Michael Barbella, Managing Editor11.29.23
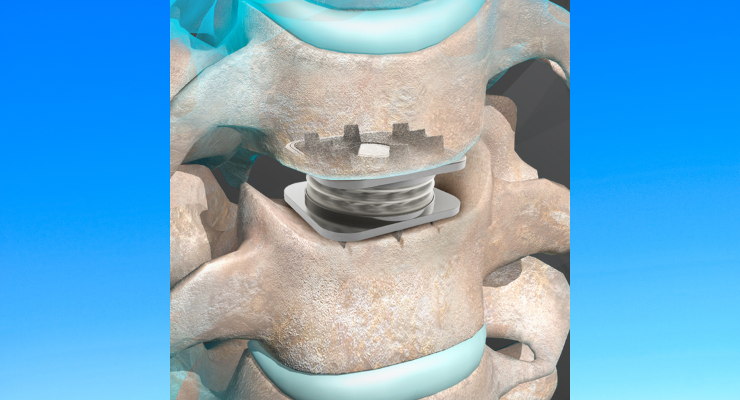
Orthofix Medical Inc. is sharing five-year results from a U.S. clinical study that compares the M6-C artificial cervical disc with anterior cervical discectomy and fusion (ACDF). Published in The Spine Journal, patients treated with the M6-C disc demonstrated superior clinical success at 60 months compared to ACDF patients.
Secondary findings indicated significant improvements in neck and arm pain, function, and quality of life scores. The M6-C patient group maintained the flexion-extension and lateral bending motion reported at earlier time points, according to study results. Publication of this data coincides with a significant milestone for the company: more than 100,000 M6-C artificial cervical disc and M6-L artificial lumbar disc implants (only available outside the United States) have been performed worldwide since the product’s first introduction in 2006.
“Publication of this data is important as it validates the strong clinical performance observed in the five-year data from the U.S. IDE study,” said lead author Dr. Frank Phillips, professor of Orthopedic Surgery at Rush University Medical Center in Chicago and principal investigator for the U.S. Food and Drug Administration (FDA) clinical trial. “Artificial cervical disc replacement is becoming the gold standard of care for indicated patients who may otherwise be facing cervical disc fusion. Data from this study show the M6-C artificial disc demonstrated superior five-year achievement of clinical success when compared to ACDF controls. In addition, significantly more subjects in the M6-C group reported improved pain and physical functioning scores than observed in ACDF subjects, with no difference in re-operation rates or safety outcomes.”
A prospective, non-randomized, concurrently controlled clinical trial, the M6-C IDE study was conducted at 23 U.S. sites with an average patient age of 44 years. The study evaluated the safety and effectiveness of the M6-C artificial cervical disc compared to ACDF for the treatment of single-level symptomatic cervical radiculopathy with or without cord compression. The overall success rate for the protocol-specified primary endpoint for the M6-C disc patients was 82.3% as compared to 67% percent in the control group. The rates of M6-C disc subsequent surgical interventions (SSI) were 3.1%, device or procedure-related serious adverse events (SAE) were 3.1%, and were similar to ACDF rates of SSI at 5.3% and SAE failure equaling 4.8%. The M6-C disc received FDA approval in February 2019 based on the two-year results of this study.
“To date there have been more than 100,000 implantations of M6 artificial disc technology in 20 countries around the world,” Orthofix Global Spine President Kevin Kenny said. “We are proud of this technology that has helped so many people get back to enjoying their lives.”
The M6‑C artificial cervical disc is designed to maintain the natural behavior of a functional spinal unit by replicating the biomechanical characteristics of the native disc and is indicated as an alternative to cervical fusion. The unique design of the M6-C disc features a compressible viscoelastic nuclear core and an annular structure construct that allows for shock absorption at the implanted level, as well as providing a controlled range of motion when the spine transitions in its combined complex movements.
