08.14.19
AT A GLANCE
Rank: #6 (Last year: #7)
$1.10 Billion
Prior Fiscal: $1.03 Billion
Percentage Change: +7.3%
No. of Employees: 2,600
Global Headquarters: San Diego, Calif.
KEY EXECUTIVES:
Chris Barry, CEO
Rajesh Asarpota, Exec. VP and CFO
Matt Link, President
Paul McClintock, President, U.S. Commercial
Nathaniel Sisitsky, Esq., Sr. VP, General Counsel and Corporate Secretary
Lucas Vitale, Chief Human Resources Officer
The SpineTRACK Registry, a NuVasive-sponsored prospective, multicenter, observational data collection quality improvement tool, was established in 2011. SpineTRACK lets surgeons use real-time reports to make better treatment decisions, and also allows patients to track progress to better understand treatment benefits. Hospitals can also use SpineTRACK to evaluate outcome variations to provide the highest value of care.
Last February, the Centers for Medicare and Medicaid Service (CMS) designated SpineTRACK as a Qualified Clinical Data Registry (QCDR) for the 2018 Merit-based Incentive Payment System reporting year. That meant the platform could be used to earn Medicare payment incentives.
“The QCDR designation helps our customers meet quality reporting criteria to deliver optimal results and earn incentives that can grow their practices,” NuVasive then-executive vice president of strategy, technology, and corporate development Matt Link told the press. “This is truly a value-add that aligns with our focus on systems-based spine solutions.”
Three months later, helped along by the significant adoption increase from QCDR designation, SpineTRACK enrolled its 10,000th patient. “SpineTRACK is foundational in our efforts to enhance the patient experience throughout the continuum of care with advanced data analytics,” Link said. “These elements will help fuel the future of Surgical Intelligence and continue to help revolutionize spine care.”
Half a year later, the company promoted Link to president. J. Christopher Barry was also welcomed as NuVasive’s new CEO then, replacing Gregory Lucier. He had previously served as senior vice president and president of Medtronic’s Surgical Innovations business, directing over 14,000 employees, diversifying the business, and overseeing Medtronic’s surgical robotics development initiative. Before that, he held commercial and leadership roles at Covidien.
“Chris has successfully led teams globally, managed complex R&D programs, driven commercial initiatives, and executed strategic acquisitions. He has built a reputation for driving employee engagement and operational excellence,” Lucier told the press at the time of Barry’s appointment.
“The combination of Chris and Matt at the helm of NuVasive creates the most dynamic leadership team in the industry,” he added.
Barry took the reins of a company that has continued to grow due to the strong demand for minimally invasive spinal procedures. NuVasive’s flagship eXtreme Lateral Interbody Fusion (XLIF) procedure, in which surgeons access the spine for fusion from the side of the patient’s body rather than the front or back, is in high demand due to decreased trauma and blood loss, as well as faster recovery times compared to open surgery. Last year’s $1.1 billion revenue continued the firm’s upward trajectory, vaulting 7.3 percent ($75 million) over the prior year.
The spinal hardware franchise, which consists of implants and fixation products, grew 7 percent ($51.1 million) last year to reach $788.6 million. Spinal hardware product volume increased the segment’s revenue by 9 percent, but unfavorable pricing impacts offset the gain by 2 percent. Foreign currency impact had no material impact on spinal hardware sales.
ANALYST INSIGHTS: NuVasive continues to be a thought leader, driving innovation in a spine segment doing its best to stagnate. Comprehensive, elegant solutions and strong leadership will continue to drive revenue and profits.
Last January, NuVasive launched the COALESCE thoracolumbar interbody fusion device (TLIF) and was granted an FDA label expansion for its COHERE cervical interbody fusion device.
COALESCE and COHERE both provide a 3D porous polyetheretherketone (PEEK) architecture to promote bone ingrowth. The launch made MR-compatible COALESCE available for both TLIF and PLIF procedures. COHERE’s label expansion allows its use at multiple cervical spine levels (C2-S1) with both autograft and allograft. COHERE was also given a new ICD code from CMS at that time.
