Michael Barbella, Managing Editor05.29.18
Maybe it’s true. Maybe there’s some credibility to all that talk about hallowed ground and the fairy tale-like ambience of the Augusta National Golf Club.
It certainly would explain a lot of things: It would, for example, lend credence to the gold dust-spouting natural spring purportedly nestled between the 13th and 14th fairways, and the exceptionally low concessions prices ($1.50/pimento cheese sandwich, $1/chips, $4/beer). It also would legitimize a handful of hole names—specifically, Nos. 11, 12, 13 (Amen Corner), No. 10 (Camillia, a Latin moniker meaning “helper to the priest”), and No. 17 (Nandina, a plant more commonly known as “heavenly” or “sacred bamboo”).
“If there’s a golf course in heaven,” three-time Masters Tournament winner Gary Player once told Reuters, “I hope it’s like Augusta National.”
Tony Finau might say the two are one in the same. During the Masters’ 2018 Par-3 contest in early April, the 28-year-old golfer injured his left ankle celebrating a hole-in-one but rebounded the next day to shoot a four-under-par 68 in the championship game’s opening round. He briefly led the tournament before slipping behind three-time major winner Jordan Spieth to tie for second place with Matthew Kuchar.
“It’s nothing short of a miracle if you ask me,” Finau said of his first-day Masters performance.
Miraculous so it seemed: After rolling his ankle and crumpling to the ground, Finau stood up and appeared to pop the mangled joint back into place before walking (limping) off the green. The exact nature of his injury, however, was unclear in the immediate aftermath of the fall. Initial media reports described the injury as a dislocated ankle, but retired orthopedic surgeon and PGA tour golfer Bill Mallon disagreed. “There’s no way he dislocated his ankle. Relocating an ankle [after a dislocation] is really hard,” Mallon told Sports Illustrated for its April 9 issue. “It takes huge amounts of strength and lots of anesthesia. It was just a sprain.”
Initial X-rays confirmed Mallon’s suspicions, as they ruled out fractures and/or any significant structural damage to the affected joint. There also was far less pain and swelling for so gruesome an injury—a testament, perhaps, to Finau’s fine athleticism, as well as his overnight regimen of ice packs and anti-inflammatory drugs.
Finau’s relatives, howbeit, would argue it was the Mormon prayer service (for healing) and “spirit of the grounds” that did the trick.
Regardless of the vehicle, Finau sustained only soft tissue damage and some torn ligaments. Masters doctors cleared him to play, fitting the 6-foot 4-inch linksman with a cold/compression treatment device called Game Ready and a compression-only product (Normatec) to further reduce the swelling. Before teeing off, the doctors heavily taped his ankle for stability.
Finau then worked with his coach, Boyd Summerhays, to develop a makeshift swing that would enable him to hit off his back foot. One of the longest hitters on the PGA Tour, Finau gradually returned to his normal swing as the Masters progressed (and his ankle improved); he finished tied for 10th place, posting a final-round 66 that included six consecutive birdies.
Though it nearly sidelined his Masters debut, Finau’s injury—ironically—is likely largely responsible for his impressive performance. He told The New York Times the impairment masked some game pressure by forcing him to think about his ankle. “I had other things on my mind,” he said.
Indeed he did, but Finau’s father Kelepi believes other forces were at play, too.
“This is a holy place in the game of golf...so we did a lot of praying,” he said. “You have to have faith and feel the spirit of the grounds. They let my kid play...”
Maybe so, but biological science had a say in the decision as well. Torn ligaments and soft tissue damage are relatively routine (and fairly common) sports injuries, requiring neither surgical intervention nor extended bouts of rehabilitation. Most slight traumas of this nature need little more than rest, elevation, and repeated ice applications to heal completely. Mother Nature does all the heavy lifting.
While ankle sprains are often considered “minor” injuries, they can have major consequences. Studies have shown that minor sprains can increase the risk of re-injury and eventually lead to joint damage, chronic instability, and ACL (anterior cruciate ligament) troubles. A 2013 National Athletic Trainers’ Association position statement on ankle sprains in athletes warns that “individuals who sustain ankle sprains demonstrate high recurrence rates, prolonged symptoms, diminished quality of life, reduced physical activity levels across the lifespan, a propensity to develop chronic ankle instability, and an increased risk for ankle osteoarthritis.”
