PR Newswire11.09.18
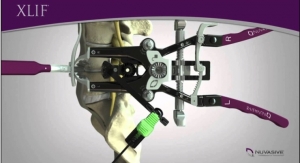
NuVasive Inc., a provider of spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for use of its COHERE Porous PEEK implant in eXtreme Lateral Interbody Fusion (XLIF) surgical spine procedures.
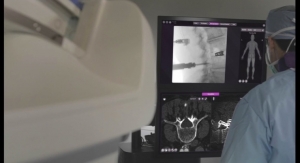
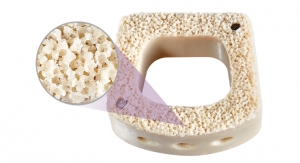
NuVasive's patented Porous PEEK technology offers a unique three-dimensional architecture combined with the radiolucent properties of PEEK. Based on preclinical studies, this porous structure promotes bone ongrowth and ingrowth, which is key to facilitating fusion that leads to better clinical outcomes. This unique, proprietary technology—combined with the radiolucent properties of PEEK—provides increased clarity in postoperative x-rays and imaging, allowing surgeons to more easily assess fusion following procedures.
"COHERE XLIF with its one-of-a-kind Porous PEEK technology provides surgeons the best implant for treating patients. It is designed to allow early incorporation of bone into the inter-body spacer, which may lead to the prevention of expulsion and promotion of early fusion which allows a quicker and better way back to an active lifestyle for my patients," said Dr. Christopher R. Brown, orthopedic spine surgeon for Duke Health. "Incorporating COHERE Porous PEEK implants into XLIF procedures keeps imaging characteristics that we all love about PEEK implants intact. COHERE XLIF takes the gold standard of lateral interbody fusion and raises the bar, allowing our patients to receive the best surgery with the best implant possible."
The 510(k) clearance supports the Company's strategic expansion of its Advanced Materials Science technologies into complementary NuVasive procedures. An abstract recently published by Biomaterials, further validates that Porous PEEK implant surfaces provide a stronger osseointegration response than smooth PEEK surfaces. The conclusion is based on results from an in vitro cell study and in vivo small animal model.
"It's exciting to extend our patented interbody technology to the Company's flagship XLIF procedure, an integral part of our single-position surgery portfolio, as we further our mission to improve patient lives," said Matt Link, president, Strategy, Technology and Corporate Development for NuVasive. "COHERE XLIF represents the first buildout of a Porous PEEK implant by NuVasive, and we look forward to extending this proprietary technology into additional applications across the Company's comprehensive procedural solutions."
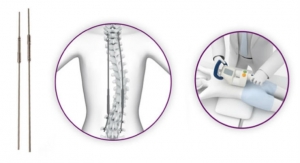
The FDA 510(k) indications for COHERE allow surgeons to use the interbody implant with autograft and/or allograft in skeletally mature patients for thoracolumbar pathologies including degenerative disc disease, degenerative spondylolisthesis, and degenerative scoliosis. NuVasive worked alongside highly experienced XLIF surgeons to pioneer the design of the COHERE XLIF implant to achieve the optimal shape and geometry for lateral insertion.
COHERE XLIF is expected to launch commercially in the U.S. in second quarter 2019.
NuVasive's patented Porous PEEK technology offers a unique three-dimensional architecture combined with the radiolucent properties of PEEK. Based on preclinical studies, this porous structure promotes bone ongrowth and ingrowth, which is key to facilitating fusion that leads to better clinical outcomes. This unique, proprietary technology—combined with the radiolucent properties of PEEK—provides increased clarity in postoperative x-rays and imaging, allowing surgeons to more easily assess fusion following procedures.
"COHERE XLIF with its one-of-a-kind Porous PEEK technology provides surgeons the best implant for treating patients. It is designed to allow early incorporation of bone into the inter-body spacer, which may lead to the prevention of expulsion and promotion of early fusion which allows a quicker and better way back to an active lifestyle for my patients," said Dr. Christopher R. Brown, orthopedic spine surgeon for Duke Health. "Incorporating COHERE Porous PEEK implants into XLIF procedures keeps imaging characteristics that we all love about PEEK implants intact. COHERE XLIF takes the gold standard of lateral interbody fusion and raises the bar, allowing our patients to receive the best surgery with the best implant possible."
The 510(k) clearance supports the Company's strategic expansion of its Advanced Materials Science technologies into complementary NuVasive procedures. An abstract recently published by Biomaterials, further validates that Porous PEEK implant surfaces provide a stronger osseointegration response than smooth PEEK surfaces. The conclusion is based on results from an in vitro cell study and in vivo small animal model.
"It's exciting to extend our patented interbody technology to the Company's flagship XLIF procedure, an integral part of our single-position surgery portfolio, as we further our mission to improve patient lives," said Matt Link, president, Strategy, Technology and Corporate Development for NuVasive. "COHERE XLIF represents the first buildout of a Porous PEEK implant by NuVasive, and we look forward to extending this proprietary technology into additional applications across the Company's comprehensive procedural solutions."
The FDA 510(k) indications for COHERE allow surgeons to use the interbody implant with autograft and/or allograft in skeletally mature patients for thoracolumbar pathologies including degenerative disc disease, degenerative spondylolisthesis, and degenerative scoliosis. NuVasive worked alongside highly experienced XLIF surgeons to pioneer the design of the COHERE XLIF implant to achieve the optimal shape and geometry for lateral insertion.
COHERE XLIF is expected to launch commercially in the U.S. in second quarter 2019.