Michael Barbella, Managing Editor09.01.23
Relievant Medsystems is touting three-year pooled results from two prospective clinical trials that validate the safety, effectiveness and long-term durability of the Intracept Procedure for patients with vertebrogenic pain. Study results, published in Interventional Pain Medicine, are consistent with previously published long-term data that measured Intracept Procedure outcomes at five years.
The study included 95 patients across 22 study sites who were successfully treated with the Intracept Procedure and completed three-year study visits. These patients achieved statistically significant, clinically meaningful and durable improvements in both pain and function.
At three years post-Intracept Procedure, statistically significant (p<0.0001) improvements of 31.2 points and 4.3 points were observed compared to baseline, for mean Oswestry Disability Index (ODI) and mean numeric pain score (NPS), respectively. The study also found that 74% fewer patients were actively using opioid medications for low back pain and 84% fewer patients required spinal injections for the same pain source and treatment level at three years following treatment.
“These aggregated three-year results demonstrate the sustained effectiveness of basivertebral nerve ablation in improving patients’ pain, productivity and quality of life while decreasing their healthcare utilization,” said Matthew Smuck, M.D., chief of Physical Medicine & Rehabilitation and professor of Orthopaedic Surgery at Stanford University and lead study author. “The data also demonstrate that vertebrogenic pain is a distinct source of anterior column low back pain that is identified using an objective imaging biomarker. The exciting thing about this is that many patients with this diagnosis were previously misdiagnosed with discogenic back pain, a condition with no reliable treatments currently, and they can now be effectively treated when correctly diagnosed with vertebrogenic pain.”
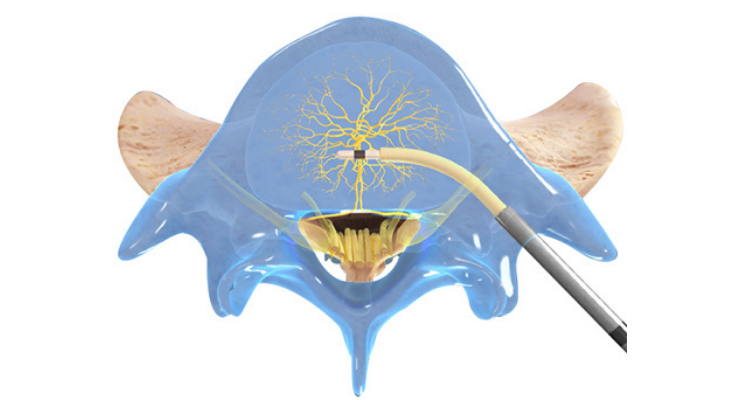
Relievant Medsystems’ minimally invasive Intracept Procedure is the only U.S. Food and Drug Administration-cleared treatment for chronic vertebrogenic low back pain. The same-day, outpatient procedure uses targeted radiofrequency energy to stop the basivertebral nerve (BVN) from transmitting pain signals to the brain and takes approximately one hour to perform.
“We are encouraged to see positive outcomes consistent with results from multiple clinical trials, including two Level I randomized controlled trials, that demonstrate the long-term durability of the Intracept Procedure,” Relievant Medsystems President/CEO Tyler Binney said. “These outcomes represent improved quality of life for patients and we are committed to continuing to provide vertebrogenic pain relief to the millions of individuals with this often debilitating condition.”
Other key findings at three years post-procedure include:
Relievant Medsystems is a commercial-stage medical device company aiming to transform vertebrogenic pain diagnosis and treatment with the Intracept Procedure. The firm is based in Sunnyvale, Calif.
The study included 95 patients across 22 study sites who were successfully treated with the Intracept Procedure and completed three-year study visits. These patients achieved statistically significant, clinically meaningful and durable improvements in both pain and function.
At three years post-Intracept Procedure, statistically significant (p<0.0001) improvements of 31.2 points and 4.3 points were observed compared to baseline, for mean Oswestry Disability Index (ODI) and mean numeric pain score (NPS), respectively. The study also found that 74% fewer patients were actively using opioid medications for low back pain and 84% fewer patients required spinal injections for the same pain source and treatment level at three years following treatment.
“These aggregated three-year results demonstrate the sustained effectiveness of basivertebral nerve ablation in improving patients’ pain, productivity and quality of life while decreasing their healthcare utilization,” said Matthew Smuck, M.D., chief of Physical Medicine & Rehabilitation and professor of Orthopaedic Surgery at Stanford University and lead study author. “The data also demonstrate that vertebrogenic pain is a distinct source of anterior column low back pain that is identified using an objective imaging biomarker. The exciting thing about this is that many patients with this diagnosis were previously misdiagnosed with discogenic back pain, a condition with no reliable treatments currently, and they can now be effectively treated when correctly diagnosed with vertebrogenic pain.”
Relievant Medsystems’ minimally invasive Intracept Procedure is the only U.S. Food and Drug Administration-cleared treatment for chronic vertebrogenic low back pain. The same-day, outpatient procedure uses targeted radiofrequency energy to stop the basivertebral nerve (BVN) from transmitting pain signals to the brain and takes approximately one hour to perform.
“We are encouraged to see positive outcomes consistent with results from multiple clinical trials, including two Level I randomized controlled trials, that demonstrate the long-term durability of the Intracept Procedure,” Relievant Medsystems President/CEO Tyler Binney said. “These outcomes represent improved quality of life for patients and we are committed to continuing to provide vertebrogenic pain relief to the millions of individuals with this often debilitating condition.”
Other key findings at three years post-procedure include:
- 26.3% of patients reported being completely pain-free
- Seventy-one percent of patients indicated they had returned to the level of activity they enjoyed before experiencing chronic low back pain
- Eighty-six percent of patients indicated they would have the procedure again for the same condition
- No serious device or device-procedure related adverse events were reported, highlighting the safety of this procedure
Relievant Medsystems is a commercial-stage medical device company aiming to transform vertebrogenic pain diagnosis and treatment with the Intracept Procedure. The firm is based in Sunnyvale, Calif.