Arthrex Inc. is helping athletes (and ordinary folk) avoid such bleak futures with a device designed to improve ankle support. Similar in function to a seatbelt, the InternalBrace is used to repair lateral or medial ankle instability and prevent the possibility of a repeat injury. The product, according to the Naples, Fla.-based firm, augments Broström reconstruction and allows ligaments to heal in a more physiologic manner, at appropriate tension. Early mobilization with the InternalBrace also supports collagen growth and health without also harboring the conditions for future instability.
The InternalBrace consists of two anchors and fiber tape. The anchors are inserted into the talus and fibula—the two bones separated by a ligament that prevents ankles from rolling or becoming sprained. The fiber tape is attached to one anchor and then crossed over the ligament and affixed to the other anchor. The tape sits on top of the ligament to provide a protective coating.
Arthrex’s brace is still a relatively new product, but it already is immensely popular with athletes. Company executives estimate about 22,000 patients annually use the “internal seatbelt” to reinforce their wobbly ankles. “A lot of professional athletes are getting this,” an Arthrex senior manager boasted last spring.
A lot more ordinary Joes are likely to get the brace, too, as they seek relief from the miseries of diabetes, obesity, arthritis, and biological aging in the coming decades. Such afflictions, along with a better awareness of small joint implant technology and its benefits, are expected to drive growth in the worldwide extremities market over the next half-dozen years. London-based Credence Research Inc. predicts the global extremities reconstructive devices market to swell 6.1 percent annually to reach $16.66 billion by 2025, with upper extremity products generating the lion’s share of profits.
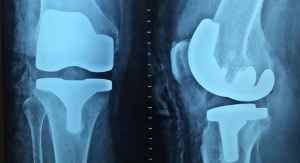
Shoulder and ankle devices are expected to lead the surge as technological advancements like reverse and stemless shoulder implants, natural biomaterials, and bone-prosthetic bonding techniques become more widely accepted in the forecast period. Shoulders are the most frequently replaced extremity—53,000 Americans have shoulder replacement surgery every year, compared with 900,000 who undergo hip or knee replacement surgery annually, according to the Agency for Healthcare Research and Quality, part of the U.S. Department of Health and Human Services.
Commensurate with the market shift toward minimally invasive surgery, stemless arthroplasty is one of the fastest-growing segments of the worldwide shoulder replacement sector. An iData Research study predicts the stemless technique will surpass conventional (stemmed) procedures by 2025 due to its host of clinical benefits and proven comparability with their stemmed counterparts. Overall clinical evidence supports the use of bone-sparing implants, with the most recent study data showing comparability between the two surgical approaches over a nine-year period.
A less invasive arthroplasty option, stemless implants are ideal for patients with post-traumatic shoulder osteoarthritis or extra-articular proximal humerous deformities, conditions that can complicate or preclude the placement of a stemmed prosthesis. The technique is gaining greater assent among surgeons and patients for its various advantages—mainly bone preservation, lower infection and fracture risk, anatomical accuracy, less blood loss, and shorter procedure times.
“We continue to see increasing adoption of stemless shoulder implants,” Steve Vankoski, senior director of Global Marketing, Extremities, for Zimmer Biomet Holdings Inc., told Orthopedic Design & Technology. “The stemless shoulder market is comparatively mature in Europe and Canada, with a 25 percent market penetration for primary shoulder arthroplasty. Within the next few years, we expect the U.S. to approach a similar level of penetration.”
That milestone may be well within reach, but it likely is more than just a “few years” away. Despite its spate of advantages and growing popularity, stemless shoulder implants are a rare commodity in the United States. Only a handful of companies offer the solution, and most of the implants have only recently received regulatory approval.
The newest market entrants include Zimmer Biomet’s Sidus Stem-Free Shoulder System, Exactech’s Equinoxe Stemless Shoulder, and Arthrosurface Inc.’s OVOMotion Shoulder Arthroplasty System. Arthrosurface is somewhat of a veteran in the American market, having introduced the OVO Primary Stemless Shoulder (with a polyethylene inlay glenoid) in 2009; Wright Medical Group N.V. followed suit six years later with Simpliciti, a stemless humeral implant featuring a three-fin design with a collar and female Morse taper that allows for full glenoid access. Simpliciti spent its formative years in Europe (first clinical use in France in 2010) before crossing the pond in 2015.
