Zimmer Biomet09.19.18
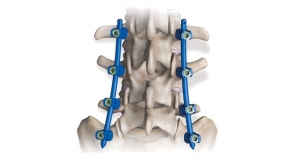
Zimmer Biomet Holdings Inc., a global leader in musculoskeletal healthcare, has launched its Mobi-C Cervical Disc for cervical total disc replacement (cTDR) in Japan. The launch follows receipt of regulatory approval from the Japan Ministry of Health, Labour and Welfare (MHLW) in May and the receipt of reimbursement from the MHLW in June. The first Mobi-C surgery in Japan was performed in July. The Mobi-C Cervical Disc is a leading option for cTDR designed to restore a healthy disc height and maintain patient range of motion at the operated level.
Rebecca Whitney, general manager of Global Spine, commented, “Mobi-C Cervical Disc is one of only two cervical discs available in Japan, both launched in 2018. This marks the expansion of Mobi-C’s innovative, motion-preserving technology in a new market with high unmet need. We look forward to serving the needs of our Japanese patients and surgeons with the Mobi-C, a proven alternative to cervical fusion.”
Cervical disc replacement is currently a regulated procedure in Japan, and surgeons wishing to perform the procedure must fulfill specific requirements, undergo a structured training program, and participate in post-market surveillance.
Mobi-C is the first cervical disc prosthesis approved by the U.S. Food and Drug Administration for reconstruction of a cervical disc at both one and two levels (C3-C7). Mobi-C is a cobalt chromium alloy and polyethylene mobile-bearing prosthesis that is inserted in a single step, without requiring bone chiseling to accommodate vertebral anchorage such as screws or keels. The Mobi-C Cervical Disc Prosthesis is indicated in skeletally mature patients for reconstruction of the disc from C3-C7 following discectomy at one level or two contiguous levels for intractable radiculopathy (arm pain and/or neurological deficit) with or without neck pain or myelopathy due to abnormality localized to the level of the disc space and at least one of the following conditions confirmed by radiographic imaging (CT, MRI or X-rays): herniated nucleus pulposus, spondylosis (defined by the presence of osteophytes) and/or visible loss of disc height compared to adjacent levels. The Mobi-C Cervical Disc Prosthesis is implanted using an anterior approach. Patients should have failed at least six weeks of conservative treatment or demonstrated progressive signs or symptoms despite nonoperative treatment prior to implantation of the Mobi-C Cervical Disc Prosthesis.
Founded in 1927 and headquartered in Warsaw, Ind., Zimmer Biomet designs, manufactures, and markets orthopedic reconstructive products; sports medicine, biologics, extremities and trauma products; office-based technologies; spine, craniomaxillofacial and thoracic products; dental implants; and related surgical products. The company's products and solutions help treat patients suffering from disorders of, or injuries to, bones, joints or supporting soft tissues. Zimmer Biomet has operations in more than 25 countries around the world and sells products in more than 100 countries.
Rebecca Whitney, general manager of Global Spine, commented, “Mobi-C Cervical Disc is one of only two cervical discs available in Japan, both launched in 2018. This marks the expansion of Mobi-C’s innovative, motion-preserving technology in a new market with high unmet need. We look forward to serving the needs of our Japanese patients and surgeons with the Mobi-C, a proven alternative to cervical fusion.”
Cervical disc replacement is currently a regulated procedure in Japan, and surgeons wishing to perform the procedure must fulfill specific requirements, undergo a structured training program, and participate in post-market surveillance.
Mobi-C is the first cervical disc prosthesis approved by the U.S. Food and Drug Administration for reconstruction of a cervical disc at both one and two levels (C3-C7). Mobi-C is a cobalt chromium alloy and polyethylene mobile-bearing prosthesis that is inserted in a single step, without requiring bone chiseling to accommodate vertebral anchorage such as screws or keels. The Mobi-C Cervical Disc Prosthesis is indicated in skeletally mature patients for reconstruction of the disc from C3-C7 following discectomy at one level or two contiguous levels for intractable radiculopathy (arm pain and/or neurological deficit) with or without neck pain or myelopathy due to abnormality localized to the level of the disc space and at least one of the following conditions confirmed by radiographic imaging (CT, MRI or X-rays): herniated nucleus pulposus, spondylosis (defined by the presence of osteophytes) and/or visible loss of disc height compared to adjacent levels. The Mobi-C Cervical Disc Prosthesis is implanted using an anterior approach. Patients should have failed at least six weeks of conservative treatment or demonstrated progressive signs or symptoms despite nonoperative treatment prior to implantation of the Mobi-C Cervical Disc Prosthesis.
Founded in 1927 and headquartered in Warsaw, Ind., Zimmer Biomet designs, manufactures, and markets orthopedic reconstructive products; sports medicine, biologics, extremities and trauma products; office-based technologies; spine, craniomaxillofacial and thoracic products; dental implants; and related surgical products. The company's products and solutions help treat patients suffering from disorders of, or injuries to, bones, joints or supporting soft tissues. Zimmer Biomet has operations in more than 25 countries around the world and sells products in more than 100 countries.