BioPoly LLC01.17.19
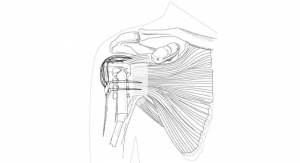
BioPoly LLC has announced that another European patent has been granted for its partial resurfacing implants. Where many of the other BioPoly patents address the material, this patent family focuses on the implant designs. With four product lines that are now regulatory approved (i.e. CE marked) in Europe, receiving additional intellectual property protection in European countries is an important part of the company’s overall risk management plan.
With BioPoly’s recent announcement that it received U.S. Food and Drug Administration approval to begin an IDE Clinical study in the United States with its BioPoly RS Partial Resurfacing Knee implant, the intellectual property portfolio provides the assurance that all aspects of the implants, material, and surgical technique are protected throughout the world.
The unique BioPoly material behaves like an artificial cartilage because of its patented combination of a molecule found in cartilage with an orthopedic plastic (polyethylene based). The BioPoly material is being used to replace only the damaged cartilage rather than to replace the entire joint, thus, allowing patients to return to active, pain-free lifestyles. Patients in Europe have been receiving the implants since 2012.
BioPoly LLC is an ISO 13485-certified orthopedic implant manufacturer located in Fort Wayne, Ind. The company is developing, manufacturing, and marketing products for use in sports medicine, orthopedics, and spinal markets. Additional medical applications of the BioPoly technology in cardiovascular and trauma markets are also being pursued.
With BioPoly’s recent announcement that it received U.S. Food and Drug Administration approval to begin an IDE Clinical study in the United States with its BioPoly RS Partial Resurfacing Knee implant, the intellectual property portfolio provides the assurance that all aspects of the implants, material, and surgical technique are protected throughout the world.
The unique BioPoly material behaves like an artificial cartilage because of its patented combination of a molecule found in cartilage with an orthopedic plastic (polyethylene based). The BioPoly material is being used to replace only the damaged cartilage rather than to replace the entire joint, thus, allowing patients to return to active, pain-free lifestyles. Patients in Europe have been receiving the implants since 2012.
BioPoly LLC is an ISO 13485-certified orthopedic implant manufacturer located in Fort Wayne, Ind. The company is developing, manufacturing, and marketing products for use in sports medicine, orthopedics, and spinal markets. Additional medical applications of the BioPoly technology in cardiovascular and trauma markets are also being pursued.