08.11.20
Rank: #3 (Last year: #3)
$7.98 Billion
Prior Fiscal: $7.93 Billion
Percentage Change: +1%
No. of Employees: 19,900
Global Headquarters: Warsaw, Ind.
KEY EXECUTIVES:
Bryan Hanson, President and CEO
Carrie Nichol, VP, Controller, and Chief Accounting Officer
Suketu Upadhyay, Exec. VP and CFO
Rachel Ellingson, Sr. VP, Strategy
David Kunz, Sr. VP, Global Quality and Regulatory Affairs
Kenneth Tripp, Sr. VP, Global Operations and Logistics
Chad Phipps, Sr. VP, General Counsel, and Secretary
Angela Main, Sr. VP, Global Chief Compliance Officer and Associate General Counsel, Asia Pacific
Ivan Tornos, Group President, Global Businesses and Americas
Didier Deltort, President, Europe, Middle East, and Africa
Sang Yi, President, Asia Pacific
Steve Jobs was a notoriously difficult boss.
He yelled a lot. He cursed a lot. He was obsessively controlling and brutally honest. He was abrasive, manipulative, impatient. He was borderline tyrannical.
Jobs was as talented a bully as a sweet-talker, capable of charming enemies just as easily as he belittled friends and co-workers. He would sulk or cry to get his way. He often fired staff without warning for mistakes or subpar work.
Yet beneath that gruff, blistering exterior lurked a hard-working, creative visionary responsible for revolutionizing computers, music and digital publishing. His success in growing Apple from its humble start in his parents’ home to a $1 trillion valuation is as much the result of Jobs’s brilliance as his perfectionism.
Jobs reportedly learned the value of perfection from his father. In a 2011 biography, Jobs recounts a fence-building project from his childhood in which his father insisted on working as hard on the back of the fence as the front. “Nobody will ever know,” Jobs said, according to the book. “But you will know,” his father, Paul, replied, noting the best craftsmen use good wood on the front and back of wall cabinets; the same philosophy should apply to the fence, he reasoned. It was the mark of true artistry to have such a passion for perfection, Paul Jobs emphasized to his son.
Years later, while overseeing development of the Apple II and Macintosh computers, Jobs applied his father’s perfection principle to the machines’ circuit board. He instructed the computers’ engineers to line the chips up neatly so the board would look nice—an odd request, considering Jobs wanted the machines tightly sealed.
“Nobody is going to see the PC board,” one engineer said, prompting a familiar reply from Jobs: “I want it to be as beautiful as possible, even if it’s inside the box. A great carpenter isn’t going to use lousy wood for the back of a cabinet, even though nobody’s going to see it.”
Jobs told the engineers they were true artists, and should act that way (one can only imagine his tone). Once the board was redesigned, the biography states, Jobs had the engineers and other Macintosh team members sign their names for engraving inside the case. “Real artists sign their work,” he told the group.
Jobs’ penchant for perfection was one of the key drivers of his success. While he was not the greatest boss—at times demanding, verbally abusive, temperamental, and uncompromising—he managed to bring out the best in his employees. He helped them achieve feats they never dreamed possible.
ANALYST INSIGHTS: Zimmer Biomet (ZB) is another company impacted with a slowdown due to COVID. CEO Bryan Hansen used this event as an opportunity to continue his corporate restructuring to focus ZB on its strengths to position the firm to gain market share in the short term. By doing so, ZB now has the ability to use M&A to supplement its market positions on an opportunistic basis. As we know, both J&J and Stryker will not be giving ground easily; this will be an interesting battleground.
Steve Jobs had high standards—for himself, his employees, and his company’s innovations. In his perpetual pursuit of perfection, Jobs would typically rework a product’s first prototype to make it more to his liking. The initial iPhone design, for example, featured a glass screen set into an aluminum case; Jobs lost sleep over the arrangement, as he wanted the iPhone display to be the device’s central focus. Jobs harbored similar misgivings about the original iPad schematic (too formal-looking and uninviting), and Apple Stores layout (too focused on product categories).
