Omnia Medical LLC10.17.19
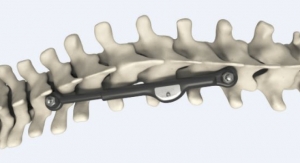
Omnia Medical’s ‘Boxcar’ has been granted the first U.S. Food and Drug Administration (FDA) 510(k) clearance for a cervical vertebral body replacement (VBR) system manufactured from PEEK-OPTIMA HA Enhanced polymer, from Invibio Biomaterial Solutions. The Boxcar system has been designed for use in cervical-corpectomy procedures—
that is, the replacement of a collapsed, damaged, or unstable vertebral body located in the cervical spine.
“I see many patients that require a cervical corpectomy, and it's exciting to have such an innovative option for these patients," said Robert Gewirtz, M.D., a neurosurgeon in Columbus, Ohio, who collaborated on the development of the Boxcar VBR product. "Boxcar is very easy to use and provides intra-operative flexibility, allowing me to dial in a VBR construct to match my patient’s anatomy. Hydroxyapatite is incorporated into the PEEK-OPTIMA structure of the implant, which delivers potentially improved outcomes for my patients”.
The single-use device is available in two footprints and allows fine adjustments of height and lordosis using spacers and endplates. It features a hollow center and holes to accommodate autograft or allograft, while encouraging the formation of new bone, and includes anti-migration features.
Steve Anderson, vice president of Marketing at Omnia Medical, added: “There is a limited number of 510(k) FDA-cleared cervical VBR products on the market, and for our company to gain the first approval for a PEEK-OPTIMA HA Enhanced cervical corpectomy device is a great achievement. I’m absolutely confident surgeons will see the clinical benefits for them and their patients. Omnia Medical is focused on bringing innovative products to market, and Boxcar is the first of many ‘first to market’ products from our company, going forward.”
In PEEK-OPTIMA HA Enhanced, hydroxyapatite (HA), a well-known osteoconductive material that enhances bone apposition, is fully integrated into the polymer matrix, and not simply coated. Invibio´s enhanced biomaterial offers all the clinical advantages of PEEK-OPTIMA Natural, including a modulus similar to that of cortical bone—unlike the metal or mesh cages in earlier use, plus reduced stress shielding and artifact-free imaging that allows for clear fusion assessment. It has been shown to improve bone apposition compared to unfilled PEEK-OPTIMA Natural in a pre-clinical ovine model at four weeks and 12 weeks,1 and also demonstrated performance advantages in a pre-clinical cervical spine fusion model in sheep at six and 12 weeks.2 In addition, as observed by surgeons, early clinical results have shown solid fusions with dense bone apposition at six months and beneficial clinical outcomes for patients, revealing specific improvements in overall pain and neurological function,3 when a PEEK-OPTIMA HA Enhanced interbody fusion device was used.
References
1 Study evaluated the in vivo response to PEEK-OPTIMA Natural and PEEK-OPTIMA HA Enhanced in a large animal model. Data on file at Invibio. This has not been correlated with human clinical experience.
2 Study evaluated the in vivo response to PEEK-OPTIMA Natural, PEEK-OPTIMA HA Enhanced and allograft in a cervical spine fusion model in sheep. Data on file at Invibio. This has not been correlated with human clinical experience.
3 Testimonials presented have been provided by participating orthopedic surgeons. Their views and experiences are their own and do not necessarily reflect those of others. “Invibio” disclaims any liabilities or loss in connection with the information herein.
“I see many patients that require a cervical corpectomy, and it's exciting to have such an innovative option for these patients," said Robert Gewirtz, M.D., a neurosurgeon in Columbus, Ohio, who collaborated on the development of the Boxcar VBR product. "Boxcar is very easy to use and provides intra-operative flexibility, allowing me to dial in a VBR construct to match my patient’s anatomy. Hydroxyapatite is incorporated into the PEEK-OPTIMA structure of the implant, which delivers potentially improved outcomes for my patients”.
The single-use device is available in two footprints and allows fine adjustments of height and lordosis using spacers and endplates. It features a hollow center and holes to accommodate autograft or allograft, while encouraging the formation of new bone, and includes anti-migration features.
Steve Anderson, vice president of Marketing at Omnia Medical, added: “There is a limited number of 510(k) FDA-cleared cervical VBR products on the market, and for our company to gain the first approval for a PEEK-OPTIMA HA Enhanced cervical corpectomy device is a great achievement. I’m absolutely confident surgeons will see the clinical benefits for them and their patients. Omnia Medical is focused on bringing innovative products to market, and Boxcar is the first of many ‘first to market’ products from our company, going forward.”
In PEEK-OPTIMA HA Enhanced, hydroxyapatite (HA), a well-known osteoconductive material that enhances bone apposition, is fully integrated into the polymer matrix, and not simply coated. Invibio´s enhanced biomaterial offers all the clinical advantages of PEEK-OPTIMA Natural, including a modulus similar to that of cortical bone—unlike the metal or mesh cages in earlier use, plus reduced stress shielding and artifact-free imaging that allows for clear fusion assessment. It has been shown to improve bone apposition compared to unfilled PEEK-OPTIMA Natural in a pre-clinical ovine model at four weeks and 12 weeks,1 and also demonstrated performance advantages in a pre-clinical cervical spine fusion model in sheep at six and 12 weeks.2 In addition, as observed by surgeons, early clinical results have shown solid fusions with dense bone apposition at six months and beneficial clinical outcomes for patients, revealing specific improvements in overall pain and neurological function,3 when a PEEK-OPTIMA HA Enhanced interbody fusion device was used.
References
1 Study evaluated the in vivo response to PEEK-OPTIMA Natural and PEEK-OPTIMA HA Enhanced in a large animal model. Data on file at Invibio. This has not been correlated with human clinical experience.
2 Study evaluated the in vivo response to PEEK-OPTIMA Natural, PEEK-OPTIMA HA Enhanced and allograft in a cervical spine fusion model in sheep. Data on file at Invibio. This has not been correlated with human clinical experience.
3 Testimonials presented have been provided by participating orthopedic surgeons. Their views and experiences are their own and do not necessarily reflect those of others. “Invibio” disclaims any liabilities or loss in connection with the information herein.