Zimmer’s dabbles in stemless shoulder technology pre-date Wright Medical’s by more than a decade. The Warsaw, Ind.-based company developed the T.E.S.S. stemless system in 2004 and its next-generation redesign, the Comprehensive Nano, an implant offered in anatomic and reverse configurations. The Sidus system—cleared by the U.S. Food and Drug Administration (FDA) in January—is intended to anatomically restore patient anatomy while preserving bone stock and improving post-operative outcomes.
The same objectives can be found in Exactech’s Equinoxe model, a system featuring a single tray of instruments; a 3D porous structure with optimized pore size, count, and porosity; and a bone cage for both biological fixation and bone growth. Company executives consider the implant’s 3D porous material and bone cage technology to be market differentiators, but industry analysts contend the product’s regulatory pathway is actually more groundbreaking.
The Equinoxe clearance (in March) is particularly significant, analysts say, because it was conducted without an Investigational Device Exemption (IDE), an FDA approval process that allows a medical device to be used in a clinical study so safety and effectiveness data can be collected. Both Sidus and Simpliciti—the Equinoxe’s direct competition—received FDA clearance with IDEs.
Clearing the Equinoxe stemless system without an IDE could bode well for other shoulder arthroplasty manufacturers, allowing them to enter the market significantly faster than anticipated (in about one year, following Exactech’s timeline), Wells Fargo Securities analyst Larry Biegelsen noted.
The IDE-less regulatory route certainly could help facilitate Catalyst OrthoScience Inc.’s commercialization efforts. The Naples, Fla.-based company was granted four U.S. patents in January for technologies incorporated into the CSR Total Shoulder System, a new, canal-sparing anatomic arthroplasty solution that features a smaller, more anatomically-shaped humeral implant and a less invasive approach than traditional surgery. The patents expand the company’s intellectual property (IP) portfolio of proprietary glenoid and humeral implant designs and instruments.
Arthrosurface skipped the IDE process in its 510(k) application for the OVOMotion, a system blessed by the FDA in April to treat damage from arthritis, trauma, or Avascular Necrosis (bone tissue death). The company considers its stemless implant to be a “truly revolutionary” product for its anatomic restoration abilities and aspherical head design that better replicates patient anatomy and improves joint access for easier glenoid preparation.
The OVOMotion is designed to be part of a three-element moduar stemless platform that will ultimately allow surgeons to intra-operatively choose between stemless, traditional, and reverse options.
The latter alternative, traditionally used to treat older patients with rotator cuff arthropathy, is gaining prevalance amid a wider spectrum of indications. Pathologies now treated include acute proximal humerus fractures, rotator cuff tears without arthropathy, osteoarthritis, chronic dislocation, failed anatomic total shoulder replacement, tumor resection, and rheumatoid arthritis in older and younger patients.
Business intelligence firm BioMedGPS LLC projects that reverse shoulder arthroplasty will comprise more than 40 percent of all U.S. shoulder replacement procedures by 2021. Research shows the reverse shoulder sector controlled over 50 percent of total U.S. shoulder market revenues in 2015; going forward, the sector is expected to grow 10 percent annually through 2020.
“Another noteworthy trend this year is the increasing use of augmented glenoid solutions for reverse shoulder arthroplasty,” Vankoski said. “A primary benefit of augmented versus non-augmented baseplates is that they are intended to better match a patient’s deformity. Rather than having to remove more of the patient’s bone to accommodate for the baseplate to fit, augmented baseplates more closely match the patient’s deformity to yield the potential for greater bone preservation. Additionally, these solutions are being introduced to the global market nearly simultaneously, leading to adoption and market presence in Europe, Canada, and the U.S.”
Concurrently indeed: The market debut of Zimmer Biomet’s latest augmented baseplate solution coincided with the March 510(k) clearance of Arthrex’s Univers Revers Modular Glenoid System, a porous-coated implant featuring an anatomically-shaped baseplate available in three sizes. The product is designed to fix badly damaged shoulder joints.