Orthofix is a global spine and orthopedics company with a portfolio of biologics, spinal hardware, bone growth therapies, specialized orthopedic solutions, and a surgical navigation system. Its products are distributed in approximately 68 countries worldwide. The company is headquartered in Lewisville, Texas, where it conducts general business, product development, medical education and manufacturing, and has primary offices in Carlsbad, Calif., with a focus on spine and biologics product innovation and surgeon education, and Verona, Italy, with an emphasis on product innovation, production, and medical education for orthopedics. The combined company’s global R&D, commercial and manufacturing footprint also includes facilities and offices in Irvine, Calif.; Toronto; Sunnyvale, Calif.; Wayne, Pa.; Olive Branch, Miss.; Maidenhead, U.K.; Munich, Germany; Paris; and São Paulo, Brazil.
Secondary findings indicated significant improvements in neck and arm pain, function, and quality of life scores. The M6-C patient group maintained the flexion-extension and lateral bending motion reported at earlier time points, according to study results. Publication of this data coincides with a significant milestone for the company: more than 100,000 M6-C artificial cervical disc and M6-L artificial lumbar disc implants (only available outside the United States) have been performed worldwide since the product’s first introduction in 2006.
“Publication of this data is important as it validates the strong clinical performance observed in the five-year data from the U.S. IDE study,” said lead author Dr. Frank Phillips, professor of Orthopedic Surgery at Rush University Medical Center in Chicago and principal investigator for the U.S. Food and Drug Administration (FDA) clinical trial. “Artificial cervical disc replacement is becoming the gold standard of care for indicated patients who may otherwise be facing cervical disc fusion. Data from this study show the M6-C artificial disc demonstrated superior five-year achievement of clinical success when compared to ACDF controls. In addition, significantly more subjects in the M6-C group reported improved pain and physical functioning scores than observed in ACDF subjects, with no difference in re-operation rates or safety outcomes.”
A prospective, non-randomized, concurrently controlled clinical trial, the M6-C IDE study was conducted at 23 U.S. sites with an average patient age of 44 years. The study evaluated the safety and effectiveness of the M6-C artificial cervical disc compared to ACDF for the treatment of single-level symptomatic cervical radiculopathy with or without cord compression. The overall success rate for the protocol-specified primary endpoint for the M6-C disc patients was 82.3% as compared to 67% percent in the control group. The rates of M6-C disc subsequent surgical interventions (SSI) were 3.1%, device or procedure-related serious adverse events (SAE) were 3.1%, and were similar to ACDF rates of SSI at 5.3% and SAE failure equaling 4.8%. The M6-C disc received FDA approval in February 2019 based on the two-year results of this study.
“To date there have been more than 100,000 implantations of M6 artificial disc technology in 20 countries around the world,” Orthofix Global Spine President Kevin Kenny said. “We are proud of this technology that has helped so many people get back to enjoying their lives.”
The M6‑C artificial cervical disc is designed to maintain the natural behavior of a functional spinal unit by replicating the biomechanical characteristics of the native disc and is indicated as an alternative to cervical fusion. The unique design of the M6-C disc features a compressible viscoelastic nuclear core and an annular structure construct that allows for shock absorption at the implanted level, as well as providing a controlled range of motion when the spine transitions in its combined complex movements.
Orthofix is a global spine and orthopedics company with a portfolio of biologics, spinal hardware, bone growth therapies, specialized orthopedic solutions, and a surgical navigation system. Its products are distributed in approximately 68 countries worldwide. The company is headquartered in Lewisville, Texas, where it conducts general business, product development, medical education and manufacturing, and has primary offices in Carlsbad, Calif., with a focus on spine and biologics product innovation and surgeon education, and Verona, Italy, with an emphasis on product innovation, production, and medical education for orthopedics. The combined company’s global R&D, commercial and manufacturing footprint also includes facilities and offices in Irvine, Calif.; Toronto; Sunnyvale, Calif.; Wayne, Pa.; Olive Branch, Miss.; Maidenhead, U.K.; Munich, Germany; Paris; and São Paulo, Brazil.