Charles Sternberg, Associate Editor05.17.22
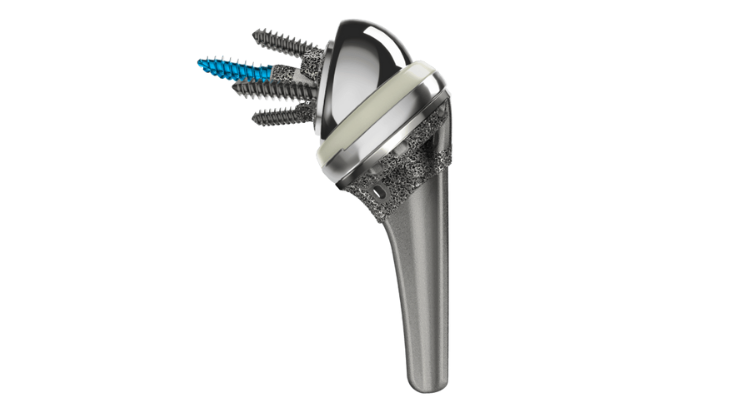
The INHANCE Shoulder System from DePuy Synthes, the Orthopedics Company of Johnson & Johnson, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for use in reverse total shoulder arthroplasty procedures.
This clearance is in addition to the system’s already cleared use in anatomic shoulder arthroplasty and provides surgeons with interoperative flexibility.
In 2020 alone, reverse shoulder arthroplasty accounted for 70% of all total shoulder arthroplasty procedures in the U.S.1 Surgeons typically perform a reverse total shoulder arthroplasty when there is arthritis of the shoulder joint, and the rotator cuff tendons are severely deficient. In complicated cases, surgeons may need to change their approach mid-surgery due to multiple factors, including humeral bone quality and severity of glenoid bone loss, or the extent of rotator cuff deficiency.
“The INHANCE Shoulder System can be used for an anatomic or reverse shoulder procedure offering the surgeon and OR staff a streamlined and effective shoulder system,” said Andrew Jawa, MD, orthopedic surgeon, New England Baptist Hospital. “This system contains the necessary tools from pre-op planning to intra-op flexibility with two trays of instruments to help ensure a consistent outcome.”
Features of the reverse system include:
The INHANCE Shoulder System is also designed for the shift in site of care of certain orthopedic procedures toward outpatient and ambulatory surgery centers where economic value and operational efficiency are critical considerations. The INHANCE Shoulder System's implants are designed to preserve bone, provide immediate and biological fixation, and facilitate intra-operative flexibility while simplifying preparation for various surgical treatment options.
“Adding the reverse total shoulder arthroplasty to our INHANCE Shoulder System is helping us transform patient care,” said Rajit Kamal, worldwide president, Sports Medicine & Shoulder Reconstruction, DePuy Synthes. “This reinforces our commitment to innovation that addresses the most pressing needs of surgeons today and the market dynamics driving clinical trends of tomorrow.”
The INHANCE Shoulder System was designed in collaboration with a team of leading shoulder arthroplasty surgeons and is competitively positioned to address the increasing complexity of shoulder replacement procedures by integrating less invasive clinical options aligned with how surgeons approach patient care.
Reference:
1 DePuy Synthes. Analysis of Primary Shoulder Arthroplasty Anatomic and Reverse Volumes from 2016‐2020 Using Medicare SAF, IBM MDCD and IBM CCAE. Raynham (MA). February 2022.
This clearance is in addition to the system’s already cleared use in anatomic shoulder arthroplasty and provides surgeons with interoperative flexibility.
In 2020 alone, reverse shoulder arthroplasty accounted for 70% of all total shoulder arthroplasty procedures in the U.S.1 Surgeons typically perform a reverse total shoulder arthroplasty when there is arthritis of the shoulder joint, and the rotator cuff tendons are severely deficient. In complicated cases, surgeons may need to change their approach mid-surgery due to multiple factors, including humeral bone quality and severity of glenoid bone loss, or the extent of rotator cuff deficiency.
“The INHANCE Shoulder System can be used for an anatomic or reverse shoulder procedure offering the surgeon and OR staff a streamlined and effective shoulder system,” said Andrew Jawa, MD, orthopedic surgeon, New England Baptist Hospital. “This system contains the necessary tools from pre-op planning to intra-op flexibility with two trays of instruments to help ensure a consistent outcome.”
Features of the reverse system include:
- 135° neck shaft angle with lateralized options
- Sizing options to address a full range of patients
- 3D laser printed R/SPEED Baseplates and Modular Baseplates with Central Screw and Central Post options
- Advanced cross-linked Vitamin E polyethylene for desired wear characteristics and oxidative stability
- One Step Prep Glenoid Reamers
The INHANCE Shoulder System is also designed for the shift in site of care of certain orthopedic procedures toward outpatient and ambulatory surgery centers where economic value and operational efficiency are critical considerations. The INHANCE Shoulder System's implants are designed to preserve bone, provide immediate and biological fixation, and facilitate intra-operative flexibility while simplifying preparation for various surgical treatment options.
“Adding the reverse total shoulder arthroplasty to our INHANCE Shoulder System is helping us transform patient care,” said Rajit Kamal, worldwide president, Sports Medicine & Shoulder Reconstruction, DePuy Synthes. “This reinforces our commitment to innovation that addresses the most pressing needs of surgeons today and the market dynamics driving clinical trends of tomorrow.”
The INHANCE Shoulder System was designed in collaboration with a team of leading shoulder arthroplasty surgeons and is competitively positioned to address the increasing complexity of shoulder replacement procedures by integrating less invasive clinical options aligned with how surgeons approach patient care.
Reference:
1 DePuy Synthes. Analysis of Primary Shoulder Arthroplasty Anatomic and Reverse Volumes from 2016‐2020 Using Medicare SAF, IBM MDCD and IBM CCAE. Raynham (MA). February 2022.