Such refinements were part of Jobs’ genius. He created impeccable products, certainly, but sacrificed human decency and rectitude in order to do so. Perfectionism was the catalyst that drove him, and it drove his management style, for better or for worse.
“Leaders are complex figures sending multiple signals at the same time,” Robert Bies, a management professor at Georgetown University who researches work practices, told The Wall Street Journal in 2016. “With Jobs, there is plenty of evidence of abuse. But you can see that he’s a motivator. He was pushing the envelope for excellence in products.”
Jobs’ legacy as a difficult, if not impossible, boss is now well-known. And though he’s frequently cited as the antithesis of inspirational leadership, Jobs’ non-conformist behavior nevertheless has served as a model for thousands of CEOs, managers, wanna be geniuses, tech startup founders, and business owners looking to improve their entities’ chances of success.
Most Jobs aspirants, however, are not mimicking his behavior as much as they are emulating his uncompromising, never-settle-for-second-best work ethic.
Zimmer Biomet Holdings Inc. President and CEO Bryan C. Hanson is one such “never settle” disciple. He displayed his Jobs-like business success strategy during a Q4 2019 earnings review earlier this year, cautioning management against resting upon the company’s solid year-end performance.
“We’re encouraged by our performance in the fourth quarter. We posted solid revenue growth, slightly above our weighted average market growth rate expectations and grew earnings per share faster than revenue,” Hanson told investors on a Feb. 4 conference call. “We also made further progress in executing on our short-term priorities: supply, quality remediation, new product introductions, and ZB’s mission and culture. We also continued to invest for growth...These are all important steps forward. And while we are happy with our progress, we’re certainly not satisfied. Rest assured that we are still striving to continuously improve our business, our performance, and the value we deliver to patients, as well as customers and investors.”
Zimmer Biomet delivered on Hanson’s pledge last year, increasing total revenue 0.63 percent (2.2 percent on a constant currency basis) to $7.98 billion. Operating profit surged more than 30-fold, reaching $1.13 billion (compared with 33.8 million in 2018), and net earnings redeemed itself from a previous year loss, going from a $379.3 million deficit to $1.13 billion. Diluted earnings per share in 2019 was $5.47; adjusted diluted earnings per share was $7.87, a 3 percent rise from the prior year.
Overall, the company’s 2019 performance was a solid one. But there are definitely areas in need of improvement. Never settle.
Europe, Middle East, and Africa sales is one such area. That region posted the only loss (by geography) in 2019—revenue fell 3.3 percent to $1.74 billion. The decrease, though, was offset by a 0.82 percent rise in Americas sales (4.87 billion) and a 5 percent jump in Asia-Pacific proceeds ($1.36 billion).
Other areas needing improvement include spine/CMF and bone cement/office-based technology—the two product categories that lost money for Zimmer Biomet last year.
Never settle.
The latter genre (bone cement/office-based technology)—bundled under the company’s “Other” product reporting group—posted the largest fiscal 2019 deficit, with sales sinking 9.9 percent to $280 million. Conversely, the 2.2 percent loss posted by Zimmer Biomet’s Spine/CMF (craniomaxillofacial) business wasn’t as steep—slipping to $747.3 million vs. 2018—but it nevertheless stung, as the decline offset strong thoracic device demand and derailed any momentum gained from three U.S. Food and Drug Administration (FDA) product approvals. (For the record, Zimmer Biomet attributed Spine/CMF’s poor performance to “ongoing sales channel consolidation, price declines, and changes in foreign currency exchange rates.”).
The FDA approval trifecta began with the February 2019 sanctioning of the ROSA ONE Brain Application, a robotic platform for planning and performing minimally invasive neurological procedures. Used in cranial biopsies, tumor treatment and removal, laser brain ablation, stereo electroencephalography, ventricular and transnasal endoscopy, and epilepsy surgery, the ROSA ONE features haptic technology to create a seamless surgical interface. Its robotic arm is engineered with six degrees of freedom, which allows exceptional dexterity and flexibility to access surgical sites.
