Miach Orthopaedics03.12.19
Miach Orthopaedics Inc., a privately held company developing bio-engineered surgical implants for connective tissue repair, today announced study results showing the Bridge-Enhanced ACL Repair (BEAR) procedure using the BEAR implant resulted in similar clinical, functional and patient-reported outcomes compared to patients undergoing autograft ACL reconstruction 24 months after surgery. The results of the BEAR I feasibility study conducted at Boston Children’s Hospital will be presented at the Arthroscopy Association of North America (AANA)/American Orthopaedic Society for Sports Medicine (AOSSM) 2019 Specialty Day on March 16.
“In this small, first-in-human study, Bridge-Enhanced ACL Repair with the BEAR implant had similar outcomes to ACL reconstruction with autograft hamstring,” said Martha Murray, M.D., professor of orthopaedic surgery at Harvard Medical School, an orthopedic surgeon in the Sports Medicine Division at Boston Children’s Hospital and founder of Miach Orthopaedics. “These results are promising and suggest the BEAR implant is worthy of further study. We’ve completed enrollment of 100 subjects in the BEAR II randomized controlled trial, and we look forward to those results.”
The BEAR I study assessed the safety and early efficacy of the BEAR implant to repair a torn anterior cruciate ligament (ACL). In addition to similar clinical, functional and patient-reported outcomes, the procedure using the BEAR implant did not result in any patients having an infection or a severe inflammatory reaction, arthrofibrosis, or a reaction that required scaffold removal. In addition, manual and instrumented measures suggest the stability of the knee after both procedures may be comparable.
“It has been four years since we initiated human studies of ACL injury repair using the BEAR scaffold,” said Lyle J. Micheli, M.D., director of Boston Children’s Hospital Division of Sports Medicine. “During this time, we have completed two clinical studies to evaluate this technique. The first was this BEAR I safety study, comparing 10 BEAR repaired knees with 10 treated with ACL reconstruction with autograft. The second was a randomized, blinded study of 100 patients, again comparing BEAR ACL repairs with autograft reconstruction. The early results of these studies have been encouraging. We are planning a third study to look further at the effects of age on the outcomes of this technique.”
BEAR I was a non-randomized, two-arm study conducted at Boston Children’s Hospital that compared 10 patients treated with the BEAR implant to 10 patients treated with hamstring autograft ACL reconstruction. It was conducted under an Investigational Device Exemption (IDE) from the U.S. Food and Drug Administration (FDA) and registered on clinicaltrials.gov. Funding support for the study came from Boston Children's Hospital, the National Institutes of Health and the National Football League Players Association.
Every year, approximately 200,000 ACL injuries occur in the United States. Without treatment, the ACL does not heal, resulting in ACL reconstruction surgery being one of the most common orthopaedic procedures in the United States Traditional ACL reconstruction using an autograft or allograft stabilizes the knee but has drawbacks— primarily that only about 60 percent of patients can return to their sport at the same level, and the ACL re-tear rate can be nearly 30 percent for teens.
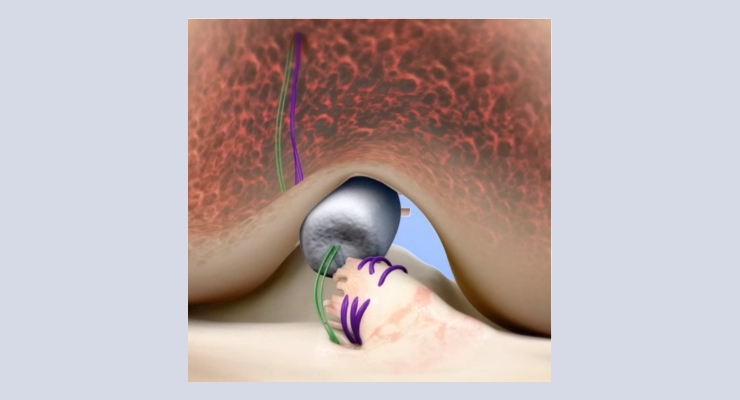
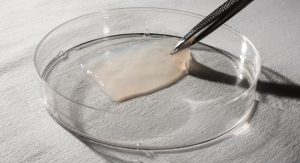
The Bridge-Enhanced ACL Repair (BEAR) implant is a proprietary bio-engineered bridging scaffold to facilitate healing of the torn ACL. It is designed to be surgically placed between the torn ACL ends at the time of repair, and to hold a small amount of the patient’s blood in the wound site. This provides a scaffold that allows the torn ends of the ACL to heal back together. It is hoped this new technology will restore more normal anatomy and function of the knee, and thus enable a higher percentage of patients to get back to activities they enjoy. The BEAR implant is an investigational device and is only available in FDA approved clinical trials.
