08.11.20
Rank: #4 (Last year: #4)
$5.13 Billion
Prior Fiscal: $4.90 Billion
Percentage Change: +4.7%
No. of Employees: 17,637
Global Headquarters: London, United Kingdom
KEY EXECUTIVES:
Roland Diggelmann, CEO
Ian Melling, Interim CFO
Brad Cannon, President, Sports Medicine and Ear, Nose and Throat
Mark Gladwell, President, Global Operations
Simon Fraser, President, Advanced Wound Management
Skil Kiil, President, Orthopaedics
Vasant Padmanabhan, President, Research & Development
“…it is an honor to become the new chief executive of Smith+Nephew, a company I greatly admire,” Namal Nawana proclaimed when he took the helm in 2018. However, as it turns out, his enthusiasm wasn’t worth the $7 million annual package the company offered him, because he quit eighteen months later.
Technically speaking, his exit was “by mutual agreement” according to Smith+Nephew. But according to The Guardian, Nawana’s request for higher pay—around the $15 million mark, comparable to packages awarded by U.S. medical device makers—couldn’t be met under UK corporate governance standards. The board even discussed relocating the company to the U.S. during the summer, The Financial Times reported.
Perhaps Nawana thought he could use the leverage gained from boosting Smith+Nephew’s share price. Under his purview the UK-based firm’s annual revenue grew 2.9 percent in 2018 and an additional 4.7 percent last year, topping $5 billion in sales for the first time with 2019 proceeds of $5.13 billion. But once the board declined and asserted UK shareholders would never approve that high of a package, Nawana reportedly said he might not hang around—likely prompting an invitation to clear his desk.
That could be perceived as “mutual agreement,” but there’s no doubt who started the fight.
Roche Diagnostics CEO Roland Diggelmann—who was considered to lead Smith+Nephew in 2017, according to Berenberg analysts—filled Nawana’s vacancy last November, to the tune of an up to $7.1 million annual package. Before his 11-year career at Roche, he spent 12 years in orthopedics in strategy and leadership roles at Sulzer Orthopedics and Zimmer. He’s currently based at the company’s site in Baar, Switzerland.
“…I look forward to playing my part in shaping the future of the company and driving the next stage of growth,” Diggelmann said when he was revealed as the new CEO.
ANALYST INSIGHTS: It is beginning to be clear that the change of CEO in the past year has had an interesting impact on the culture of S&N. Moving from an aggressive CEO in Namal Nawana to a more conservative (former Roche) CEO in Roland Diggelman, expect M&A to play a lesser role in the culture of S&N going forward. It’ll be interesting to observe if S&N can stand in its own right against the major players or need to be acquired to stay competitive in a very dynamic market segment.
Smith+Nephew was well-positioned for growth as of fiscal 2019’s end (Dec 31). The company’s Orthopaedic franchise revenue grew 2.5 percent to $2.22 billion last year. Smith+Nephew commands 12 percent of the hip and knee implant and 8 percent of the trauma and extremities markets per its 2019 annual report. Sales from knee implants rose 2.5 percent to $1.04 billion, led by strong demand outside the U.S. for the JOURNEY II and LEGION revision knee systems. Hip implant earnings grew 2.1 percent to $613 million, led by demand for the POLAR3 total hip and REDAPT revision hip systems.
Hip and knee product releases were quiet until Q4, when Smith+Nephew addressed the growing dual mobility segment—which, according to the Journal of Arthroplasty account for 9 percent of primary and 28 percent of revision hip replacements.1 The OR3O Dual Mobility System for primary and revision hip replacements hit the shelves in November. According to The Bone & Joint Journal, dual mobility hip implant components boost stability and range of motion thanks to a smaller diameter femoral head that locks into a larger polyethylene insert compared with traditional hip implants.2 OR3O is cross-compatible with the company’s R3 and REDAPT acetabular systems.
Trauma and extremities revenue swelled 2.4 percent to reach $488 million. Sustained double-digit growth from the INTERTAN intertrochateric antegrade nail, as well as the EVOS System’s roll-out, provoked this segment’s boost.
June saw the unveiling of CONQUEST FN, a telescoping compression screw and proximal femoral locking plate system for femoral neck fractures. Femoral neck procedures exhibit complication rates as high as 69 percent,3 according to Advanced Biomedical Research. The POGO screw provides continuous post-op compression to decrease hip pain and complications, which the plate’s dynamic locking fixation offers more control during treatment.
