Charles Sternberg, Associate Editor07.26.23
Total Joint Orthopedics has completed its first revision total knee arthroplasty using the Klassic Knee Femoral Cemented Stem.
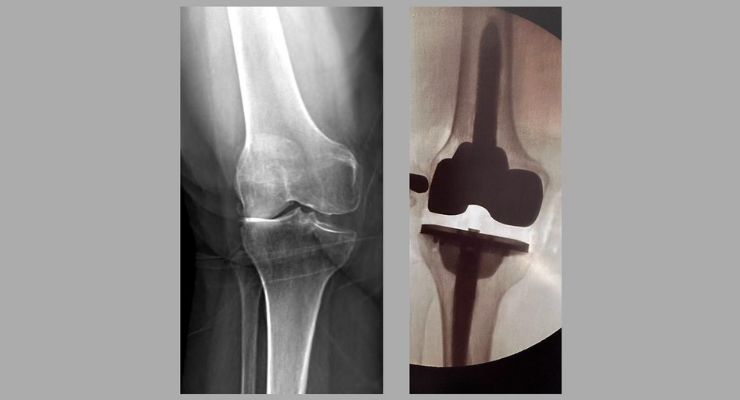
510(k) market clearance for the Klassic Knee Cemented Femoral Stem was received from the U.S. Food and Drug Administration on May 19, 2023. The clearance also includes approval for designs of femoral augments, tibial augments, and revision cones, signaling the first step for TJO to enter the revision knee market.
“With more complex cases being done in the outpatient setting, we set out to expand our primary system to allow surgeons the confidence for any patient while still realizing the value of an efficient system. With just one additional tray, a standard cruciate retaining primary knee can be converted to a constrained, stemmed construct with minimal inventory on hand, making this the ideal solution for an ASC,” said Erin Hofmann, TJO’s CEO.
The clearance of the Femoral Cemented Stem represents TJO’s first step in developing a revision system designed for the outpatient setting, optimizing surgical efficiency. Utilizing streamlined instrumentation facilitates efficient surgical workflows, thus reducing procedure time and minimizing cognitive load. The Klassic Knee System’s advanced implant portfolio offers a wide range of options to suit varying patient anatomies to ensure optimal fit, stability and long-term performance.
"This was the perfect solution for a bad valgus knee and will definitely be in my lineup for most revision cases. As with most of TJO’s lineup, efficiency and simplicity are at the forefront. It fits well into my normal surgical workflow, and even putting in my first one was not a stressful experience,” said Jonathan Peterson, MD, of Hofmann Arthritis Institute in Salt Lake City, Utah.
510(k) market clearance for the Klassic Knee Cemented Femoral Stem was received from the U.S. Food and Drug Administration on May 19, 2023. The clearance also includes approval for designs of femoral augments, tibial augments, and revision cones, signaling the first step for TJO to enter the revision knee market.
“With more complex cases being done in the outpatient setting, we set out to expand our primary system to allow surgeons the confidence for any patient while still realizing the value of an efficient system. With just one additional tray, a standard cruciate retaining primary knee can be converted to a constrained, stemmed construct with minimal inventory on hand, making this the ideal solution for an ASC,” said Erin Hofmann, TJO’s CEO.
The clearance of the Femoral Cemented Stem represents TJO’s first step in developing a revision system designed for the outpatient setting, optimizing surgical efficiency. Utilizing streamlined instrumentation facilitates efficient surgical workflows, thus reducing procedure time and minimizing cognitive load. The Klassic Knee System’s advanced implant portfolio offers a wide range of options to suit varying patient anatomies to ensure optimal fit, stability and long-term performance.
"This was the perfect solution for a bad valgus knee and will definitely be in my lineup for most revision cases. As with most of TJO’s lineup, efficiency and simplicity are at the forefront. It fits well into my normal surgical workflow, and even putting in my first one was not a stressful experience,” said Jonathan Peterson, MD, of Hofmann Arthritis Institute in Salt Lake City, Utah.