The firm introduced its Maximum Access Surgery (MAS) anterior lumbar interbody fusion (ALIF) retractor platform last April. The retractor system helps retain traditional ALIF characteristics while allowing surgeons either a lateral or supine approach to spine surgery—accessing L5/S1 laterally has historically had shortcomings because existing systems weren’t designed for those approaches. The Lateral ALIF Access System enables single-position surgery for multilevel cases during the company’s proprietary XLIF procedure.
MAGEC X for early-onset scoliosis treatment was launched in the U.S. last July. MAGEC X features updates for all rod diameters in the MAGEC portfolio to deliver 68 percent performance increase over the previous generation (according to NuVasive). Improvements were made to the internal rod mechanism as well, including a reinforced locking pin and robust actuator seal to further contain and reduce the release of titanium wear debris. An anti-jam feature simplifies intraoperative and post-op rod lengthening, and laser-marked sagittal bending lines enhance workflow efficiency and ease of use. MAGEC X was launched in the U.K. two months later.
The Reline MAS Midline (RMM) low-profile modular implants for posterior fixation were also released last July. The RMM implants and instrumentation address alignment and aim to reduce adjacent segment transition syndrome, providing a minimally invasive approach to bilateral decompression under complete visualization, interbody fusion, and stabilization with pedicle screws. The RMM solution supports both cortical and standard trajectory techniques.
The company unveiled the XLIF Lordotic Expandable (XLX) Interbody System at last July’s International Meeting on Advanced Spine Techniques. The XLIF anterior column realignment (ACR) technique, together with XLX ACR, addresses sagittal alignment from the anterior column while reducing blood loss and hospital stays. It’s used to treat symptomatic disc degeneration or degenerative spondylolisthesis at one or two adjacent levels—including thoracic disc herniation—from L1 to S1. It can be used as an adjunct to fusion in patients with multilevel degenerative scoliosis as well.
The TLX 20 degree expandable spinal interbody implant was introduced last September. It touts a first-of-its-kind oblique profile for transforaminal lumbar interbody fusion (TLIF) procedures. The TLX 20 expandable implant’s integrated auto-lock feature allows customizable expansion for up to 20 degrees of oblique lordosis and tailoring implant expansion based on clinical need. The system also includes a single, low-profile instrument to position, expand, and post-pack the implant with bone graft, and distal tip tapering to aid insertion in a collapsed disc space.
Last November saw the U.S. launch of Brigade Lateral, at the time the industry’s first interbody system optimized for ALIF spine surgery. Brigade Lateral features improved instrumentation, a new insertion method, and an expanded lordotic implant offering for NuVasive’s proprietary Lateral ALIF procedure, which enables access to L5-S1 from a lateral position. A threaded in-line inserter angled off the midline and positioned on the implant’s lateral edge also allows better L5-S1 access.
That same month the COHERE porous PEEK implant won FDA clearance for the firm’s proprietary XLIF surgical spine procedure. The porous structure supports bone ongrowth and ingrowth, and PEEK’s radiolucent properties increase clarity in post-op X-rays and imaging. The nod allows surgeons to use the implant with autograft and/or allograft in skeletally mature patients for thoracolumbar pathologies including degenerative disc disease, degenerative spondylolisthesis, and degenerative scoliosis.
The company obtained FDA clearance for expanded use of the Monolith Corpectomy System, a modular PEEK interbody implant for cervical corpectomy procedures, last December. The expansion includes procedures to treat C3-C7 vertebral bodies damaged by fracture, tumors, or osteomyelitis, or to support reconstruction after corpectomy is performed to achieve spinal cord and neural tissue decompression in cervical degenerative disorders.
The surgical support business, which is comprised of intraoperative monitoring services, disposables, and biologics, increased sales $23.9 million—8 percent—over the previous year. Most of the growth (7 percent) came as a result of the company’s purchase of intraoperative neurophysiological monitoring (IONM) services firm SafePassage in early 2018. SafePassage provided IONM and EEG services to surgeons and healthcare facilities across the eastern U.S., and NuVasive expects to support over 100,000 IONM cases annually in the U.S. thanks to the purchase.