Miach Orthopaedics Inc. is a privately held company located in Westborough, Mass., developing bio-engineered surgical implants for connective tissue repair. The company’s initial focus is on the Bridge-Enhanced ACL Repair (BEAR) technology as a viable alternative to conventional ACL reconstruction for patients who have sustained an ACL injury. The BEAR technology was pioneered by Martha Murray, M.D., at the Boston Children’s Hospital Department of Orthopaedic Surgery with initial research funding provided by the NFL Players Association, Boston Children’s Hospital and the National Institutes of Health.
“In this small, first-in-human study, Bridge-Enhanced ACL Repair with the BEAR implant had similar outcomes to ACL reconstruction with autograft hamstring,” said Martha Murray, M.D., professor of orthopaedic surgery at Harvard Medical School, an orthopedic surgeon in the Sports Medicine Division at Boston Children’s Hospital and founder of Miach Orthopaedics. “These results are promising and suggest the BEAR implant is worthy of further study. We’ve completed enrollment of 100 subjects in the BEAR II randomized controlled trial, and we look forward to those results.”
The BEAR I study assessed the safety and early efficacy of the BEAR implant to repair a torn anterior cruciate ligament (ACL). In addition to similar clinical, functional and patient-reported outcomes, the procedure using the BEAR implant did not result in any patients having an infection or a severe inflammatory reaction, arthrofibrosis, or a reaction that required scaffold removal. In addition, manual and instrumented measures suggest the stability of the knee after both procedures may be comparable.
“It has been four years since we initiated human studies of ACL injury repair using the BEAR scaffold,” said Lyle J. Micheli, M.D., director of Boston Children’s Hospital Division of Sports Medicine. “During this time, we have completed two clinical studies to evaluate this technique. The first was this BEAR I safety study, comparing 10 BEAR repaired knees with 10 treated with ACL reconstruction with autograft. The second was a randomized, blinded study of 100 patients, again comparing BEAR ACL repairs with autograft reconstruction. The early results of these studies have been encouraging. We are planning a third study to look further at the effects of age on the outcomes of this technique.”
BEAR I was a non-randomized, two-arm study conducted at Boston Children’s Hospital that compared 10 patients treated with the BEAR implant to 10 patients treated with hamstring autograft ACL reconstruction. It was conducted under an Investigational Device Exemption (IDE) from the U.S. Food and Drug Administration (FDA) and registered on clinicaltrials.gov. Funding support for the study came from Boston Children's Hospital, the National Institutes of Health and the National Football League Players Association.
Every year, approximately 200,000 ACL injuries occur in the United States. Without treatment, the ACL does not heal, resulting in ACL reconstruction surgery being one of the most common orthopaedic procedures in the United States Traditional ACL reconstruction using an autograft or allograft stabilizes the knee but has drawbacks— primarily that only about 60 percent of patients can return to their sport at the same level, and the ACL re-tear rate can be nearly 30 percent for teens.
The Bridge-Enhanced ACL Repair (BEAR) implant is a proprietary bio-engineered bridging scaffold to facilitate healing of the torn ACL. It is designed to be surgically placed between the torn ACL ends at the time of repair, and to hold a small amount of the patient’s blood in the wound site. This provides a scaffold that allows the torn ends of the ACL to heal back together. It is hoped this new technology will restore more normal anatomy and function of the knee, and thus enable a higher percentage of patients to get back to activities they enjoy. The BEAR implant is an investigational device and is only available in FDA approved clinical trials.
Miach Orthopaedics Inc. is a privately held company located in Westborough, Mass., developing bio-engineered surgical implants for connective tissue repair. The company’s initial focus is on the Bridge-Enhanced ACL Repair (BEAR) technology as a viable alternative to conventional ACL reconstruction for patients who have sustained an ACL injury. The BEAR technology was pioneered by Martha Murray, M.D., at the Boston Children’s Hospital Department of Orthopaedic Surgery with initial research funding provided by the NFL Players Association, Boston Children’s Hospital and the National Institutes of Health.