Michael Barbella, Managing Editor02.07.24
AddUp Inc. and Anatomic Implants are working together to submit a 510(k) application to the U.S. Food and Drug Administration (FDA) for the world’s first 3D printed toe joint replacement. The product will be showcased next week at the American Academy of Orthopaedic Surgeons (AAOS) 2024 Annual Meeting in San Francisco, Calif. (Feb. 13-15).
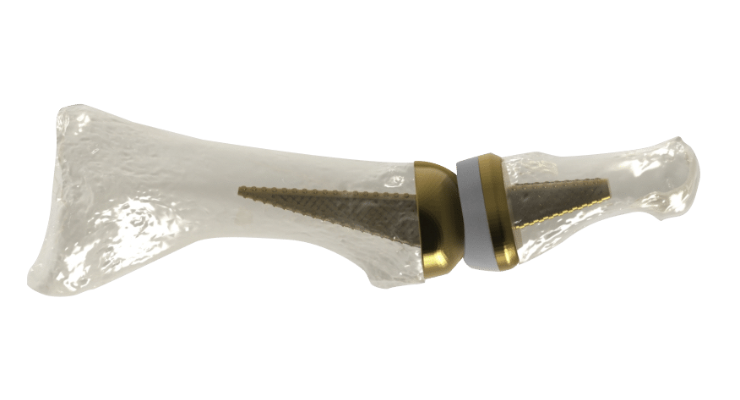
Washington, D.C.-based Anatomic Implants claims to be the first medical device startup company to patent and develop a first metatarsophalangeal (MTP) joint replacement that nearly perfectly replicates the human anatomy via titanium 3D printing technology. The MTP toe joint is located at the base of the big toe and is one of the three main points used for balance. It is often the first joint in the foot to develop osteoarthritis.
The Anatomic Great Toe Joint
To bring its product to market, Anatomic Implants has chosen AddUp’s FormUp 350 Powder Bed Fusion (PBF) machine to qualify the implant for submission to the FDA. The FormUp 350 can produce varying complex geometries with fine detailed lattice structures—ideal for implantable medical devices. The FDA’s Center for Devices and Radiological Health (CDRH) and the Center for Drug Evaluation and Research (CDER) has approved many 3D printed class II medical devices through the 510(k) pathway for more than 15 years now.
“With first MTP joint replacement being a largely underserved market, and medical device companies building lattice structures into implantables since the mid 2000s, Dr. Nutter and I sought out to make a more anatomic design by leveraging the latest technologies adopted by the industry and the FDA,” Anatomic Implants President David Nutter said. “We were excited to partner with AddUp to achieve 510(k) clearance after learning about their proprietary 3D printing technology and seeing how it could benefit the development of the Anatomic Great Toe Joint. We look forward to leveraging the AddUp team and their expertise to validate the world’s first 3D printed toe joint replacement on their FormUp 350.”
The 510(k) clearance process involves a comprehensive review of safety and performance data for the implant to determine if it is substantially equivalent to an implant that is already on the market. Several tests have already completed and 510(k) clearance is anticipated later in the third quarter this year. Anatomic Implants has been working on the Anatomic Great Toe Joint since its inception late 2016 and has already secured design patents in the United States, Canada, and throughout Europe, which represents the majority of the global 1st MTP joint reconstruction market.
The global first MTP joint reconstruction market is estimated to be worth more than $500 million annually and is currently underserved with very few products. The few devices that do exist do not have the potential to support bone-in growth as well as the Anatomic Great Toe Joint, according to Anatomic Implants. Only through the use of additive manufacturing can a porous structure be integrated into the design to promote osseointegration, which increases implants' chances of bonding to the bone and reducing rejection by the body. This leads to better patient outcomes after surgery.
AddUp has vast experience in the medical industry with global OEMs relying on the FormUp 350 for serial production of their medical implants. The company’s North American subsidiary, The AddUp Solution Center, is located in Cincinnati, Ohio, and is ISO13485 certified. AddUp has experience partnering with various medical customers and supporting their path to FDA clearance.
“AddUp is committed to supporting the development of cutting-edge solutions for the medical market,” AddUp Deputy CEO Nick Estock stated. “Our team at the AddUp Solution Center has the expertise on FDA regulations and qualification protocols to provide a proactive approach to regulatory compliance essential for a successful 510(k) submission. We are excited to be supporting Anatomic Implants through this process to bring the first additively manufactured toe joint replacement to market.”
Washington, D.C.-based Anatomic Implants claims to be the first medical device startup company to patent and develop a first metatarsophalangeal (MTP) joint replacement that nearly perfectly replicates the human anatomy via titanium 3D printing technology. The MTP toe joint is located at the base of the big toe and is one of the three main points used for balance. It is often the first joint in the foot to develop osteoarthritis.
The Anatomic Great Toe Joint
To bring its product to market, Anatomic Implants has chosen AddUp’s FormUp 350 Powder Bed Fusion (PBF) machine to qualify the implant for submission to the FDA. The FormUp 350 can produce varying complex geometries with fine detailed lattice structures—ideal for implantable medical devices. The FDA’s Center for Devices and Radiological Health (CDRH) and the Center for Drug Evaluation and Research (CDER) has approved many 3D printed class II medical devices through the 510(k) pathway for more than 15 years now.
“With first MTP joint replacement being a largely underserved market, and medical device companies building lattice structures into implantables since the mid 2000s, Dr. Nutter and I sought out to make a more anatomic design by leveraging the latest technologies adopted by the industry and the FDA,” Anatomic Implants President David Nutter said. “We were excited to partner with AddUp to achieve 510(k) clearance after learning about their proprietary 3D printing technology and seeing how it could benefit the development of the Anatomic Great Toe Joint. We look forward to leveraging the AddUp team and their expertise to validate the world’s first 3D printed toe joint replacement on their FormUp 350.”
The 510(k) clearance process involves a comprehensive review of safety and performance data for the implant to determine if it is substantially equivalent to an implant that is already on the market. Several tests have already completed and 510(k) clearance is anticipated later in the third quarter this year. Anatomic Implants has been working on the Anatomic Great Toe Joint since its inception late 2016 and has already secured design patents in the United States, Canada, and throughout Europe, which represents the majority of the global 1st MTP joint reconstruction market.
The global first MTP joint reconstruction market is estimated to be worth more than $500 million annually and is currently underserved with very few products. The few devices that do exist do not have the potential to support bone-in growth as well as the Anatomic Great Toe Joint, according to Anatomic Implants. Only through the use of additive manufacturing can a porous structure be integrated into the design to promote osseointegration, which increases implants' chances of bonding to the bone and reducing rejection by the body. This leads to better patient outcomes after surgery.
AddUp has vast experience in the medical industry with global OEMs relying on the FormUp 350 for serial production of their medical implants. The company’s North American subsidiary, The AddUp Solution Center, is located in Cincinnati, Ohio, and is ISO13485 certified. AddUp has experience partnering with various medical customers and supporting their path to FDA clearance.
“AddUp is committed to supporting the development of cutting-edge solutions for the medical market,” AddUp Deputy CEO Nick Estock stated. “Our team at the AddUp Solution Center has the expertise on FDA regulations and qualification protocols to provide a proactive approach to regulatory compliance essential for a successful 510(k) submission. We are excited to be supporting Anatomic Implants through this process to bring the first additively manufactured toe joint replacement to market.”