The AttraX Scaffold biologic, an absorbent ceramic-collagen bone graft with an optimized surface, launched last June. AttraX’s surface touts carefully defined and tightly controlled features at the submicron scale, which drives differentiation of mesenchymal stem cells into bone-forming osteoblasts without added growth factors. It’s used in the posterolateral spine to promote fusions, having demonstrated equivalent or better fusion rates than autograft and faster than ceramic grafts, according to the company.
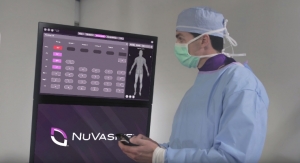
July certainly was a busy month for NuVasive—during its last week the firm gained FDA clearance for its Pulse surgical automation platform, billed as spine’s first integrated surgical automation platform. Pulse combines 2D and 3D navigation and smart imaging, neuromonitoring, surgical planning, radiation reduction, and patient-specific rod bending technologies in one platform.
Imaging and insights can be simultaneously viewed in real-time through Wi-Fi connectivity and independent device access. Multiple high-resolution cameras combined with a low-profile, 360-degree array drives uninterrupted line-of-sight and optimized procedural workflow.
“Currently, a number of hospitals and healthcare systems treat patients undergoing spine surgery through often cost-intensive, intra-operative CT scans with a general navigation system with limited utilization in spine surgery cases. The combination of NuVasive’s Pulse system and the Siemens Healthineers’ next-generation advanced imaging technology provides a compelling offering for hospitals and surgeons who require a scalable, cost-efficient technology that maximizes OR workflow efficiency and significantly improves visualization for spine surgery,” then-CEO Lucier told the press. “We firmly believe this combined offering will enable advanced clinical benefits driving superior patient outcomes, delivered through greatly improved visualization of the anatomy promoting successful anatomical access and spinal implant placement.”
Rank: #6 (Last year: #7)
$1.10 Billion
Prior Fiscal: $1.03 Billion
Percentage Change: +7.3%
No. of Employees: 2,600
Global Headquarters: San Diego, Calif.
KEY EXECUTIVES:
Chris Barry, CEO
Rajesh Asarpota, Exec. VP and CFO
Matt Link, President
Paul McClintock, President, U.S. Commercial
Nathaniel Sisitsky, Esq., Sr. VP, General Counsel and Corporate Secretary
Lucas Vitale, Chief Human Resources Officer
The SpineTRACK Registry, a NuVasive-sponsored prospective, multicenter, observational data collection quality improvement tool, was established in 2011. SpineTRACK lets surgeons use real-time reports to make better treatment decisions, and also allows patients to track progress to better understand treatment benefits. Hospitals can also use SpineTRACK to evaluate outcome variations to provide the highest value of care.
Last February, the Centers for Medicare and Medicaid Service (CMS) designated SpineTRACK as a Qualified Clinical Data Registry (QCDR) for the 2018 Merit-based Incentive Payment System reporting year. That meant the platform could be used to earn Medicare payment incentives.
“The QCDR designation helps our customers meet quality reporting criteria to deliver optimal results and earn incentives that can grow their practices,” NuVasive then-executive vice president of strategy, technology, and corporate development Matt Link told the press. “This is truly a value-add that aligns with our focus on systems-based spine solutions.”
Three months later, helped along by the significant adoption increase from QCDR designation, SpineTRACK enrolled its 10,000th patient. “SpineTRACK is foundational in our efforts to enhance the patient experience throughout the continuum of care with advanced data analytics,” Link said. “These elements will help fuel the future of Surgical Intelligence and continue to help revolutionize spine care.”
Half a year later, the company promoted Link to president. J. Christopher Barry was also welcomed as NuVasive’s new CEO then, replacing Gregory Lucier. He had previously served as senior vice president and president of Medtronic’s Surgical Innovations business, directing over 14,000 employees, diversifying the business, and overseeing Medtronic’s surgical robotics development initiative. Before that, he held commercial and leadership roles at Covidien.