Synchronous with Arthrex’s low-key (non-publicized) FDA nod, Zimmer Biomet showcased its new FDA-cleared augmented baseplate solution and heralded the product’s first use at the renowned Mayo Clinic in Rochester, Minn. The company’s Comprehensive Augmented Baseplate features a simplified circular design at three augment heights (3 mm, 5 mm, 7 mm buildup) for improved accommodation of various bone anatomies. The circular structure also allows for bone ingrowth into the baseplate rim for optimal fixation. Moreover, the baseplate leverages Mayo Clinic’s patented methodolody for providing relevant component sizing options and preserving bone stock.
Ankle Market on Solid Footing
Nearly two months have passed since Tony Finau’s unusual (albeit embarrassing) golfing injury, and the Utah native is well on the road to a full recovery. He might be wise, though, to refrain from running backwards for the foreseeable future, as studies show he’s at high risk of re-injuring his ankle. Finau also should take a cue from his own (medical) history: He’s hurt his right ankle five times, according to published reports.
Injuries (sprains) like those suffered by Finau and approximately 2 million other Americans each year are turning the foot and ankle sector into a formidable catalyst for growth within the worldwide extremities market. Silicon Valley-based Grand View Research Inc. predicts the global foot and ankle segment to grow 10.1 percent annually over the next seven years to reach $3.4 billion by 2025; the increase is slated to be driven by mounting incidents of ankle sprains, hammertoe, bunions, osteoporosis, rheumatoid arthritis, and fractures.
“The foot and ankle and wrist/hand segments of the orthopedic market are among the fastest-growing segments in the U.S.,” noted Emad Abdelnaby, senior director of U.S. marketing for Trauma & CMF at DePuy Synthes Inc. “According to the U.S. Census Bureau, in 2050 the population aged 65 and over is projected to be 83.7 million, almost double its estimated population of 43.1 million in 2012. This population may be more susceptible to slips and falls, particularly as they remain more active throughout their lifetime. As that population grows, so does the number of physicians performing orthopedic injury procedures. To that end, it is critical for DePuy Synthes to continue to find different ways to support these healthcare professionals so they can best care for their patients...”
DePuy’s latest means of support include the MAXFRAME Multi-Axial Correction System and Continuous Compression Implants. Launched last fall, the MAXFRAME system is an external circular fixation device designed to gradually correct bone or soft tissue deformities in the arm, leg, foot, or ankle. The product’s 3D planning software crafts accurate deformity treatments through advanced algorithms (eliminating the need for manual measurements) and requires fewer inputs than competitive systems. DePuy claims the MAXFRAME software can potentially reduce the number of patient X-rays required, thus mitigating procedural complexity, radiation exposure for both patient and surgeon, and overall costs, while minimizing treatment time.
Patients undergoing treatment with the MAXFRAME System adjust the struts on the device themselves in accordance with their customized treatment plan. The system’s ASSURE-STRUT technology makes adjustments easy by sounding an audible “click” as the strut correctly locks into place.
DePuy’s Continuous Compression Implants were released in December 2017 in Europe, and the Middle East and Africa. The implants are made with nitinol (a “shape memory” metal) to continuously maintain bone compression during recovery from foot and ankle surgery. DePuy contends the compression can help reduce the risk of complications and enable patients to more quickly resume normal activities.
The Continuous Compression implants are pre-sterilized and pre-loaded onto an insertion tool for improved operating room efficiency; there is no need to heat or freeze the implants prior to use.
* * * *
Buoyed by rising numbers of joint disorders and the planet’s aging populace, the worldwide extremities market is becoming quite crowded with players seeking to capitalize on its high-growth opportunities, particularly those in the burgeoning shoulder and ankle sectors. In the past, large OEMs tended to ignore this market, opting instead to concentrate on the larger (and more profitable) hip and knee segments. In recent years, however, these companies have seen the error of their ways and added extremities technologies to their portfolios through either in-house product development or acquisition.
“The trend that is most prevalent to our company is the increasing competition in the marketplace,” said Jason Pringle, market coordinator for BioPro Inc., a Port Huron, Mich.-based designer and manufacturer of joint replacement implants and surgical devices. “When BioPro first started focusing on the extremities market in the early 2000s, there were fewer large orthopedic players focused on extremities. Now with the acquisitions of startup extremity device companies, the large orthopedic companies have really started to focus on the market and expand their portfolios.”
