Michael Barbella, Managing Editor10.24.23
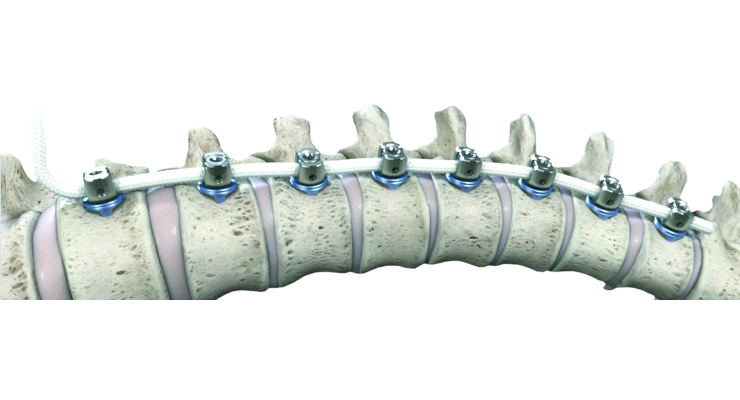
ZimVie Inc.'s Tether Vertebral Body Tethering System has been used to treat more than 2,000 adolescent idiopathic scoliosis patients worldwide, the company announced today.
The Tether was approved as a Humanitarian Device by the U.S. Food and Drug Administration in 2019, based on more than seven years of clinical data. Over 50 surgeons have performed VBT using the Tether system to treat adolescent idiopathic scoliosis patients.
“I have been in practice dedicating my career to the treatment of patients with scoliosis for 28 years. The Tether has been an amazing advance that has allowed my patients to have correction of their scoliosis while avoiding a fusion, with a faster return to sports and other activities. It has been a game changer,” said Dr. Baron Lonner, chief of Minimally Invasive Scoliosis Surgery and Pediatric Spine at Mount Sinai Hospital and professor of Orthopaedic Surgery at Icahn School of Medicine in New York, N.Y.
The Tether is a Humanitarian Device authorized by federal law for use in treating pediatric patients to correct progressive idiopathic scoliosis, with a major Cobb angle of 30 to 65 degrees whose osseous structure is dimensionally adequate to accommodate screw fixation, as determined by radiographic imaging. Patients should have failed bracing and/or be intolerant to brace wear. Post-operative risks include inadequate curve correction, overcorrection of the instrumented curve, cord breakage, and bone screw migration.
“Having brought this technology to over 2,000 children is a milestone that serves as a reminder of our commitment to put patients first, especially those for whom our products can have such profound and prolonged impact,” ZimVie Spine Global President Rebecca Whitney said. “The Tether is an important and inspirational part of our motion preservation portfolio. We remain dedicated to developing the market for vertebral body tethering and restoring daily life for this special group of patients.”
ZimVie develops, manufactures, and delivers products and solutions designed to treat spine pathologies and support dental tooth replacement and restoration procedures. The company was founded in March 2022 as an independent, publicly traded spin-off of the Dental and Spine business units of Zimmer Biomet to breathe new life, dedicated energy, and strategic focus to its portfolio of trusted brands and products. From its headquarters in Westminster, Colo., and additional facilities worldwide, the company serves customers in more than 70 countries with dental and spine solutions including differentiated product platforms supported by extensive clinical evidence.
The Tether was approved as a Humanitarian Device by the U.S. Food and Drug Administration in 2019, based on more than seven years of clinical data. Over 50 surgeons have performed VBT using the Tether system to treat adolescent idiopathic scoliosis patients.
“I have been in practice dedicating my career to the treatment of patients with scoliosis for 28 years. The Tether has been an amazing advance that has allowed my patients to have correction of their scoliosis while avoiding a fusion, with a faster return to sports and other activities. It has been a game changer,” said Dr. Baron Lonner, chief of Minimally Invasive Scoliosis Surgery and Pediatric Spine at Mount Sinai Hospital and professor of Orthopaedic Surgery at Icahn School of Medicine in New York, N.Y.
The Tether is a Humanitarian Device authorized by federal law for use in treating pediatric patients to correct progressive idiopathic scoliosis, with a major Cobb angle of 30 to 65 degrees whose osseous structure is dimensionally adequate to accommodate screw fixation, as determined by radiographic imaging. Patients should have failed bracing and/or be intolerant to brace wear. Post-operative risks include inadequate curve correction, overcorrection of the instrumented curve, cord breakage, and bone screw migration.
“Having brought this technology to over 2,000 children is a milestone that serves as a reminder of our commitment to put patients first, especially those for whom our products can have such profound and prolonged impact,” ZimVie Spine Global President Rebecca Whitney said. “The Tether is an important and inspirational part of our motion preservation portfolio. We remain dedicated to developing the market for vertebral body tethering and restoring daily life for this special group of patients.”
ZimVie develops, manufactures, and delivers products and solutions designed to treat spine pathologies and support dental tooth replacement and restoration procedures. The company was founded in March 2022 as an independent, publicly traded spin-off of the Dental and Spine business units of Zimmer Biomet to breathe new life, dedicated energy, and strategic focus to its portfolio of trusted brands and products. From its headquarters in Westminster, Colo., and additional facilities worldwide, the company serves customers in more than 70 countries with dental and spine solutions including differentiated product platforms supported by extensive clinical evidence.