“Chris has successfully led teams globally, managed complex R&D programs, driven commercial initiatives, and executed strategic acquisitions. He has built a reputation for driving employee engagement and operational excellence,” Lucier told the press at the time of Barry’s appointment.
“The combination of Chris and Matt at the helm of NuVasive creates the most dynamic leadership team in the industry,” he added.
Barry took the reins of a company that has continued to grow due to the strong demand for minimally invasive spinal procedures. NuVasive’s flagship eXtreme Lateral Interbody Fusion (XLIF) procedure, in which surgeons access the spine for fusion from the side of the patient’s body rather than the front or back, is in high demand due to decreased trauma and blood loss, as well as faster recovery times compared to open surgery. Last year’s $1.1 billion revenue continued the firm’s upward trajectory, vaulting 7.3 percent ($75 million) over the prior year.
The spinal hardware franchise, which consists of implants and fixation products, grew 7 percent ($51.1 million) last year to reach $788.6 million. Spinal hardware product volume increased the segment’s revenue by 9 percent, but unfavorable pricing impacts offset the gain by 2 percent. Foreign currency impact had no material impact on spinal hardware sales.
ANALYST INSIGHTS: NuVasive continues to be a thought leader, driving innovation in a spine segment doing its best to stagnate. Comprehensive, elegant solutions and strong leadership will continue to drive revenue and profits.
—Patrick West, Partner, Mirus Capital Advisors
Last January, NuVasive launched the COALESCE thoracolumbar interbody fusion device (TLIF) and was granted an FDA label expansion for its COHERE cervical interbody fusion device.
COALESCE and COHERE both provide a 3D porous polyetheretherketone (PEEK) architecture to promote bone ingrowth. The launch made MR-compatible COALESCE available for both TLIF and PLIF procedures. COHERE’s label expansion allows its use at multiple cervical spine levels (C2-S1) with both autograft and allograft. COHERE was also given a new ICD code from CMS at that time.
The firm introduced its Maximum Access Surgery (MAS) anterior lumbar interbody fusion (ALIF) retractor platform last April. The retractor system helps retain traditional ALIF characteristics while allowing surgeons either a lateral or supine approach to spine surgery—accessing L5/S1 laterally has historically had shortcomings because existing systems weren’t designed for those approaches. The Lateral ALIF Access System enables single-position surgery for multilevel cases during the company’s proprietary XLIF procedure.
MAGEC X for early-onset scoliosis treatment was launched in the U.S. last July. MAGEC X features updates for all rod diameters in the MAGEC portfolio to deliver 68 percent performance increase over the previous generation (according to NuVasive). Improvements were made to the internal rod mechanism as well, including a reinforced locking pin and robust actuator seal to further contain and reduce the release of titanium wear debris. An anti-jam feature simplifies intraoperative and post-op rod lengthening, and laser-marked sagittal bending lines enhance workflow efficiency and ease of use. MAGEC X was launched in the U.K. two months later.
The Reline MAS Midline (RMM) low-profile modular implants for posterior fixation were also released last July. The RMM implants and instrumentation address alignment and aim to reduce adjacent segment transition syndrome, providing a minimally invasive approach to bilateral decompression under complete visualization, interbody fusion, and stabilization with pedicle screws. The RMM solution supports both cortical and standard trajectory techniques.
The company unveiled the XLIF Lordotic Expandable (XLX) Interbody System at last July’s International Meeting on Advanced Spine Techniques. The XLIF anterior column realignment (ACR) technique, together with XLX ACR, addresses sagittal alignment from the anterior column while reducing blood loss and hospital stays. It’s used to treat symptomatic disc degeneration or degenerative spondylolisthesis at one or two adjacent levels—including thoracic disc herniation—from L1 to S1. It can be used as an adjunct to fusion in patients with multilevel degenerative scoliosis as well.
The TLX 20 degree expandable spinal interbody implant was introduced last September. It touts a first-of-its-kind oblique profile for transforaminal lumbar interbody fusion (TLIF) procedures. The TLX 20 expandable implant’s integrated auto-lock feature allows customizable expansion for up to 20 degrees of oblique lordosis and tailoring implant expansion based on clinical need. The system also includes a single, low-profile instrument to position, expand, and post-pack the implant with bone graft, and distal tip tapering to aid insertion in a collapsed disc space.