Acquired through the $132 million purchase of French robot-assisted surgical firm Medtech in 2016, the ROSA ONE technology is Zimmer Biomet’s competitive retort to rival robots jockeying for market share in the fast-growing (and increasingly crowded) orthopedic robotics sector.
Zimmer Biomet intensified the battle for that sector last year with FDA clearances for ROSA One Knee and Spine applications, but its war plan suffered an unexpected setback with the September recall of ROSA Brain. Categorized as a Class I recall, the regulatory action stemmed from a software defect that potentially compromised ROSA’s robotic arm positioning accuracy. In its recall notice, the FDA cited five complaints related to the issue and one patient injury; no related deaths were reported.
The recall involved 86 ROSA Brain 3.0 devices distributed in the United States between April 2016 and March 2019, manufactured between February 2016 and December 2019.
Though it was considerable step backward in Zimmer Biomet’s quest for orthopedic robot market share, the recall had little, if any, impact on 2019 company earnings or Spine/CMF sales. Contrarily, the main culprit impacting Spine/CMF revenue currently is the segment’s “unpredictability,” according Hanson.
“ROSA is definitely part of the equation, it’s just not the whole equation,” Hanson said on the Q4 2019 earnings conference call. “...if I just take a step back and I think about the spine business overall, I think about it in a few different ways. We clearly have not been performing the way I would like, and we’re not performing right now with predictability in that business. There are key areas that we need to concentrate on. The No. 1 thing is we have to get the channel that we have decided on to be able to take advantage of the portfolio that we have.”
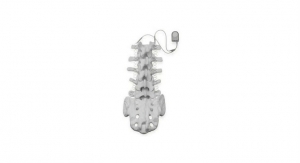
Zimmer Biomet added to that portfolio with the August 2019 clearance of a fusion-less alternative to pediatric scoliosis treatment. The Tether, a rope-like implant that pulls the vertebrae into shape, is designed for children and adolescents with severe sideways spinal curvature; the device aims to correct the curvature during a child’s growth while allowing a fuller range of motion when compared to spinal fusion. The Tether uses anchors and vertebral screws to connect a flexible cord that pulls the spine’s curved side straight (via compression) and maintains alignment as the child grows taller.
Zimmer Biomet is partnering with the Harms Study Group, an international cohort of surgeons dedicated to advancing pediatric spinal deformity treatments, to track the Tether’s long-term performance through patient registry data.
“This collaboration demonstrates how a focused, shared purpose can fundamentally change the way we approach treatment of diseases like scoliosis,” Jim Cloar, Zimmer Biomet Spine president, said upon the Tether’s FDA clearance. “Working together, clinicians, the FDA, and Zimmer Biomet have given surgeons an important fusion-less scoliosis treatment option for their pediatric patients. This procedure gives kids the best option for maintaining spine mobility and reaching their full potential.”
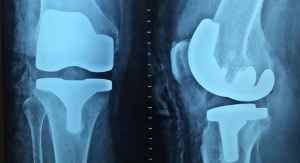
Clinicians, the FDA and Zimmer Biomet also helped adults reach their full potential last year with new knee products. Besides the ROSA ONE Knee System in January, the FDA cleared the Persona Revision Knee System, which is designed to match a patient’s anatomy for a more personalized fit. The system uses Zimmer Biomet’s Trabecular Metal technology and the Vivacit-E Highly Crosslinked Polyethylene bearing surface that protects against oxidation and maintains wear resistance and strength.
Just a week after Persona clearance (early September 2019), the FDA gave its blessing to Zimmer Biomet’s JuggerStitch Meniscal Repair Device in the United States. The implant uses two soft anchors connected by a knotless, self-locking suture loop to lock the repair, and is designed to improve both tissue preservation and tissue compression control at the surgical repair site.
