PR Newswire11.14.19
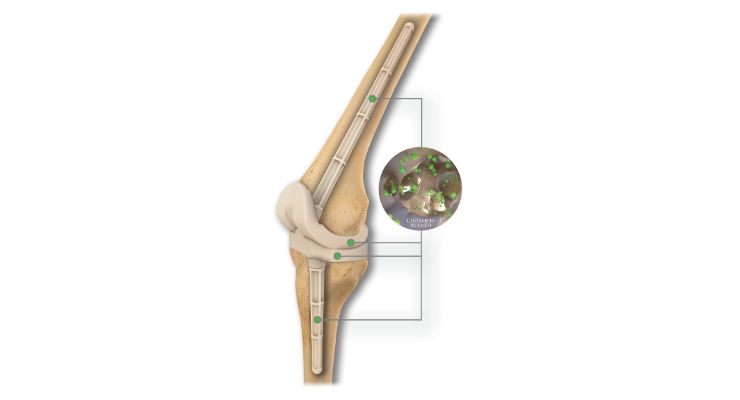
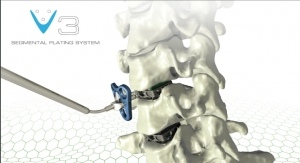
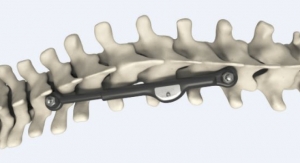
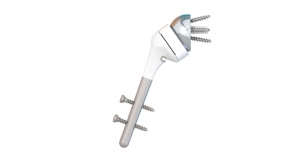
OsteoRemedies LLC, a company focused on providing simple solutions to complex disorders, has commercially launched the REMEDY Stemmed Knee Spacer. Building on its portfolio consisting of the only preformed modular spacer systems with Gentamicin for the hip, knee, and shoulder, as well as the only Gentamicin + Vancomycin preformed modular spacer and Bone Cement for the hip, the REMEDY Stemmed Knee will provide surgeons with the first ever preformed knee spacer with attachable modular stems that can connect to the femoral and tibial component.
"Our vision is to continue to provide simple but comprehensive solutions to the growing clinical challenges surrounding infected total joints. By providing surgeons with the only modular knee stems cleared by the FDA, we believe our portfolio remains the most robust which allows us to bolster our leadership position," stated Chris Hughes, president and CEO, OsteoRemedies.
Eric Stookey, chief operating officer of OsteoRemedies, noted, "After five years clinical experience with the current knee spacer design, our surgeon design team and users clearly identified the need for additional knee stem extensions for use in more complex two-stage revisions for infection. By incorporating our proprietary premolded and modular approach to the knee stem design, we can offer surgeons the opportunity to improve OR efficiency vs handmade IM dowels."
OsteoRemedies provides simple solutions to complex musculoskeletal disorders. Its first introduction was the REMEDY Spacer System. This was the first available modular system for hip, knee and shoulder two-stage infection revision arthroplasty. The REMEDY Spacer System and REMEDY SPECTRUM GV Hip Spacers are indicated for temporary use (maximum 180 days) as an adjunct to total joint replacement in skeletally mature patients undergoing a two-stage procedure due to a septic process and where Gentamicin or Gentamicin and Vancomycin are the most appropriate antibiotics based on the susceptibility pattern of the infecting micro-organisms. SPECTRUM GV Bone Cement is indicated for use with the REMEDY SPECTRUM GV Hip Spacer System. Other products marketed by OsteoRemedies include OSTEOBOOST RBK and FLORASEAL Microbial Sealant and UNITE AB Bone Cement.
"Our vision is to continue to provide simple but comprehensive solutions to the growing clinical challenges surrounding infected total joints. By providing surgeons with the only modular knee stems cleared by the FDA, we believe our portfolio remains the most robust which allows us to bolster our leadership position," stated Chris Hughes, president and CEO, OsteoRemedies.
Eric Stookey, chief operating officer of OsteoRemedies, noted, "After five years clinical experience with the current knee spacer design, our surgeon design team and users clearly identified the need for additional knee stem extensions for use in more complex two-stage revisions for infection. By incorporating our proprietary premolded and modular approach to the knee stem design, we can offer surgeons the opportunity to improve OR efficiency vs handmade IM dowels."
OsteoRemedies provides simple solutions to complex musculoskeletal disorders. Its first introduction was the REMEDY Spacer System. This was the first available modular system for hip, knee and shoulder two-stage infection revision arthroplasty. The REMEDY Spacer System and REMEDY SPECTRUM GV Hip Spacers are indicated for temporary use (maximum 180 days) as an adjunct to total joint replacement in skeletally mature patients undergoing a two-stage procedure due to a septic process and where Gentamicin or Gentamicin and Vancomycin are the most appropriate antibiotics based on the susceptibility pattern of the infecting micro-organisms. SPECTRUM GV Bone Cement is indicated for use with the REMEDY SPECTRUM GV Hip Spacer System. Other products marketed by OsteoRemedies include OSTEOBOOST RBK and FLORASEAL Microbial Sealant and UNITE AB Bone Cement.