Sam Brusco, Associate Editor03.24.22
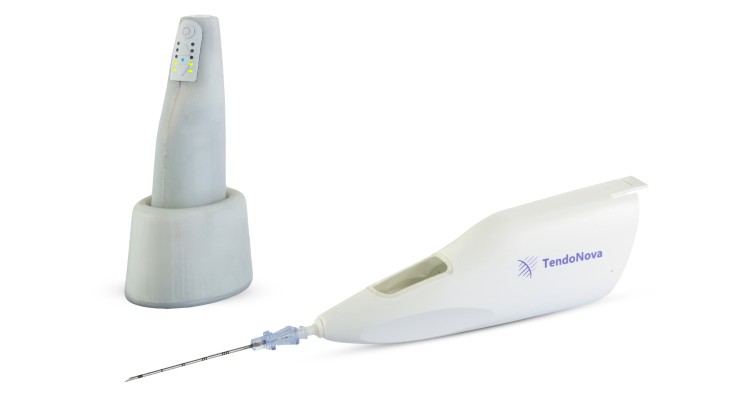
Microinvasive sports medicine company TendoNova has gained U.S. Food and Drug Administration (FDA) 510(k) clearance for its new Ocelot surgical tool to fragment or debride soft tissue. It can be used for percutaneous tenotomy in orthopedics, sports medicine, pain management, and podiatry.
Ocelot helps procedures needing soft tissue fragmentation and debridement under external image guidance that need a handheld format.
"According to studies cited by the Centers for Disease Control and Prevention, more than 30 million people in the U.S. suffer from chronic tendon pain, and half of those have little or no relief from physical therapy or medication," TendoNova CEO Mark A. Samuels told the press. "The Ocelot is the company's first product and was conceived in response to a request by Atlanta Hawks head team physician Dr. Ken Mautner for an improved tool."
Dr. Mautner, who practices at Emory, commented that, "After 15 years of treating elite and recreational athletes for tendinopathy, I have been frustrated by the tools we have had to help heal these injuries. The TendoNova device could be a game changer in getting our athletes back to the healthiest level possible."
Using fragmentation and debridement is part of treatment to encourage healthy tendon growth and supplant painful, pathologic tendon tissue. Chronic tendon pain disorders are prevalent in athletes or anyone else engaging in repetitive tasks.
The Ocelot will be manufactured in Georgia and the company is working on validating the Ocelot disposable component production. It’s expected to be available in the second half of this year.
Ocelot helps procedures needing soft tissue fragmentation and debridement under external image guidance that need a handheld format.
"According to studies cited by the Centers for Disease Control and Prevention, more than 30 million people in the U.S. suffer from chronic tendon pain, and half of those have little or no relief from physical therapy or medication," TendoNova CEO Mark A. Samuels told the press. "The Ocelot is the company's first product and was conceived in response to a request by Atlanta Hawks head team physician Dr. Ken Mautner for an improved tool."
Dr. Mautner, who practices at Emory, commented that, "After 15 years of treating elite and recreational athletes for tendinopathy, I have been frustrated by the tools we have had to help heal these injuries. The TendoNova device could be a game changer in getting our athletes back to the healthiest level possible."
Using fragmentation and debridement is part of treatment to encourage healthy tendon growth and supplant painful, pathologic tendon tissue. Chronic tendon pain disorders are prevalent in athletes or anyone else engaging in repetitive tasks.
The Ocelot will be manufactured in Georgia and the company is working on validating the Ocelot disposable component production. It’s expected to be available in the second half of this year.