The ROSA ONE and Persona Knee systems, along with the Oxford Partial Knee, were largely responsible for the 1.3 percent rise in 2019 Knee sales ($2.81 billion total). The expansion, however, was curbed by price declines and fluctuating foreign currency rates.
Those same two impediments limited fiscal 2019 growth in Hips and S.E.T. (Surgical, Sports Medicine, Biologics, Foot and Ankle, Extremities, Trauma). Robust demand for the Taperloc Complete Hip and G7 Acetabular systems drove up Hip sales 0.7 percent to $1.93 billion, while supply stability, salesforce specialization, and new product launches increased S.E.T. revenue 2.5 percent to $1.79 billion.
Dental sales climbed 0.7 percent to $414 million due to investment of resources in priority areas, and other operational improvements.
COVID-19 Consequences
Zimmer Biomet Holdings Inc. executives figured there’d be a dropoff in company earnings during Q2, but they hoped the loss might be minimized by a gradual recovery in elective surgeries.
No such luck.
Net sales plummeted 38.3 percent to $1.22 billion as COVID-19 crushed revenue around the world. Americas proceeds shrank 39.6 percent to $733.7 million, and Asia-Pacific sales slid 18.6 percent to $273.7 million. EMEA (Europe, Middle East, Africa) revenue, meanwhile, dropped an astonishing 50 percent to $218.7 million.
The losses in each product category were equally as staggering: Knees fell 46.8 percent to $374.2 million, Hips revenue declined 31 percent to $329.7 million, and SET sales (sports medicine, surgical, extremities and trauma) were down 29.2 percent to $252.6 million. Dental, Spine, and CMFT proceeds decreased 37.6 percent to $182.5 million. Zimmer Biomet’s “Other” product category posted a 44.5 percent loss against Q2 2019 figures, garnering only $87.1 million.
Despite its grim financial outlook, however, Zimmer Biomet has provided moral and monetary support to healthcare professionals, hospitals, and communities during the pandemic. It donated PPE to local hospitals (Warsaw, Ind., area) and money to non-profit organizations; it also partnered with an industry coalition, and used 3D printing technology to produce critical components for ventilators and face shields for healthcare professionals.
$7.98 Billion
Prior Fiscal: $7.93 Billion
Percentage Change: +1%
No. of Employees: 19,900
Global Headquarters: Warsaw, Ind.
KEY EXECUTIVES:
Bryan Hanson, President and CEO
Carrie Nichol, VP, Controller, and Chief Accounting Officer
Suketu Upadhyay, Exec. VP and CFO
Rachel Ellingson, Sr. VP, Strategy
David Kunz, Sr. VP, Global Quality and Regulatory Affairs
Kenneth Tripp, Sr. VP, Global Operations and Logistics
Chad Phipps, Sr. VP, General Counsel, and Secretary
Angela Main, Sr. VP, Global Chief Compliance Officer and Associate General Counsel, Asia Pacific
Ivan Tornos, Group President, Global Businesses and Americas
Didier Deltort, President, Europe, Middle East, and Africa
Sang Yi, President, Asia Pacific
Steve Jobs was a notoriously difficult boss.
He yelled a lot. He cursed a lot. He was obsessively controlling and brutally honest. He was abrasive, manipulative, impatient. He was borderline tyrannical.
Jobs was as talented a bully as a sweet-talker, capable of charming enemies just as easily as he belittled friends and co-workers. He would sulk or cry to get his way. He often fired staff without warning for mistakes or subpar work.
Yet beneath that gruff, blistering exterior lurked a hard-working, creative visionary responsible for revolutionizing computers, music and digital publishing. His success in growing Apple from its humble start in his parents’ home to a $1 trillion valuation is as much the result of Jobs’s brilliance as his perfectionism.
Jobs reportedly learned the value of perfection from his father. In a 2011 biography, Jobs recounts a fence-building project from his childhood in which his father insisted on working as hard on the back of the fence as the front. “Nobody will ever know,” Jobs said, according to the book. “But you will know,” his father, Paul, replied, noting the best craftsmen use good wood on the front and back of wall cabinets; the same philosophy should apply to the fence, he reasoned. It was the mark of true artistry to have such a passion for perfection, Paul Jobs emphasized to his son.