And they haven’t looked back since.
It certainly would explain a lot of things: It would, for example, lend credence to the gold dust-spouting natural spring purportedly nestled between the 13th and 14th fairways, and the exceptionally low concessions prices ($1.50/pimento cheese sandwich, $1/chips, $4/beer). It also would legitimize a handful of hole names—specifically, Nos. 11, 12, 13 (Amen Corner), No. 10 (Camillia, a Latin moniker meaning “helper to the priest”), and No. 17 (Nandina, a plant more commonly known as “heavenly” or “sacred bamboo”).
“If there’s a golf course in heaven,” three-time Masters Tournament winner Gary Player once told Reuters, “I hope it’s like Augusta National.”
Tony Finau might say the two are one in the same. During the Masters’ 2018 Par-3 contest in early April, the 28-year-old golfer injured his left ankle celebrating a hole-in-one but rebounded the next day to shoot a four-under-par 68 in the championship game’s opening round. He briefly led the tournament before slipping behind three-time major winner Jordan Spieth to tie for second place with Matthew Kuchar.
“It’s nothing short of a miracle if you ask me,” Finau said of his first-day Masters performance.
Miraculous so it seemed: After rolling his ankle and crumpling to the ground, Finau stood up and appeared to pop the mangled joint back into place before walking (limping) off the green. The exact nature of his injury, however, was unclear in the immediate aftermath of the fall. Initial media reports described the injury as a dislocated ankle, but retired orthopedic surgeon and PGA tour golfer Bill Mallon disagreed. “There’s no way he dislocated his ankle. Relocating an ankle [after a dislocation] is really hard,” Mallon told Sports Illustrated for its April 9 issue. “It takes huge amounts of strength and lots of anesthesia. It was just a sprain.”
Initial X-rays confirmed Mallon’s suspicions, as they ruled out fractures and/or any significant structural damage to the affected joint. There also was far less pain and swelling for so gruesome an injury—a testament, perhaps, to Finau’s fine athleticism, as well as his overnight regimen of ice packs and anti-inflammatory drugs.
Finau’s relatives, howbeit, would argue it was the Mormon prayer service (for healing) and “spirit of the grounds” that did the trick.
Regardless of the vehicle, Finau sustained only soft tissue damage and some torn ligaments. Masters doctors cleared him to play, fitting the 6-foot 4-inch linksman with a cold/compression treatment device called Game Ready and a compression-only product (Normatec) to further reduce the swelling. Before teeing off, the doctors heavily taped his ankle for stability.
Finau then worked with his coach, Boyd Summerhays, to develop a makeshift swing that would enable him to hit off his back foot. One of the longest hitters on the PGA Tour, Finau gradually returned to his normal swing as the Masters progressed (and his ankle improved); he finished tied for 10th place, posting a final-round 66 that included six consecutive birdies.
Though it nearly sidelined his Masters debut, Finau’s injury—ironically—is likely largely responsible for his impressive performance. He told The New York Times the impairment masked some game pressure by forcing him to think about his ankle. “I had other things on my mind,” he said.
Indeed he did, but Finau’s father Kelepi believes other forces were at play, too.
“This is a holy place in the game of golf...so we did a lot of praying,” he said. “You have to have faith and feel the spirit of the grounds. They let my kid play...”
Maybe so, but biological science had a say in the decision as well. Torn ligaments and soft tissue damage are relatively routine (and fairly common) sports injuries, requiring neither surgical intervention nor extended bouts of rehabilitation. Most slight traumas of this nature need little more than rest, elevation, and repeated ice applications to heal completely. Mother Nature does all the heavy lifting.
While ankle sprains are often considered “minor” injuries, they can have major consequences. Studies have shown that minor sprains can increase the risk of re-injury and eventually lead to joint damage, chronic instability, and ACL (anterior cruciate ligament) troubles. A 2013 National Athletic Trainers’ Association position statement on ankle sprains in athletes warns that “individuals who sustain ankle sprains demonstrate high recurrence rates, prolonged symptoms, diminished quality of life, reduced physical activity levels across the lifespan, a propensity to develop chronic ankle instability, and an increased risk for ankle osteoarthritis.”
