Michael Barbella, Managing Editor06.30.23
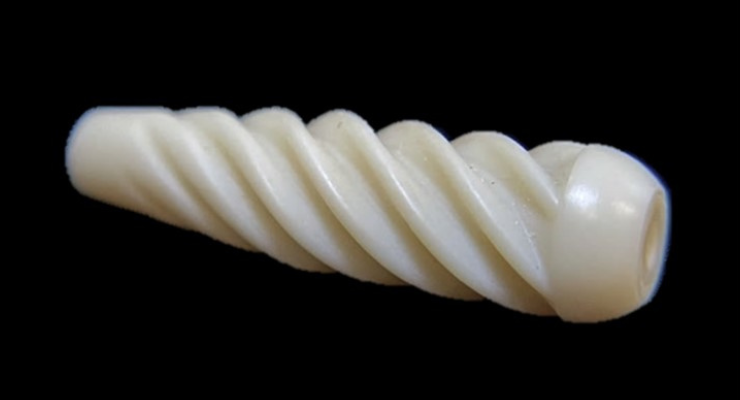
Acuitive Technologies Inc. announced the first surgical use of its Citrelock ACL Tendon Fixation Device. The new product provides surgeons a differentiated design through a tendon-friendly spiral thread featuring a next-generation resorbable technology, known as Citregen, that has unique molecular and mechanical properties for orthopedic surgical applications.
Robin Gehrmann M.D., president of the New Jersey Orthopedic Society and clinical assistant professor of Orthopedics, Rutgers-New Jersey Medical School, successfully implanted the first Citrelock ACL Tendon Fixation Device on June 29. “By leveraging Citregen’s unique material properties, the Citrelock Tendon Fixation Device introduces design features to ease insertion and secure fixation without damaging the tendon during placement,” Gehrmann said. “It has been exciting to participate in the development of a completely novel biomaterial for use in orthopedic applications. I believe this unique material technology will improve tendon fixation as it participates in the natural healing process while avoiding chronic inflammation seen with other biodegradable devices.”
The Citrelock ACL Tendon Fixation Device offers surgeons:
Acuitive Technologies develops biomaterials and orthopedic devices that engage the natural healing process of musculoskeletal tissues while addressing the shortcomings of current biomaterials. Together with its surgeon customers, Acuitive is driven to make healthcare outcomes better for patients.
Robin Gehrmann M.D., president of the New Jersey Orthopedic Society and clinical assistant professor of Orthopedics, Rutgers-New Jersey Medical School, successfully implanted the first Citrelock ACL Tendon Fixation Device on June 29. “By leveraging Citregen’s unique material properties, the Citrelock Tendon Fixation Device introduces design features to ease insertion and secure fixation without damaging the tendon during placement,” Gehrmann said. “It has been exciting to participate in the development of a completely novel biomaterial for use in orthopedic applications. I believe this unique material technology will improve tendon fixation as it participates in the natural healing process while avoiding chronic inflammation seen with other biodegradable devices.”
The Citrelock ACL Tendon Fixation Device offers surgeons:
- Biomimetic material composition to match the native bone inorganic content
- A soft spiral thread design to avoid tendon laceration
- Compressive strength similar to cortical bone and a modulus similar to trabecular bone
- Elastomeric properties for improved pull-out strength
- A controlled resorption rate to mitigate chronic inflammation
Acuitive Technologies develops biomaterials and orthopedic devices that engage the natural healing process of musculoskeletal tissues while addressing the shortcomings of current biomaterials. Together with its surgeon customers, Acuitive is driven to make healthcare outcomes better for patients.