Last November saw the U.S. launch of Brigade Lateral, at the time the industry’s first interbody system optimized for ALIF spine surgery. Brigade Lateral features improved instrumentation, a new insertion method, and an expanded lordotic implant offering for NuVasive’s proprietary Lateral ALIF procedure, which enables access to L5-S1 from a lateral position. A threaded in-line inserter angled off the midline and positioned on the implant’s lateral edge also allows better L5-S1 access.
That same month the COHERE porous PEEK implant won FDA clearance for the firm’s proprietary XLIF surgical spine procedure. The porous structure supports bone ongrowth and ingrowth, and PEEK’s radiolucent properties increase clarity in post-op X-rays and imaging. The nod allows surgeons to use the implant with autograft and/or allograft in skeletally mature patients for thoracolumbar pathologies including degenerative disc disease, degenerative spondylolisthesis, and degenerative scoliosis.
The company obtained FDA clearance for expanded use of the Monolith Corpectomy System, a modular PEEK interbody implant for cervical corpectomy procedures, last December. The expansion includes procedures to treat C3-C7 vertebral bodies damaged by fracture, tumors, or osteomyelitis, or to support reconstruction after corpectomy is performed to achieve spinal cord and neural tissue decompression in cervical degenerative disorders.
The surgical support business, which is comprised of intraoperative monitoring services, disposables, and biologics, increased sales $23.9 million—8 percent—over the previous year. Most of the growth (7 percent) came as a result of the company’s purchase of intraoperative neurophysiological monitoring (IONM) services firm SafePassage in early 2018. SafePassage provided IONM and EEG services to surgeons and healthcare facilities across the eastern U.S., and NuVasive expects to support over 100,000 IONM cases annually in the U.S. thanks to the purchase.
The AttraX Scaffold biologic, an absorbent ceramic-collagen bone graft with an optimized surface, launched last June. AttraX’s surface touts carefully defined and tightly controlled features at the submicron scale, which drives differentiation of mesenchymal stem cells into bone-forming osteoblasts without added growth factors. It’s used in the posterolateral spine to promote fusions, having demonstrated equivalent or better fusion rates than autograft and faster than ceramic grafts, according to the company.
July certainly was a busy month for NuVasive—during its last week the firm gained FDA clearance for its Pulse surgical automation platform, billed as spine’s first integrated surgical automation platform. Pulse combines 2D and 3D navigation and smart imaging, neuromonitoring, surgical planning, radiation reduction, and patient-specific rod bending technologies in one platform.
Imaging and insights can be simultaneously viewed in real-time through Wi-Fi connectivity and independent device access. Multiple high-resolution cameras combined with a low-profile, 360-degree array drives uninterrupted line-of-sight and optimized procedural workflow.
- NuVasive launched three more biologics last September as well:
- Traditional Bone Allograft—cancellous chips, demineralized cancellous chips, cortical cancellous chips, and cancellous crushed as a scaffold for bone growth.
- Amniotic Membrane DS—a double-sided layer of human amniotic membrane as a biologic barrier to prevent adhesions and reduce scar tissue formation near adjacent muscle, nerve, and fascia layer tissue.
- Propel DBM—gel, gel plus, and putty plus as additional form factors to the product line (which launched in 2017.)
“Currently, a number of hospitals and healthcare systems treat patients undergoing spine surgery through often cost-intensive, intra-operative CT scans with a general navigation system with limited utilization in spine surgery cases. The combination of NuVasive’s Pulse system and the Siemens Healthineers’ next-generation advanced imaging technology provides a compelling offering for hospitals and surgeons who require a scalable, cost-efficient technology that maximizes OR workflow efficiency and significantly improves visualization for spine surgery,” then-CEO Lucier told the press. “We firmly believe this combined offering will enable advanced clinical benefits driving superior patient outcomes, delivered through greatly improved visualization of the anatomy promoting successful anatomical access and spinal implant placement.”