Arthrex Inc. is helping athletes (and ordinary folk) avoid such bleak futures with a device designed to improve ankle support. Similar in function to a seatbelt, the InternalBrace is used to repair lateral or medial ankle instability and prevent the possibility of a repeat injury. The product, according to the Naples, Fla.-based firm, augments Broström reconstruction and allows ligaments to heal in a more physiologic manner, at appropriate tension. Early mobilization with the InternalBrace also supports collagen growth and health without also harboring the conditions for future instability.
The InternalBrace consists of two anchors and fiber tape. The anchors are inserted into the talus and fibula—the two bones separated by a ligament that prevents ankles from rolling or becoming sprained. The fiber tape is attached to one anchor and then crossed over the ligament and affixed to the other anchor. The tape sits on top of the ligament to provide a protective coating.
Arthrex’s brace is still a relatively new product, but it already is immensely popular with athletes. Company executives estimate about 22,000 patients annually use the “internal seatbelt” to reinforce their wobbly ankles. “A lot of professional athletes are getting this,” an Arthrex senior manager boasted last spring.
A lot more ordinary Joes are likely to get the brace, too, as they seek relief from the miseries of diabetes, obesity, arthritis, and biological aging in the coming decades. Such afflictions, along with a better awareness of small joint implant technology and its benefits, are expected to drive growth in the worldwide extremities market over the next half-dozen years. London-based Credence Research Inc. predicts the global extremities reconstructive devices market to swell 6.1 percent annually to reach $16.66 billion by 2025, with upper extremity products generating the lion’s share of profits.
Shoulder and ankle devices are expected to lead the surge as technological advancements like reverse and stemless shoulder implants, natural biomaterials, and bone-prosthetic bonding techniques become more widely accepted in the forecast period. Shoulders are the most frequently replaced extremity—53,000 Americans have shoulder replacement surgery every year, compared with 900,000 who undergo hip or knee replacement surgery annually, according to the Agency for Healthcare Research and Quality, part of the U.S. Department of Health and Human Services.
Commensurate with the market shift toward minimally invasive surgery, stemless arthroplasty is one of the fastest-growing segments of the worldwide shoulder replacement sector. An iData Research study predicts the stemless technique will surpass conventional (stemmed) procedures by 2025 due to its host of clinical benefits and proven comparability with their stemmed counterparts. Overall clinical evidence supports the use of bone-sparing implants, with the most recent study data showing comparability between the two surgical approaches over a nine-year period.
A less invasive arthroplasty option, stemless implants are ideal for patients with post-traumatic shoulder osteoarthritis or extra-articular proximal humerous deformities, conditions that can complicate or preclude the placement of a stemmed prosthesis. The technique is gaining greater assent among surgeons and patients for its various advantages—mainly bone preservation, lower infection and fracture risk, anatomical accuracy, less blood loss, and shorter procedure times.
“We continue to see increasing adoption of stemless shoulder implants,” Steve Vankoski, senior director of Global Marketing, Extremities, for Zimmer Biomet Holdings Inc., told Orthopedic Design & Technology. “The stemless shoulder market is comparatively mature in Europe and Canada, with a 25 percent market penetration for primary shoulder arthroplasty. Within the next few years, we expect the U.S. to approach a similar level of penetration.”
That milestone may be well within reach, but it likely is more than just a “few years” away. Despite its spate of advantages and growing popularity, stemless shoulder implants are a rare commodity in the United States. Only a handful of companies offer the solution, and most of the implants have only recently received regulatory approval.
The newest market entrants include Zimmer Biomet’s Sidus Stem-Free Shoulder System, Exactech’s Equinoxe Stemless Shoulder, and Arthrosurface Inc.’s OVOMotion Shoulder Arthroplasty System. Arthrosurface is somewhat of a veteran in the American market, having introduced the OVO Primary Stemless Shoulder (with a polyethylene inlay glenoid) in 2009; Wright Medical Group N.V. followed suit six years later with Simpliciti, a stemless humeral implant featuring a three-fin design with a collar and female Morse taper that allows for full glenoid access. Simpliciti spent its formative years in Europe (first clinical use in France in 2010) before crossing the pond in 2015.