Years later, while overseeing development of the Apple II and Macintosh computers, Jobs applied his father’s perfection principle to the machines’ circuit board. He instructed the computers’ engineers to line the chips up neatly so the board would look nice—an odd request, considering Jobs wanted the machines tightly sealed.
“Nobody is going to see the PC board,” one engineer said, prompting a familiar reply from Jobs: “I want it to be as beautiful as possible, even if it’s inside the box. A great carpenter isn’t going to use lousy wood for the back of a cabinet, even though nobody’s going to see it.”
Jobs told the engineers they were true artists, and should act that way (one can only imagine his tone). Once the board was redesigned, the biography states, Jobs had the engineers and other Macintosh team members sign their names for engraving inside the case. “Real artists sign their work,” he told the group.
Jobs’ penchant for perfection was one of the key drivers of his success. While he was not the greatest boss—at times demanding, verbally abusive, temperamental, and uncompromising—he managed to bring out the best in his employees. He helped them achieve feats they never dreamed possible.
ANALYST INSIGHTS: Zimmer Biomet (ZB) is another company impacted with a slowdown due to COVID. CEO Bryan Hansen used this event as an opportunity to continue his corporate restructuring to focus ZB on its strengths to position the firm to gain market share in the short term. By doing so, ZB now has the ability to use M&A to supplement its market positions on an opportunistic basis. As we know, both J&J and Stryker will not be giving ground easily; this will be an interesting battleground.
—Dave Sheppard, Co-Founder and Managing Director, MedWorld Advisors
Steve Jobs had high standards—for himself, his employees, and his company’s innovations. In his perpetual pursuit of perfection, Jobs would typically rework a product’s first prototype to make it more to his liking. The initial iPhone design, for example, featured a glass screen set into an aluminum case; Jobs lost sleep over the arrangement, as he wanted the iPhone display to be the device’s central focus. Jobs harbored similar misgivings about the original iPad schematic (too formal-looking and uninviting), and Apple Stores layout (too focused on product categories).
Such refinements were part of Jobs’ genius. He created impeccable products, certainly, but sacrificed human decency and rectitude in order to do so. Perfectionism was the catalyst that drove him, and it drove his management style, for better or for worse.
“Leaders are complex figures sending multiple signals at the same time,” Robert Bies, a management professor at Georgetown University who researches work practices, told The Wall Street Journal in 2016. “With Jobs, there is plenty of evidence of abuse. But you can see that he’s a motivator. He was pushing the envelope for excellence in products.”
Jobs’ legacy as a difficult, if not impossible, boss is now well-known. And though he’s frequently cited as the antithesis of inspirational leadership, Jobs’ non-conformist behavior nevertheless has served as a model for thousands of CEOs, managers, wanna be geniuses, tech startup founders, and business owners looking to improve their entities’ chances of success.
Most Jobs aspirants, however, are not mimicking his behavior as much as they are emulating his uncompromising, never-settle-for-second-best work ethic.
Zimmer Biomet Holdings Inc. President and CEO Bryan C. Hanson is one such “never settle” disciple. He displayed his Jobs-like business success strategy during a Q4 2019 earnings review earlier this year, cautioning management against resting upon the company’s solid year-end performance.
“We’re encouraged by our performance in the fourth quarter. We posted solid revenue growth, slightly above our weighted average market growth rate expectations and grew earnings per share faster than revenue,” Hanson told investors on a Feb. 4 conference call. “We also made further progress in executing on our short-term priorities: supply, quality remediation, new product introductions, and ZB’s mission and culture. We also continued to invest for growth...These are all important steps forward. And while we are happy with our progress, we’re certainly not satisfied. Rest assured that we are still striving to continuously improve our business, our performance, and the value we deliver to patients, as well as customers and investors.”