“…post-operatively, patients often follow-up very comfortable with minimal pain, and have progressed to weight-bearing more quickly. I feel this speaks to the enhanced stability of the implant,” Daniel Chan, M.D., an Orthopaedic Trauma Institute orthopedic traumatologist told the press.
The EVOS WRIST plating system launched at last September’s American Society for Surgery of the Hand annual meeting. Stainless steel and titanium volar plate options with variable- and fixed-angle locking, dorsal plates, and forearm-specific plates can facilitate simple and complex wrist fracture surgeries. Titanium distal ulna, intermediate column, radial column, and dorsal buttress plates encompass the suite of fragment-specific plates.
The Other Reconstruction franchise, which contains the NAVIO robotics-assisted surgical systems swelled an impressive 27.9 percent to reach $79 million. NAVIO can be used for both partial and total knee procedures and according to the company is the first and only robotics-assisted bi-cruciate retaining knee procedure available.
Smith+Nephew bought Brainlab’s orthopedic joint reconstruction business for an undisclosed amount in June. Used by over 500 hospitals, the portfolio includes digital workflow tools for pre-operative planning, intraoperative navigation, post-operative evaluation, and sharing. The technologies encompass cloud computing, tracking, augmented reality, robotics, AI, machine learning, image fusion, and anatomical segmentation. Smith+Nephew plans to integrate the technology into the next iteration of its surgical robotics system, NAVIO 7.0. The two companies will also collaborate to develop additional applications to bolster Smith+Nephew’s digital surgery system, including orthopedic reconstruction and sports medicine.
The company also acquired Switzerland-based Atracsys, maker of optical tracking technology for computer-assisted surgery, in June. Its fusionTrack 500 optical tracking camera will enhance Smith+Nephew’s digital surgery and robotic ecosystem, reducing procedure times and increasing accuracy in precision surgical tasks. Atracsys’ portfolio supports orthopedic, neurosurgical, spinal, and dental applications.
Sports Medicine & ENT segment revenue rose 5.1 percent to $1.54 billion. Sports Medicine Joint Repair proceeds ballooned 10.8 percent to $794 million, exhibiting four straight quarters of double-digit growth. There was consistent performance along knee and shoulder repair ranges, thanks to increased fervor for the REGENETEN bioinductive rotator cuff implant and NOVOSTITCH meniscal repair system.
Smith+Nephew released results for the STITCH Study in July. This study used NOVOSTITCH for the first-ever trial evaluating horizontal cleavage tear repair effectiveness. It was based on three endpoints: meniscal healing upon second look, freedom from re-operation, and knee pain/function improvements according to patient reported outcome (PRO) scores. Six-month results showed significant PRO improvement, 0 percent reoperation rate, and 100 percent meniscal healing.
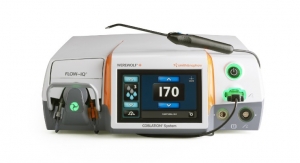
Arthroscopic Enabling Technology income dropped 1.5 percent to $591 million last year, but two strong product launches finished off the year. The FLOW 90 Wand for the firm’s WEREWOLF COBLATION radio-frequency arthroscopy system was released last May. The 90-degree wand was designed for shoulder procedures but is indicated for all soft tissue types.
“…It excels at rapid bulk tissue removal while remaining very precise. FLOW 90’s triangular tip also enables you to do excellent dissection,” Dr. Scott Trenhaile, assistant professor, Rush University Medical Center, commented to the press.
The LENS 4K Surgical Imaging System for arthroscopic procedures arrived in late September. Its 4K 3CMOS ultra high definition camera’s control unit features an integrated light source, autoclavable camera head with programmable buttons, image management options, and tablet application to control the camera outside of the surgical field. Smith+Nephew expects LENS 4K to be particularly helpful to ambulatory and multi-surgery centers.
Advanced Wound Management sales grew 8.2 percent to achieve $1.38 billion in earnings. Advanced Wound Devices proceeds expanded 12.8 percent to $242 million, thanks to continued strong demand for PICO negative pressure wound therapy system and increasing contribution from the RENASYS negative pressure wound therapy system.