Zimmer’s dabbles in stemless shoulder technology pre-date Wright Medical’s by more than a decade. The Warsaw, Ind.-based company developed the T.E.S.S. stemless system in 2004 and its next-generation redesign, the Comprehensive Nano, an implant offered in anatomic and reverse configurations. The Sidus system—cleared by the U.S. Food and Drug Administration (FDA) in January—is intended to anatomically restore patient anatomy while preserving bone stock and improving post-operative outcomes.
The same objectives can be found in Exactech’s Equinoxe model, a system featuring a single tray of instruments; a 3D porous structure with optimized pore size, count, and porosity; and a bone cage for both biological fixation and bone growth. Company executives consider the implant’s 3D porous material and bone cage technology to be market differentiators, but industry analysts contend the product’s regulatory pathway is actually more groundbreaking.
The Equinoxe clearance (in March) is particularly significant, analysts say, because it was conducted without an Investigational Device Exemption (IDE), an FDA approval process that allows a medical device to be used in a clinical study so safety and effectiveness data can be collected. Both Sidus and Simpliciti—the Equinoxe’s direct competition—received FDA clearance with IDEs.
Clearing the Equinoxe stemless system without an IDE could bode well for other shoulder arthroplasty manufacturers, allowing them to enter the market significantly faster than anticipated (in about one year, following Exactech’s timeline), Wells Fargo Securities analyst Larry Biegelsen noted.
The IDE-less regulatory route certainly could help facilitate Catalyst OrthoScience Inc.’s commercialization efforts. The Naples, Fla.-based company was granted four U.S. patents in January for technologies incorporated into the CSR Total Shoulder System, a new, canal-sparing anatomic arthroplasty solution that features a smaller, more anatomically-shaped humeral implant and a less invasive approach than traditional surgery. The patents expand the company’s intellectual property (IP) portfolio of proprietary glenoid and humeral implant designs and instruments.
Arthrosurface skipped the IDE process in its 510(k) application for the OVOMotion, a system blessed by the FDA in April to treat damage from arthritis, trauma, or Avascular Necrosis (bone tissue death). The company considers its stemless implant to be a “truly revolutionary” product for its anatomic restoration abilities and aspherical head design that better replicates patient anatomy and improves joint access for easier glenoid preparation.
The OVOMotion is designed to be part of a three-element moduar stemless platform that will ultimately allow surgeons to intra-operatively choose between stemless, traditional, and reverse options.
The latter alternative, traditionally used to treat older patients with rotator cuff arthropathy, is gaining prevalance amid a wider spectrum of indications. Pathologies now treated include acute proximal humerus fractures, rotator cuff tears without arthropathy, osteoarthritis, chronic dislocation, failed anatomic total shoulder replacement, tumor resection, and rheumatoid arthritis in older and younger patients.
Business intelligence firm BioMedGPS LLC projects that reverse shoulder arthroplasty will comprise more than 40 percent of all U.S. shoulder replacement procedures by 2021. Research shows the reverse shoulder sector controlled over 50 percent of total U.S. shoulder market revenues in 2015; going forward, the sector is expected to grow 10 percent annually through 2020.
“Another noteworthy trend this year is the increasing use of augmented glenoid solutions for reverse shoulder arthroplasty,” Vankoski said. “A primary benefit of augmented versus non-augmented baseplates is that they are intended to better match a patient’s deformity. Rather than having to remove more of the patient’s bone to accommodate for the baseplate to fit, augmented baseplates more closely match the patient’s deformity to yield the potential for greater bone preservation. Additionally, these solutions are being introduced to the global market nearly simultaneously, leading to adoption and market presence in Europe, Canada, and the U.S.”
Concurrently indeed: The market debut of Zimmer Biomet’s latest augmented baseplate solution coincided with the March 510(k) clearance of Arthrex’s Univers Revers Modular Glenoid System, a porous-coated implant featuring an anatomically-shaped baseplate available in three sizes. The product is designed to fix badly damaged shoulder joints.
