Sam Brusco, Associate Editor05.31.22
Soft tissue healing firm Embody Inc. has received U.S. Food and Drug Administration (FDA) clearances for its TAPESTRY RC system to treat rotator cuff disease.
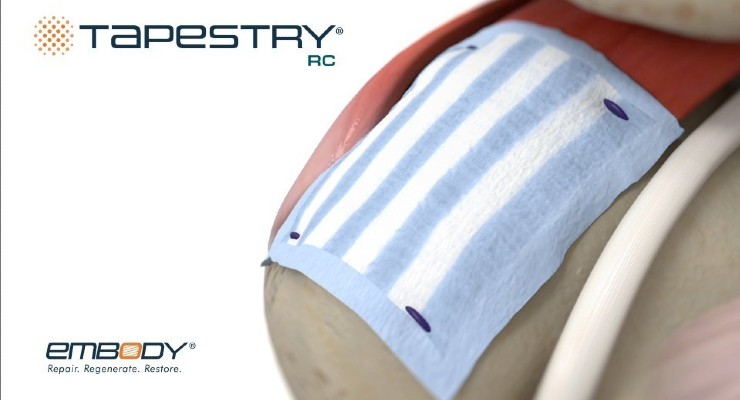
TAPESTRY RC is designed for arthroscopic procedures. It combines the TAPESTRY biointegrative collagen implant’s healing benefits with streamlined delivery and bioabsorbable anchor fixation.
The goal of rotator cuff surgery is repairing the damaged tendon and restoring mobility. However, failure can occur due to inadequate soft tissue healing, resulting in pain, loss of function, and costly reoperations.
"We believe TAPESTRY RC sets a new standard in arthroscopic rotator cuff surgery," Embody CEO Jeff Conroy told the press. "Building on the clinical utility of TAPESTRY, now in a streamlined, all-in-one solution for arthroscopic rotator cuff surgery."
TAPESTRY RC’s arthroscopic introducer is pre-loaded with the implant. The fully bioabsorbable fixation anchors are the first of their kind cleared for tendon and bone fixation in a pre-loaded anchor inserter, according to the company.
"There remains an important clinical need to further improve upon the current healing rates in the treatment of rotator cuff disease," said Nicholas Sgaglione, MD, senior VP and executive director of Northwell Health Orthopaedic Institute and Chair and Professor of orthopedic surgery at Northwell Health. "Embody's TAPESTRY RC System allows for efficient and versatile delivery in a fully bioabsorbable solution to biologic augmentation of the rotator cuff in arthroscopic procedures."
"Historical focus has been on the mechanical repair of tendon and ligaments, not the biologic augmentation," said Dr. Wasik Ashraf, Director of Sport Medicine at St. Luke's Cornwall Hospital. "TAPESTRY has enabled me to bring the power of biostimulative collagen to improve healing of my patients, now in a quick and easy solution to address rotator cuff disease."
The company aims to extend its postmarket studies in shoulder arthroplasty, foot and ankle, and gluteus medius repair to further include arthroscopic rotator cuff repair.
TAPESTRY RC is designed for arthroscopic procedures. It combines the TAPESTRY biointegrative collagen implant’s healing benefits with streamlined delivery and bioabsorbable anchor fixation.
The goal of rotator cuff surgery is repairing the damaged tendon and restoring mobility. However, failure can occur due to inadequate soft tissue healing, resulting in pain, loss of function, and costly reoperations.
"We believe TAPESTRY RC sets a new standard in arthroscopic rotator cuff surgery," Embody CEO Jeff Conroy told the press. "Building on the clinical utility of TAPESTRY, now in a streamlined, all-in-one solution for arthroscopic rotator cuff surgery."
TAPESTRY RC’s arthroscopic introducer is pre-loaded with the implant. The fully bioabsorbable fixation anchors are the first of their kind cleared for tendon and bone fixation in a pre-loaded anchor inserter, according to the company.
"There remains an important clinical need to further improve upon the current healing rates in the treatment of rotator cuff disease," said Nicholas Sgaglione, MD, senior VP and executive director of Northwell Health Orthopaedic Institute and Chair and Professor of orthopedic surgery at Northwell Health. "Embody's TAPESTRY RC System allows for efficient and versatile delivery in a fully bioabsorbable solution to biologic augmentation of the rotator cuff in arthroscopic procedures."
"Historical focus has been on the mechanical repair of tendon and ligaments, not the biologic augmentation," said Dr. Wasik Ashraf, Director of Sport Medicine at St. Luke's Cornwall Hospital. "TAPESTRY has enabled me to bring the power of biostimulative collagen to improve healing of my patients, now in a quick and easy solution to address rotator cuff disease."
The company aims to extend its postmarket studies in shoulder arthroplasty, foot and ankle, and gluteus medius repair to further include arthroscopic rotator cuff repair.