Sam Brusco, Associate Editor01.19.22
Orthofix began a partnership and investment agreement with nView Medical, a Salt Lake City-based developer of artificial intelligence (AI)-enabled surgical imaging and guidance systems. The companies will jointly develop and co-market nView systems with Orthofix’s cervical spine and pediatric limb deformity correction technologies.
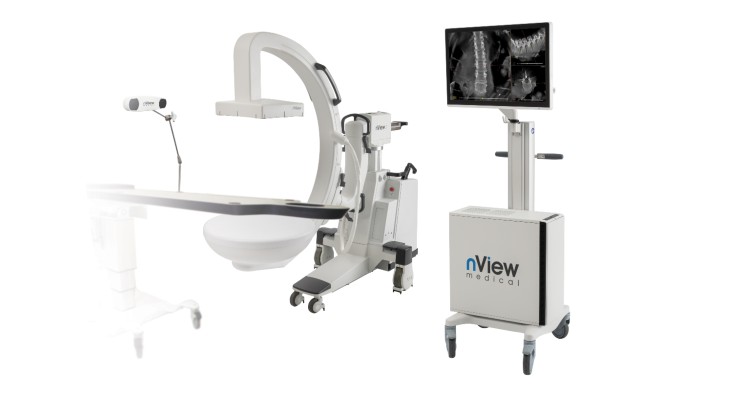
nView’s U.S. Food and Drug Administration (FDA)-cleared s1 imaging system with proprietary insta-3D tech instantly captures 3D images with very low-dose radiation, making the images available throughout surgery for real-time visualization. It also functions as a surgical guidance system with GPS-like tech using true anatomical representation as maps, and incorporates AI tech to augment the surgeon’s understanding of the procedure.
Orthofix will use an nView s1 imaging and navigation system in its Lewisville, Texas training facility and conduct joint education programs for surgeons. The companies intend to earn 510(k) clearances for Orthofix pediatric limb deformity tech navigated with nView s1. The companies will also pursue cervical spine applications.
“The nView s1 system’s insta-3D imaging technology complements Orthofix’s pre-planning and post-operative software platforms,” Orthofix president and CEO Jon Serbousek told the press. “It is an innovative digital solution aimed at better streamlining the surgery by reducing radiation exposure and decreasing OR time, which is so important for pediatric surgeons and their patients. We also look forward to working with nView to expand application of this technology for use in cervical spine procedures, utilizing Orthofix cervical solutions.”
“We are very excited to partner with Orthofix, who has a long history in pediatric limb deformity correction, and deep expertise with adult cervical spine procedures,” added nView medical founder and CEO Cristian Atria. “As we continue the initial commercial rollout of our technology in pediatrics, we look forward to bringing our value proposition to more patients and surgeons, who are also served by Orthofix.”
nView’s U.S. Food and Drug Administration (FDA)-cleared s1 imaging system with proprietary insta-3D tech instantly captures 3D images with very low-dose radiation, making the images available throughout surgery for real-time visualization. It also functions as a surgical guidance system with GPS-like tech using true anatomical representation as maps, and incorporates AI tech to augment the surgeon’s understanding of the procedure.
Orthofix will use an nView s1 imaging and navigation system in its Lewisville, Texas training facility and conduct joint education programs for surgeons. The companies intend to earn 510(k) clearances for Orthofix pediatric limb deformity tech navigated with nView s1. The companies will also pursue cervical spine applications.
“The nView s1 system’s insta-3D imaging technology complements Orthofix’s pre-planning and post-operative software platforms,” Orthofix president and CEO Jon Serbousek told the press. “It is an innovative digital solution aimed at better streamlining the surgery by reducing radiation exposure and decreasing OR time, which is so important for pediatric surgeons and their patients. We also look forward to working with nView to expand application of this technology for use in cervical spine procedures, utilizing Orthofix cervical solutions.”
“We are very excited to partner with Orthofix, who has a long history in pediatric limb deformity correction, and deep expertise with adult cervical spine procedures,” added nView medical founder and CEO Cristian Atria. “As we continue the initial commercial rollout of our technology in pediatrics, we look forward to bringing our value proposition to more patients and surgeons, who are also served by Orthofix.”