Sam Brusco, Associate Editor10.05.22
SpineX has published the first in human study of its SCiP (Spinal Cord Innovation in Pediatrics) non-invasive spinal neuromodulation in children with cerebral palsy in the journal Nature Communications.
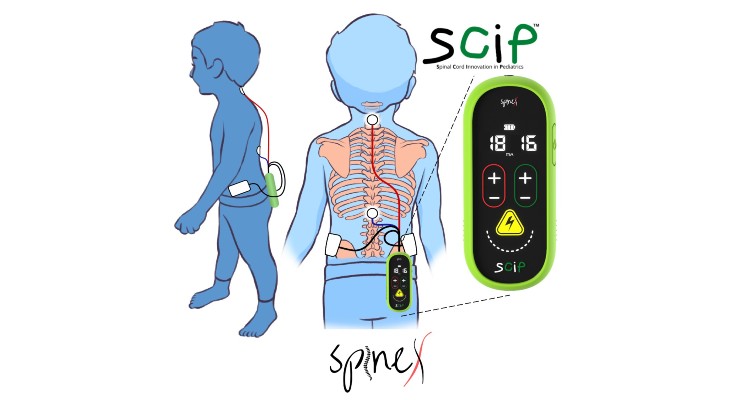
SCiP provides transcutaneous spinal cord neurostimulation to potentially treat underlying neurological dysfunction in pediatric cerebral palsy patients. Spine hopes for SCiP to be the first U.S. medical device to treat cerebral palsy by transforming the brain and spinal cord’s dysfunctional connectivity into highly functional systems.
The study showed how using SCiP during physical therapy improved voluntary sensorimotor function in 16 out of 16 children over a range of ages and cerebral palsy severities. SpineX is also engaged with the U.S. Food and Drug Administration (FDA) for alignment on a proposed clinical trial in 2023. The results of those are anticipated to lead to FDA clearance of SCiP for cerebral palsy treatment.
“At just three years old and highly affected by CP, our son has shown such exciting progress since he began using SCiP,” Dana, the mother of a young boy diagnosed with cerebral palsy who was enrolled in a clinical study sponsored by SpineX, told the press. “We’re so grateful that our son was included in the clinical study with SCiP as it has opened up a world of possibilities for our son, and we hope others will be able to witness it soon.”
SCiP provides transcutaneous spinal cord neurostimulation to potentially treat underlying neurological dysfunction in pediatric cerebral palsy patients. Spine hopes for SCiP to be the first U.S. medical device to treat cerebral palsy by transforming the brain and spinal cord’s dysfunctional connectivity into highly functional systems.
The study showed how using SCiP during physical therapy improved voluntary sensorimotor function in 16 out of 16 children over a range of ages and cerebral palsy severities. SpineX is also engaged with the U.S. Food and Drug Administration (FDA) for alignment on a proposed clinical trial in 2023. The results of those are anticipated to lead to FDA clearance of SCiP for cerebral palsy treatment.
“At just three years old and highly affected by CP, our son has shown such exciting progress since he began using SCiP,” Dana, the mother of a young boy diagnosed with cerebral palsy who was enrolled in a clinical study sponsored by SpineX, told the press. “We’re so grateful that our son was included in the clinical study with SCiP as it has opened up a world of possibilities for our son, and we hope others will be able to witness it soon.”