Sam Brusco, Associate Editor04.21.23
Centinel Spine has completed the 1,000th surgery using its latest U.S. Food and Drug Administration (FDA)-approved total disc replacement (TDR) system of prodisc cervical solutions, prodisc C Vivo and prodisc C SK.
This milestone passes two months after Centinel Spine completed 500 procedures with the systems. The limited launch of prodisc C Vivo and prodisc C SK began in September 2022 and procedure volume has grown exponentially according to the company—an increase of over 60% in Q1 2023 vs. Q4 2022.
Spine surgeon Bradley Duhon, MD, of Lone Tree, Colo. told the press, "prodisc C Vivo comes with the trusted reputation of its Centinel Spine predecessors, but now with much easier implantation. It has become my 'go-to' disc replacement because it comes with very little bone work or prep. This not only makes for easier implantation, but also less endplate disruption, less bleeding, and less potential for heterotopic ossification."
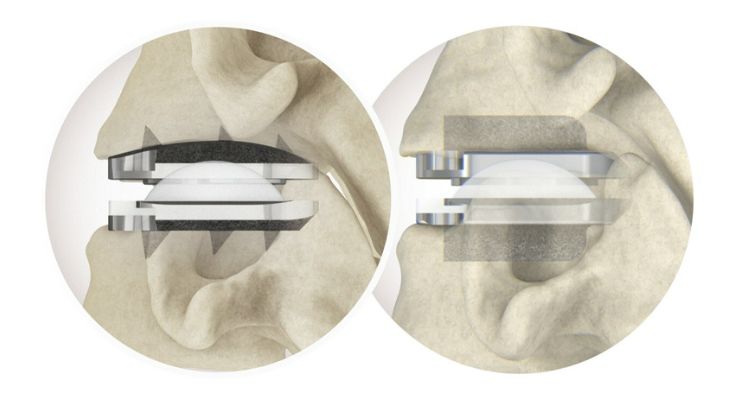
prodisc C Vivo has been in use since 2009 and according to Centinel Spine is one of the most frequently implanted TDR devices in the world. It has keel-less fixation and an anatomically-design superior endplate with lateral spikes.
prodisc C SK has a flat endplate to optimize implant positioning and a low-profile central keel for a streamlined keel preparation technique.
"The Match-the-Disc system of prodisc C Vivo and prodisc C SK, along with the original prodisc C, is exhibiting exponential growth," said Centinel Spine CEO Steve Murray. "With the addition of more instrument sets throughout the year, we will strive to support the unprecedented demand for this unique, paradigm-shifting total disc replacement system."
Centinel Spine is the only company with FDA approval for both cervical and lumbar TDR systems.
This milestone passes two months after Centinel Spine completed 500 procedures with the systems. The limited launch of prodisc C Vivo and prodisc C SK began in September 2022 and procedure volume has grown exponentially according to the company—an increase of over 60% in Q1 2023 vs. Q4 2022.
Spine surgeon Bradley Duhon, MD, of Lone Tree, Colo. told the press, "prodisc C Vivo comes with the trusted reputation of its Centinel Spine predecessors, but now with much easier implantation. It has become my 'go-to' disc replacement because it comes with very little bone work or prep. This not only makes for easier implantation, but also less endplate disruption, less bleeding, and less potential for heterotopic ossification."
prodisc C Vivo has been in use since 2009 and according to Centinel Spine is one of the most frequently implanted TDR devices in the world. It has keel-less fixation and an anatomically-design superior endplate with lateral spikes.
prodisc C SK has a flat endplate to optimize implant positioning and a low-profile central keel for a streamlined keel preparation technique.
"The Match-the-Disc system of prodisc C Vivo and prodisc C SK, along with the original prodisc C, is exhibiting exponential growth," said Centinel Spine CEO Steve Murray. "With the addition of more instrument sets throughout the year, we will strive to support the unprecedented demand for this unique, paradigm-shifting total disc replacement system."
Centinel Spine is the only company with FDA approval for both cervical and lumbar TDR systems.