September proved to be a busy month, as the PICO 7Y single-use negative pressure wound therapy system (sNPWT) also hit the shelves. Its integrated Y connector enables two dressing to be used concurrently from one pump, meaning two wounds or incisions can be simultaneously addressed. (According to the company, PICO 7Y is particularly suited for breast procedures.) It also includes a check dressing indicator and is quieter than the first-generation PICO.
The firm acquired 85.5 percent of the share capital of Leaf Healthcare in April for an undisclosed amount (bringing Smith+Nephew’s total shareholding to 100 percent) following a successful two-year partnership. Leaf Healthcare’s Leaf Patient Monitoring System is a small, lightweight sensor that wirelessly monitors a patient’s position and mobility in the hospital. Tracking data gathered by the sensor helps to automate and document compliance with prescribed turn protocols for patients at risk of pressure injuries. In January 2018, a Stanford clinical trial found the system induced a 43 percent relative increase in turning protocol compliance in high-risk patients. Patients treated using the Leaf sensor were also found to be 73 percent less likely to develop a pressure injury.
Advanced Wound Care declined 3.5 percent, falling to $714 million because U.S. price pressure could not offset stronger European market performance. Advanced Wound Bioactives, however, skyrocketed 32.3 percent to $424 million thanks to the acquisition of regenerative medicine company Osiris Therapeutics.
Purchased for $660.5 million last April, Osiris manufactures placental, bone, and cartilage tissue allografts. A tissue cryopreservation technique is used to retain native tissue components (like mesynchymal stem cells), growth factors and extracellular matrix, and the tissue’s inherent functionalities. The firm’s Grafix and Stravix are used for wound covers, BIO4 for bone repair and regeneration, and Cartiform for articular cartilage repair and regeneration. According to Smith+Nephew, the skin substitute market is one of the highest growth and high potential wound management markets.
The company also prevailed in another battle in a patent dispute with Arthrex in August. A U.S. Court of Appeals panel rejected Arthrex’s attempt to overturn a ruling that two claims of an Arthrex surgical suture anchor patent are unpatentable. This loss was the second blow to Arthrex in a long-running dispute.
References
COVID-19 Consequences
“There remain many uncertainties as countries continue to battle COVID-19, but with our unique portfolio, proven strategy, strong balance sheet, and motivated workforce we are ready to take advantage as markets recover,” CEO Roland Diggelmann commented in the company’s press release detailing Q2 performance.
Smith+Nephew confirmed a contract with the UK government on April 1 to build OxVent ventilators, which are designed to allow for large scale production. OxVent ventilators are the result of joint collaboration between Smith+Nephew, the University of Oxford, and King’s College London. Government support was pulled from the project on May 4 due to actual clinical need and reviews of those put forward. The OxVent team is engaging with colleagues abroad from the U.K., where the need for ventilators is greater.
$5.13 Billion
Prior Fiscal: $4.90 Billion
Percentage Change: +4.7%
No. of Employees: 17,637
Global Headquarters: London, United Kingdom
KEY EXECUTIVES:
Roland Diggelmann, CEO
Ian Melling, Interim CFO
Brad Cannon, President, Sports Medicine and Ear, Nose and Throat
Mark Gladwell, President, Global Operations
Simon Fraser, President, Advanced Wound Management
Skil Kiil, President, Orthopaedics
Vasant Padmanabhan, President, Research & Development
“…it is an honor to become the new chief executive of Smith+Nephew, a company I greatly admire,” Namal Nawana proclaimed when he took the helm in 2018. However, as it turns out, his enthusiasm wasn’t worth the $7 million annual package the company offered him, because he quit eighteen months later.
Technically speaking, his exit was “by mutual agreement” according to Smith+Nephew. But according to The Guardian, Nawana’s request for higher pay—around the $15 million mark, comparable to packages awarded by U.S. medical device makers—couldn’t be met under UK corporate governance standards. The board even discussed relocating the company to the U.S. during the summer, The Financial Times reported.
Perhaps Nawana thought he could use the leverage gained from boosting Smith+Nephew’s share price. Under his purview the UK-based firm’s annual revenue grew 2.9 percent in 2018 and an additional 4.7 percent last year, topping $5 billion in sales for the first time with 2019 proceeds of $5.13 billion. But once the board declined and asserted UK shareholders would never approve that high of a package, Nawana reportedly said he might not hang around—likely prompting an invitation to clear his desk.