Synchronous with Arthrex’s low-key (non-publicized) FDA nod, Zimmer Biomet showcased its new FDA-cleared augmented baseplate solution and heralded the product’s first use at the renowned Mayo Clinic in Rochester, Minn. The company’s Comprehensive Augmented Baseplate features a simplified circular design at three augment heights (3 mm, 5 mm, 7 mm buildup) for improved accommodation of various bone anatomies. The circular structure also allows for bone ingrowth into the baseplate rim for optimal fixation. Moreover, the baseplate leverages Mayo Clinic’s patented methodolody for providing relevant component sizing options and preserving bone stock.
Ankle Market on Solid Footing
Nearly two months have passed since Tony Finau’s unusual (albeit embarrassing) golfing injury, and the Utah native is well on the road to a full recovery. He might be wise, though, to refrain from running backwards for the foreseeable future, as studies show he’s at high risk of re-injuring his ankle. Finau also should take a cue from his own (medical) history: He’s hurt his right ankle five times, according to published reports.
Injuries (sprains) like those suffered by Finau and approximately 2 million other Americans each year are turning the foot and ankle sector into a formidable catalyst for growth within the worldwide extremities market. Silicon Valley-based Grand View Research Inc. predicts the global foot and ankle segment to grow 10.1 percent annually over the next seven years to reach $3.4 billion by 2025; the increase is slated to be driven by mounting incidents of ankle sprains, hammertoe, bunions, osteoporosis, rheumatoid arthritis, and fractures.
“The foot and ankle and wrist/hand segments of the orthopedic market are among the fastest-growing segments in the U.S.,” noted Emad Abdelnaby, senior director of U.S. marketing for Trauma & CMF at DePuy Synthes Inc. “According to the U.S. Census Bureau, in 2050 the population aged 65 and over is projected to be 83.7 million, almost double its estimated population of 43.1 million in 2012. This population may be more susceptible to slips and falls, particularly as they remain more active throughout their lifetime. As that population grows, so does the number of physicians performing orthopedic injury procedures. To that end, it is critical for DePuy Synthes to continue to find different ways to support these healthcare professionals so they can best care for their patients...”
DePuy’s latest means of support include the MAXFRAME Multi-Axial Correction System and Continuous Compression Implants. Launched last fall, the MAXFRAME system is an external circular fixation device designed to gradually correct bone or soft tissue deformities in the arm, leg, foot, or ankle. The product’s 3D planning software crafts accurate deformity treatments through advanced algorithms (eliminating the need for manual measurements) and requires fewer inputs than competitive systems. DePuy claims the MAXFRAME software can potentially reduce the number of patient X-rays required, thus mitigating procedural complexity, radiation exposure for both patient and surgeon, and overall costs, while minimizing treatment time.
Patients undergoing treatment with the MAXFRAME System adjust the struts on the device themselves in accordance with their customized treatment plan. The system’s ASSURE-STRUT technology makes adjustments easy by sounding an audible “click” as the strut correctly locks into place.
DePuy’s Continuous Compression Implants were released in December 2017 in Europe, and the Middle East and Africa. The implants are made with nitinol (a “shape memory” metal) to continuously maintain bone compression during recovery from foot and ankle surgery. DePuy contends the compression can help reduce the risk of complications and enable patients to more quickly resume normal activities.
The Continuous Compression implants are pre-sterilized and pre-loaded onto an insertion tool for improved operating room efficiency; there is no need to heat or freeze the implants prior to use.
* * * *
Buoyed by rising numbers of joint disorders and the planet’s aging populace, the worldwide extremities market is becoming quite crowded with players seeking to capitalize on its high-growth opportunities, particularly those in the burgeoning shoulder and ankle sectors. In the past, large OEMs tended to ignore this market, opting instead to concentrate on the larger (and more profitable) hip and knee segments. In recent years, however, these companies have seen the error of their ways and added extremities technologies to their portfolios through either in-house product development or acquisition.
“The trend that is most prevalent to our company is the increasing competition in the marketplace,” said Jason Pringle, market coordinator for BioPro Inc., a Port Huron, Mich.-based designer and manufacturer of joint replacement implants and surgical devices. “When BioPro first started focusing on the extremities market in the early 2000s, there were fewer large orthopedic players focused on extremities. Now with the acquisitions of startup extremity device companies, the large orthopedic companies have really started to focus on the market and expand their portfolios.”
And they haven’t looked back since.