Michael Barbella, Managing Editor11.30.23
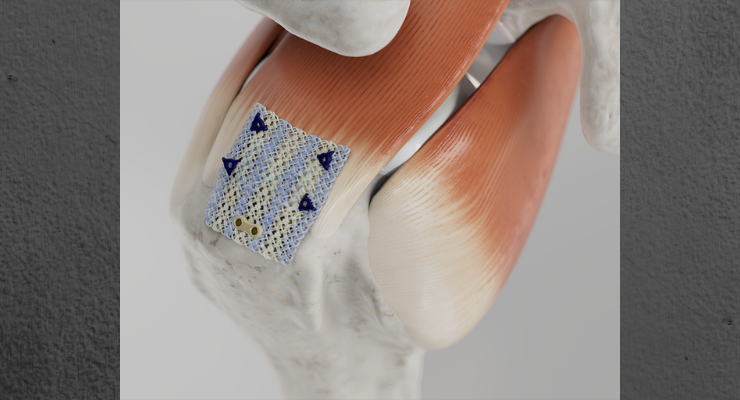
The first surgeries using Anika Therapeutics Inc.'s Integrity Implant System have been successfully carried out by Dr. Christopher Baker at the Florida Orthopaedic Institute in Tampa. Integrity, comprised of a hyaluronic acid-based scaffold with bone and tendon fixation components and single use arthroscopic delivery instruments, is designed to protect an injured tendon and promote healing in rotator cuff repair and other tendon procedures. Fully cleared by the U.S. Food and Drug Administration (FDA) in August, Integrity's O.R. debut kicks off a limited U.S. market release ahead of schedule that will ramp to full commercialization next quarter.
“The successful first surgeries using Anika’s new Integrity Implant System mark another key milestone in the build out of our regenerative product portfolio as we continue to provide differentiated solutions to surgeons for rotator cuff procedures,” Anika Therapeutics President/CEO Cheryl R. Blanchard, Ph.D., said. “The HA-based scaffold, together with the instrumentation and fixation components, provide a seamless, efficient, and elegant rotator cuff repair solution. While having a strong implant at time zero is critically important in rotator cuff repairs, the instrumentation and delivery are just as important, and we believe we have nailed it. The feedback received after the procedure exceeded our expectations highlighting the system’s ease of use and strength of the HA-based scaffold. We believe that Integrity is truly a game changer for surgeons and their patients and look forward to extending this exciting technology to other tendon repairs, for example, in the foot and ankle.”
The Integrity implant is a flexible, knitted, HA-based scaffold that provides improved dry and wet strength and regenerative capacity over first generation collagen patches,1 and supports regenerative healing through improved cell infiltration,1 tissue remodeling,1 and tendon thickening.1 In an independent head-to-head animal study1 comparing Anika’s Integrity system and the market-leading collagen implant, fibroblast infiltration and regularly oriented new collagenous tissue formation had occurred within the Integrity repair, demonstrating greater regenerative capacity as early as 12 weeks post-implantation. At 26 weeks, within the resorbing Integrity structure, new collagenous tissue infiltration had occurred, forming a new network of tendon tissue. This resulted in an average repaired tendon thickness nearly three times greater than with the market-leading collagen device. The scaffold component of the Integrity system is a porous, knitted, flexible construct combining Anika’s proprietary HYAFF fibers with polyethylene terephthalate (PET) and is designed to support cell infiltration and regenerative healing. Integrity is inherently strong and can be confidently manipulated arthroscopically, which offers a truly unique and differentiated solution for shoulder surgeons to treat rotator cuff tears. Integrity is fixated using PEEK bone staples, resorbable PLGA soft tissue tendon tacks or suture fixation, as desired, at the site of the rotator cuff repair. The fixation components and instrumentation are delivered single-use and sterile for added efficiency.
“The hybrid, multifilament structure provides superior implant handling and strength when compared to collagen-based products alone. This combination of materials is comprised of 80% HYAFF, a hyaluronic acid-based material in clinical use for more than 20 years, and 20% PET fiber, i.e. surgical suture, which also has a long and established clinical history," noted Christopher Baker, M.D., of the Florida Orthopaedic Institute. "The material and knitted structure result in a compelling blend of strength and healing that stands out from other products. The suture material remaining after full resorption of the HYAFF component is less than 30% of the total amount of suture used in a typical double-row repair. Handling of the implant has met every expectation that I had for this new device.”
The U.S. rotator cuff augmentation market is currently worth more than $150 million2 and is estimated to grow at a nearly 7% annually2 over the next five years, representing one of the high opportunity spaces in orthopedics. Integrity's launch continues Anika’s release of differentiated products to support shoulder surgeons and a portfolio built around rotator cuff disease. Integrity, combined with recent product launches including X-Twist Fixation System, RevoMotion Reverse Total Arthroplasty System, and Tactoset Injectable Bone Substitute for hardware augmentation, provide a comprehensive rotator cuff solution portfolio.