That could be perceived as “mutual agreement,” but there’s no doubt who started the fight.
Roche Diagnostics CEO Roland Diggelmann—who was considered to lead Smith+Nephew in 2017, according to Berenberg analysts—filled Nawana’s vacancy last November, to the tune of an up to $7.1 million annual package. Before his 11-year career at Roche, he spent 12 years in orthopedics in strategy and leadership roles at Sulzer Orthopedics and Zimmer. He’s currently based at the company’s site in Baar, Switzerland.
“…I look forward to playing my part in shaping the future of the company and driving the next stage of growth,” Diggelmann said when he was revealed as the new CEO.
ANALYST INSIGHTS: It is beginning to be clear that the change of CEO in the past year has had an interesting impact on the culture of S&N. Moving from an aggressive CEO in Namal Nawana to a more conservative (former Roche) CEO in Roland Diggelman, expect M&A to play a lesser role in the culture of S&N going forward. It’ll be interesting to observe if S&N can stand in its own right against the major players or need to be acquired to stay competitive in a very dynamic market segment.
—Dave Sheppard, Co-Founder and Managing Director, MedWorld Advisors
Smith+Nephew was well-positioned for growth as of fiscal 2019’s end (Dec 31). The company’s Orthopaedic franchise revenue grew 2.5 percent to $2.22 billion last year. Smith+Nephew commands 12 percent of the hip and knee implant and 8 percent of the trauma and extremities markets per its 2019 annual report. Sales from knee implants rose 2.5 percent to $1.04 billion, led by strong demand outside the U.S. for the JOURNEY II and LEGION revision knee systems. Hip implant earnings grew 2.1 percent to $613 million, led by demand for the POLAR3 total hip and REDAPT revision hip systems.
Hip and knee product releases were quiet until Q4, when Smith+Nephew addressed the growing dual mobility segment—which, according to the Journal of Arthroplasty account for 9 percent of primary and 28 percent of revision hip replacements.1 The OR3O Dual Mobility System for primary and revision hip replacements hit the shelves in November. According to The Bone & Joint Journal, dual mobility hip implant components boost stability and range of motion thanks to a smaller diameter femoral head that locks into a larger polyethylene insert compared with traditional hip implants.2 OR3O is cross-compatible with the company’s R3 and REDAPT acetabular systems.
Trauma and extremities revenue swelled 2.4 percent to reach $488 million. Sustained double-digit growth from the INTERTAN intertrochateric antegrade nail, as well as the EVOS System’s roll-out, provoked this segment’s boost.
June saw the unveiling of CONQUEST FN, a telescoping compression screw and proximal femoral locking plate system for femoral neck fractures. Femoral neck procedures exhibit complication rates as high as 69 percent,3 according to Advanced Biomedical Research. The POGO screw provides continuous post-op compression to decrease hip pain and complications, which the plate’s dynamic locking fixation offers more control during treatment.
“…post-operatively, patients often follow-up very comfortable with minimal pain, and have progressed to weight-bearing more quickly. I feel this speaks to the enhanced stability of the implant,” Daniel Chan, M.D., an Orthopaedic Trauma Institute orthopedic traumatologist told the press.
The EVOS WRIST plating system launched at last September’s American Society for Surgery of the Hand annual meeting. Stainless steel and titanium volar plate options with variable- and fixed-angle locking, dorsal plates, and forearm-specific plates can facilitate simple and complex wrist fracture surgeries. Titanium distal ulna, intermediate column, radial column, and dorsal buttress plates encompass the suite of fragment-specific plates.
The Other Reconstruction franchise, which contains the NAVIO robotics-assisted surgical systems swelled an impressive 27.9 percent to reach $79 million. NAVIO can be used for both partial and total knee procedures and according to the company is the first and only robotics-assisted bi-cruciate retaining knee procedure available.
Smith+Nephew bought Brainlab’s orthopedic joint reconstruction business for an undisclosed amount in June. Used by over 500 hospitals, the portfolio includes digital workflow tools for pre-operative planning, intraoperative navigation, post-operative evaluation, and sharing. The technologies encompass cloud computing, tracking, augmented reality, robotics, AI, machine learning, image fusion, and anatomical segmentation. Smith+Nephew plans to integrate the technology into the next iteration of its surgical robotics system, NAVIO 7.0. The two companies will also collaborate to develop additional applications to bolster Smith+Nephew’s digital surgery system, including orthopedic reconstruction and sports medicine.
