Promimic08.28.19
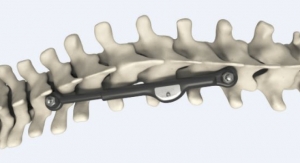
The first spinal device utilizing HAnano Surface to improve osseointegration has now been 510(k) approved by the FDA. Through partnership with Promimic, Cutting Edge Spine developed the EVOL SI Sacroiliac Joint Fusion System, which will be commercially launched later this year.
The HAnano Surface has been in clinical use on the dental implant market for several years, showing faster integration between the bone and the implant. Promimic has established U.S. operations, and is now entering the orthopedic market with multiple OEM partners.
“Cutting Edge Spine represents a perfect partner for Promimic, with their tradition of pioneering novel technologies and successfully bringing them to the market,” says Magnus Larsson.
HAnano Surface has proven to accelerate bone growth in over 30 in vivo and in vitro studies. Faster and stronger osseointegration has been demonstrated through biomechanical, histomorphological, and biological evaluations, in combination with over 150,000 clinical applications to date.
“In our quest to improve patient care, we wanted a bioactive solution to secure faster and better implant integration in SI fusion procedures. HAnano Surface was the obvious choice for us when we looked for an implant surface modification to achieve those goals,” says Randy Roof, CEO, President and Founder of Cutting Edge Spine.
Promimic will be exhibiting at the North American Spine Society (NASS) Annual Meeting in Chicago, Ill., September 25-28, Booth #3901.
The HAnano Surface has been in clinical use on the dental implant market for several years, showing faster integration between the bone and the implant. Promimic has established U.S. operations, and is now entering the orthopedic market with multiple OEM partners.
“Cutting Edge Spine represents a perfect partner for Promimic, with their tradition of pioneering novel technologies and successfully bringing them to the market,” says Magnus Larsson.
HAnano Surface has proven to accelerate bone growth in over 30 in vivo and in vitro studies. Faster and stronger osseointegration has been demonstrated through biomechanical, histomorphological, and biological evaluations, in combination with over 150,000 clinical applications to date.
“In our quest to improve patient care, we wanted a bioactive solution to secure faster and better implant integration in SI fusion procedures. HAnano Surface was the obvious choice for us when we looked for an implant surface modification to achieve those goals,” says Randy Roof, CEO, President and Founder of Cutting Edge Spine.
Promimic will be exhibiting at the North American Spine Society (NASS) Annual Meeting in Chicago, Ill., September 25-28, Booth #3901.