Zimmer Biomet delivered on Hanson’s pledge last year, increasing total revenue 0.63 percent (2.2 percent on a constant currency basis) to $7.98 billion. Operating profit surged more than 30-fold, reaching $1.13 billion (compared with 33.8 million in 2018), and net earnings redeemed itself from a previous year loss, going from a $379.3 million deficit to $1.13 billion. Diluted earnings per share in 2019 was $5.47; adjusted diluted earnings per share was $7.87, a 3 percent rise from the prior year.
Overall, the company’s 2019 performance was a solid one. But there are definitely areas in need of improvement. Never settle.
Europe, Middle East, and Africa sales is one such area. That region posted the only loss (by geography) in 2019—revenue fell 3.3 percent to $1.74 billion. The decrease, though, was offset by a 0.82 percent rise in Americas sales (4.87 billion) and a 5 percent jump in Asia-Pacific proceeds ($1.36 billion).
Other areas needing improvement include spine/CMF and bone cement/office-based technology—the two product categories that lost money for Zimmer Biomet last year.
Never settle.
The latter genre (bone cement/office-based technology)—bundled under the company’s “Other” product reporting group—posted the largest fiscal 2019 deficit, with sales sinking 9.9 percent to $280 million. Conversely, the 2.2 percent loss posted by Zimmer Biomet’s Spine/CMF (craniomaxillofacial) business wasn’t as steep—slipping to $747.3 million vs. 2018—but it nevertheless stung, as the decline offset strong thoracic device demand and derailed any momentum gained from three U.S. Food and Drug Administration (FDA) product approvals. (For the record, Zimmer Biomet attributed Spine/CMF’s poor performance to “ongoing sales channel consolidation, price declines, and changes in foreign currency exchange rates.”).
The FDA approval trifecta began with the February 2019 sanctioning of the ROSA ONE Brain Application, a robotic platform for planning and performing minimally invasive neurological procedures. Used in cranial biopsies, tumor treatment and removal, laser brain ablation, stereo electroencephalography, ventricular and transnasal endoscopy, and epilepsy surgery, the ROSA ONE features haptic technology to create a seamless surgical interface. Its robotic arm is engineered with six degrees of freedom, which allows exceptional dexterity and flexibility to access surgical sites.
Acquired through the $132 million purchase of French robot-assisted surgical firm Medtech in 2016, the ROSA ONE technology is Zimmer Biomet’s competitive retort to rival robots jockeying for market share in the fast-growing (and increasingly crowded) orthopedic robotics sector.
Zimmer Biomet intensified the battle for that sector last year with FDA clearances for ROSA One Knee and Spine applications, but its war plan suffered an unexpected setback with the September recall of ROSA Brain. Categorized as a Class I recall, the regulatory action stemmed from a software defect that potentially compromised ROSA’s robotic arm positioning accuracy. In its recall notice, the FDA cited five complaints related to the issue and one patient injury; no related deaths were reported.
The recall involved 86 ROSA Brain 3.0 devices distributed in the United States between April 2016 and March 2019, manufactured between February 2016 and December 2019.
Though it was considerable step backward in Zimmer Biomet’s quest for orthopedic robot market share, the recall had little, if any, impact on 2019 company earnings or Spine/CMF sales. Contrarily, the main culprit impacting Spine/CMF revenue currently is the segment’s “unpredictability,” according Hanson.
“ROSA is definitely part of the equation, it’s just not the whole equation,” Hanson said on the Q4 2019 earnings conference call. “...if I just take a step back and I think about the spine business overall, I think about it in a few different ways. We clearly have not been performing the way I would like, and we’re not performing right now with predictability in that business. There are key areas that we need to concentrate on. The No. 1 thing is we have to get the channel that we have decided on to be able to take advantage of the portfolio that we have.”