“Integrity’s instrumentation, delivery and surgical technique are a game-changer. Securing the implant laterally first helps ensure proper coverage across the repair site and the rolling deployment tool provides consistent and repeatable implant placement,” stated Timothy Codd, M.D., of the University of Maryland St. Joseph Medical Center.
Anika Therapeutics Inc. creates and delivers advancements in early intervention orthopedic care. Leveraging its core expertise in hyaluronic acid and implant solutions, the company partners with clinicians to provide minimally invasive products that restore active living for people worldwide. Its focus is on high opportunity spaces within orthopedics, including osteoarthritis pain management, regenerative solutions, sports medicine, and arthrosurface joint solutions, and Anika's products are delivered in key sites of care, including ambulatory surgery centers. The firm is headquartered outside Boston.
References
1 Data on File
2 2023 SmartTRAK
“The successful first surgeries using Anika’s new Integrity Implant System mark another key milestone in the build out of our regenerative product portfolio as we continue to provide differentiated solutions to surgeons for rotator cuff procedures,” Anika Therapeutics President/CEO Cheryl R. Blanchard, Ph.D., said. “The HA-based scaffold, together with the instrumentation and fixation components, provide a seamless, efficient, and elegant rotator cuff repair solution. While having a strong implant at time zero is critically important in rotator cuff repairs, the instrumentation and delivery are just as important, and we believe we have nailed it. The feedback received after the procedure exceeded our expectations highlighting the system’s ease of use and strength of the HA-based scaffold. We believe that Integrity is truly a game changer for surgeons and their patients and look forward to extending this exciting technology to other tendon repairs, for example, in the foot and ankle.”
The Integrity implant is a flexible, knitted, HA-based scaffold that provides improved dry and wet strength and regenerative capacity over first generation collagen patches,1 and supports regenerative healing through improved cell infiltration,1 tissue remodeling,1 and tendon thickening.1 In an independent head-to-head animal study1 comparing Anika’s Integrity system and the market-leading collagen implant, fibroblast infiltration and regularly oriented new collagenous tissue formation had occurred within the Integrity repair, demonstrating greater regenerative capacity as early as 12 weeks post-implantation. At 26 weeks, within the resorbing Integrity structure, new collagenous tissue infiltration had occurred, forming a new network of tendon tissue. This resulted in an average repaired tendon thickness nearly three times greater than with the market-leading collagen device. The scaffold component of the Integrity system is a porous, knitted, flexible construct combining Anika’s proprietary HYAFF fibers with polyethylene terephthalate (PET) and is designed to support cell infiltration and regenerative healing. Integrity is inherently strong and can be confidently manipulated arthroscopically, which offers a truly unique and differentiated solution for shoulder surgeons to treat rotator cuff tears. Integrity is fixated using PEEK bone staples, resorbable PLGA soft tissue tendon tacks or suture fixation, as desired, at the site of the rotator cuff repair. The fixation components and instrumentation are delivered single-use and sterile for added efficiency.
“The hybrid, multifilament structure provides superior implant handling and strength when compared to collagen-based products alone. This combination of materials is comprised of 80% HYAFF, a hyaluronic acid-based material in clinical use for more than 20 years, and 20% PET fiber, i.e. surgical suture, which also has a long and established clinical history," noted Christopher Baker, M.D., of the Florida Orthopaedic Institute. "The material and knitted structure result in a compelling blend of strength and healing that stands out from other products. The suture material remaining after full resorption of the HYAFF component is less than 30% of the total amount of suture used in a typical double-row repair. Handling of the implant has met every expectation that I had for this new device.”
The U.S. rotator cuff augmentation market is currently worth more than $150 million2 and is estimated to grow at a nearly 7% annually2 over the next five years, representing one of the high opportunity spaces in orthopedics. Integrity's launch continues Anika’s release of differentiated products to support shoulder surgeons and a portfolio built around rotator cuff disease. Integrity, combined with recent product launches including X-Twist Fixation System, RevoMotion Reverse Total Arthroplasty System, and Tactoset Injectable Bone Substitute for hardware augmentation, provide a comprehensive rotator cuff solution portfolio.
“Integrity’s instrumentation, delivery and surgical technique are a game-changer. Securing the implant laterally first helps ensure proper coverage across the repair site and the rolling deployment tool provides consistent and repeatable implant placement,” stated Timothy Codd, M.D., of the University of Maryland St. Joseph Medical Center.
Anika Therapeutics Inc. creates and delivers advancements in early intervention orthopedic care. Leveraging its core expertise in hyaluronic acid and implant solutions, the company partners with clinicians to provide minimally invasive products that restore active living for people worldwide. Its focus is on high opportunity spaces within orthopedics, including osteoarthritis pain management, regenerative solutions, sports medicine, and arthrosurface joint solutions, and Anika's products are delivered in key sites of care, including ambulatory surgery centers. The firm is headquartered outside Boston.
References
1 Data on File
2 2023 SmartTRAK