Sam Brusco, Associate Editor03.30.22
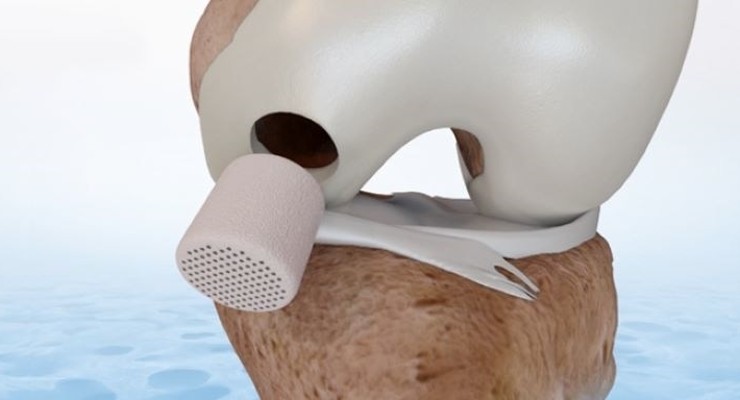
CartiHeal has earned U.S. Food and Drug Administration (FDA) premarket approval for its Agili-C implant to treat International Cartilage Repair Society (ICRS) grade III or above knee-joint surface lesions with total treatable area of 1-7cm2 and without severe osteoarthritis.
A two-year, 251-patient IDE clinical study demonstrated Agili-C’s superiority over microfracture and debridement standard of care to treat knee joint surface lesions, chondral and osteochondral defects.
After 24 months, patients using Agili-C showed knee injury and arthritis score (KOOS) improvement of about 43 points compared to about 20 points for those who underwent standard of care treatments.
"The 2-year study results, which demonstrated superiority of the Agili-C implant over the current surgical standard of care, offers an important potential benefit to millions of patients", Nir Altschuler, CartiHeal's founder and CEO told the press. "This milestone achievement was made possible due to the support of our regulatory advisors, Hogan Lovells, our statistical consultants, Biomedical Statistical Consulting, and the many dedicated investigators and patients who participated in our studies. We are grateful for all their help. FDAs approval enables us to initiate commercialization and provide a superior solution for patients compared to the current standard of care options."
A two-year, 251-patient IDE clinical study demonstrated Agili-C’s superiority over microfracture and debridement standard of care to treat knee joint surface lesions, chondral and osteochondral defects.
After 24 months, patients using Agili-C showed knee injury and arthritis score (KOOS) improvement of about 43 points compared to about 20 points for those who underwent standard of care treatments.
"The 2-year study results, which demonstrated superiority of the Agili-C implant over the current surgical standard of care, offers an important potential benefit to millions of patients", Nir Altschuler, CartiHeal's founder and CEO told the press. "This milestone achievement was made possible due to the support of our regulatory advisors, Hogan Lovells, our statistical consultants, Biomedical Statistical Consulting, and the many dedicated investigators and patients who participated in our studies. We are grateful for all their help. FDAs approval enables us to initiate commercialization and provide a superior solution for patients compared to the current standard of care options."