Zimmer Biomet added to that portfolio with the August 2019 clearance of a fusion-less alternative to pediatric scoliosis treatment. The Tether, a rope-like implant that pulls the vertebrae into shape, is designed for children and adolescents with severe sideways spinal curvature; the device aims to correct the curvature during a child’s growth while allowing a fuller range of motion when compared to spinal fusion. The Tether uses anchors and vertebral screws to connect a flexible cord that pulls the spine’s curved side straight (via compression) and maintains alignment as the child grows taller.
Zimmer Biomet is partnering with the Harms Study Group, an international cohort of surgeons dedicated to advancing pediatric spinal deformity treatments, to track the Tether’s long-term performance through patient registry data.
“This collaboration demonstrates how a focused, shared purpose can fundamentally change the way we approach treatment of diseases like scoliosis,” Jim Cloar, Zimmer Biomet Spine president, said upon the Tether’s FDA clearance. “Working together, clinicians, the FDA, and Zimmer Biomet have given surgeons an important fusion-less scoliosis treatment option for their pediatric patients. This procedure gives kids the best option for maintaining spine mobility and reaching their full potential.”
Clinicians, the FDA and Zimmer Biomet also helped adults reach their full potential last year with new knee products. Besides the ROSA ONE Knee System in January, the FDA cleared the Persona Revision Knee System, which is designed to match a patient’s anatomy for a more personalized fit. The system uses Zimmer Biomet’s Trabecular Metal technology and the Vivacit-E Highly Crosslinked Polyethylene bearing surface that protects against oxidation and maintains wear resistance and strength.
Just a week after Persona clearance (early September 2019), the FDA gave its blessing to Zimmer Biomet’s JuggerStitch Meniscal Repair Device in the United States. The implant uses two soft anchors connected by a knotless, self-locking suture loop to lock the repair, and is designed to improve both tissue preservation and tissue compression control at the surgical repair site.
The ROSA ONE and Persona Knee systems, along with the Oxford Partial Knee, were largely responsible for the 1.3 percent rise in 2019 Knee sales ($2.81 billion total). The expansion, however, was curbed by price declines and fluctuating foreign currency rates.
Those same two impediments limited fiscal 2019 growth in Hips and S.E.T. (Surgical, Sports Medicine, Biologics, Foot and Ankle, Extremities, Trauma). Robust demand for the Taperloc Complete Hip and G7 Acetabular systems drove up Hip sales 0.7 percent to $1.93 billion, while supply stability, salesforce specialization, and new product launches increased S.E.T. revenue 2.5 percent to $1.79 billion.
Dental sales climbed 0.7 percent to $414 million due to investment of resources in priority areas, and other operational improvements.
COVID-19 Consequences
Q2 2019 Revenue: $1.22 Billion
Q2 2020 Revenue: $1.98 Billion
Percentage Change: -38.3%
Zimmer Biomet Holdings Inc. executives figured there’d be a dropoff in company earnings during Q2, but they hoped the loss might be minimized by a gradual recovery in elective surgeries.
No such luck.
Net sales plummeted 38.3 percent to $1.22 billion as COVID-19 crushed revenue around the world. Americas proceeds shrank 39.6 percent to $733.7 million, and Asia-Pacific sales slid 18.6 percent to $273.7 million. EMEA (Europe, Middle East, Africa) revenue, meanwhile, dropped an astonishing 50 percent to $218.7 million.
The losses in each product category were equally as staggering: Knees fell 46.8 percent to $374.2 million, Hips revenue declined 31 percent to $329.7 million, and SET sales (sports medicine, surgical, extremities and trauma) were down 29.2 percent to $252.6 million. Dental, Spine, and CMFT proceeds decreased 37.6 percent to $182.5 million. Zimmer Biomet’s “Other” product category posted a 44.5 percent loss against Q2 2019 figures, garnering only $87.1 million.
Despite its grim financial outlook, however, Zimmer Biomet has provided moral and monetary support to healthcare professionals, hospitals, and communities during the pandemic. It donated PPE to local hospitals (Warsaw, Ind., area) and money to non-profit organizations; it also partnered with an industry coalition, and used 3D printing technology to produce critical components for ventilators and face shields for healthcare professionals.