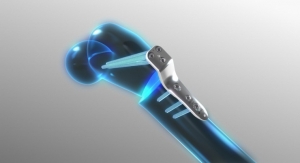
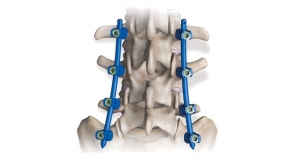
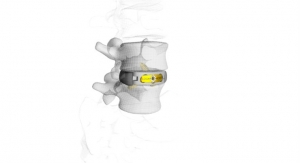
Meditech Spine has received FDA 510(k) clearance to market the Cure Opel-C Plate System, an add-on technology to its already cleared CURE ACP System and Talos-C (HA) Interbody Systems. With this approval, Meditech will now offer two additional CURE ACP plating options with its Talos line of cervical interbodies made with the osteoconductive PEEK-OPTIMA HA Enhanced material. Since Meditech's introduction of the CURE ACP System in late 2016, its adoption and usage have seen double-digit growth. By adding the Opel-C plating options to the CURE ACP product line, surgeons now have two modular snap-on plate styles providing rigid fixation while minimizing adjacent level impact. The Cure Opel-C Plate System incorporates the same unique, patented, one-step locking mechanism of the CURE ACP System preventing screws from backing out when anchored in the vertebral body. Meditech will be demonstrating the newly approved system at the North American Spine Society (NASS) meeting held in Los Angeles, Calif. from September 26-28 along with its proprietary Talos HA Peek Interbody Systems in booth 1757.
Meditech is excited to continue its relationship with CURE International, the namesake of the CURE ACP, an organization serving children with physical disabilities in underserved areas of the world. A portion of proceeds from the sale of every plate helps CURE International treat patients in need of surgical care.
Meditech is excited to continue its relationship with CURE International, the namesake of the CURE ACP, an organization serving children with physical disabilities in underserved areas of the world. A portion of proceeds from the sale of every plate helps CURE International treat patients in need of surgical care.