Sam Brusco, Associate Editor10.31.23
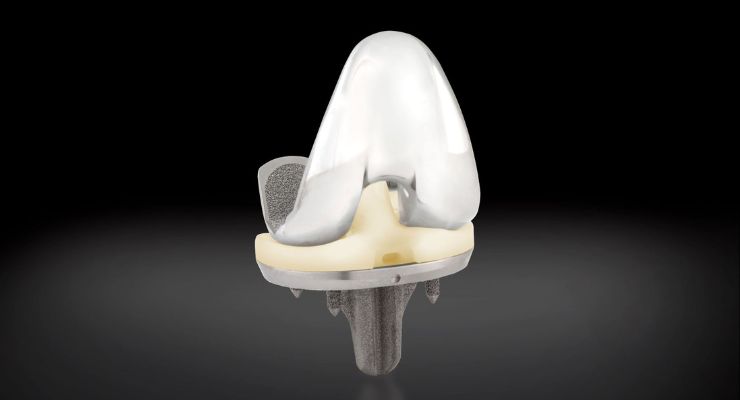
Exactech announced the first successful surgery using its new, advanced Activit-E polyethylene for the Truliant knee replacement system.
Activit-E chemically crosslinked polyethylene provides a balance of material strength and toughness, according to the company. It also eliminates the need for gamma irradiation technology used in past generations. It allows for active oxidative resistance and a long-term polyethylene bearing.
Hany Bedair, M.D. performed the procedure at Massachusetts General Hospital (MGH) in Boston, where the material was developed by renowned polyethylene expert Orhun Muratoglu, Ph.D., director of the Harris Orthopaedic Laboratory at MGH. Dr. Muratoglu was also in attendance for the knee replacement case.
Muratoglu invented the first crosslinked polyethylene, and the first of multiple generations of vitamin E, antioxidant polyethylene for leading orthopedic companies.
“It is a profound moment when you have the inventor who developed our advanced polyethylene in the case, performed at the hospital where it was developed,” Adam Hayden, CMO and SVP, Large Joints Business Unit at Exactech, told the press. “Exactech is extremely proud to offer this next generation of highly crosslinked polyethylene with vitamin E antioxidant.”
Activit-E was recently 510(k) cleared by the U.S. Food and Drug Administration (FDA) for Exactech knee and ankle replacements.
“It is exciting to perform the first surgical procedure with an implant material that was both conceived and developed within our hospital,” said Dr. Bedair. “With this advancement, we anticipate better outcomes compared to knees with traditional, highly crosslinked polyethylene.”
Activit-E chemically crosslinked polyethylene provides a balance of material strength and toughness, according to the company. It also eliminates the need for gamma irradiation technology used in past generations. It allows for active oxidative resistance and a long-term polyethylene bearing.
Hany Bedair, M.D. performed the procedure at Massachusetts General Hospital (MGH) in Boston, where the material was developed by renowned polyethylene expert Orhun Muratoglu, Ph.D., director of the Harris Orthopaedic Laboratory at MGH. Dr. Muratoglu was also in attendance for the knee replacement case.
Muratoglu invented the first crosslinked polyethylene, and the first of multiple generations of vitamin E, antioxidant polyethylene for leading orthopedic companies.
“It is a profound moment when you have the inventor who developed our advanced polyethylene in the case, performed at the hospital where it was developed,” Adam Hayden, CMO and SVP, Large Joints Business Unit at Exactech, told the press. “Exactech is extremely proud to offer this next generation of highly crosslinked polyethylene with vitamin E antioxidant.”
Activit-E was recently 510(k) cleared by the U.S. Food and Drug Administration (FDA) for Exactech knee and ankle replacements.
“It is exciting to perform the first surgical procedure with an implant material that was both conceived and developed within our hospital,” said Dr. Bedair. “With this advancement, we anticipate better outcomes compared to knees with traditional, highly crosslinked polyethylene.”