The company also acquired Switzerland-based Atracsys, maker of optical tracking technology for computer-assisted surgery, in June. Its fusionTrack 500 optical tracking camera will enhance Smith+Nephew’s digital surgery and robotic ecosystem, reducing procedure times and increasing accuracy in precision surgical tasks. Atracsys’ portfolio supports orthopedic, neurosurgical, spinal, and dental applications.
Sports Medicine & ENT segment revenue rose 5.1 percent to $1.54 billion. Sports Medicine Joint Repair proceeds ballooned 10.8 percent to $794 million, exhibiting four straight quarters of double-digit growth. There was consistent performance along knee and shoulder repair ranges, thanks to increased fervor for the REGENETEN bioinductive rotator cuff implant and NOVOSTITCH meniscal repair system.
Smith+Nephew released results for the STITCH Study in July. This study used NOVOSTITCH for the first-ever trial evaluating horizontal cleavage tear repair effectiveness. It was based on three endpoints: meniscal healing upon second look, freedom from re-operation, and knee pain/function improvements according to patient reported outcome (PRO) scores. Six-month results showed significant PRO improvement, 0 percent reoperation rate, and 100 percent meniscal healing.
Arthroscopic Enabling Technology income dropped 1.5 percent to $591 million last year, but two strong product launches finished off the year. The FLOW 90 Wand for the firm’s WEREWOLF COBLATION radio-frequency arthroscopy system was released last May. The 90-degree wand was designed for shoulder procedures but is indicated for all soft tissue types.
“…It excels at rapid bulk tissue removal while remaining very precise. FLOW 90’s triangular tip also enables you to do excellent dissection,” Dr. Scott Trenhaile, assistant professor, Rush University Medical Center, commented to the press.
The LENS 4K Surgical Imaging System for arthroscopic procedures arrived in late September. Its 4K 3CMOS ultra high definition camera’s control unit features an integrated light source, autoclavable camera head with programmable buttons, image management options, and tablet application to control the camera outside of the surgical field. Smith+Nephew expects LENS 4K to be particularly helpful to ambulatory and multi-surgery centers.
Advanced Wound Management sales grew 8.2 percent to achieve $1.38 billion in earnings. Advanced Wound Devices proceeds expanded 12.8 percent to $242 million, thanks to continued strong demand for PICO negative pressure wound therapy system and increasing contribution from the RENASYS negative pressure wound therapy system.
September proved to be a busy month, as the PICO 7Y single-use negative pressure wound therapy system (sNPWT) also hit the shelves. Its integrated Y connector enables two dressing to be used concurrently from one pump, meaning two wounds or incisions can be simultaneously addressed. (According to the company, PICO 7Y is particularly suited for breast procedures.) It also includes a check dressing indicator and is quieter than the first-generation PICO.
The firm acquired 85.5 percent of the share capital of Leaf Healthcare in April for an undisclosed amount (bringing Smith+Nephew’s total shareholding to 100 percent) following a successful two-year partnership. Leaf Healthcare’s Leaf Patient Monitoring System is a small, lightweight sensor that wirelessly monitors a patient’s position and mobility in the hospital. Tracking data gathered by the sensor helps to automate and document compliance with prescribed turn protocols for patients at risk of pressure injuries. In January 2018, a Stanford clinical trial found the system induced a 43 percent relative increase in turning protocol compliance in high-risk patients. Patients treated using the Leaf sensor were also found to be 73 percent less likely to develop a pressure injury.
Advanced Wound Care declined 3.5 percent, falling to $714 million because U.S. price pressure could not offset stronger European market performance. Advanced Wound Bioactives, however, skyrocketed 32.3 percent to $424 million thanks to the acquisition of regenerative medicine company Osiris Therapeutics.
Purchased for $660.5 million last April, Osiris manufactures placental, bone, and cartilage tissue allografts. A tissue cryopreservation technique is used to retain native tissue components (like mesynchymal stem cells), growth factors and extracellular matrix, and the tissue’s inherent functionalities. The firm’s Grafix and Stravix are used for wound covers, BIO4 for bone repair and regeneration, and Cartiform for articular cartilage repair and regeneration. According to Smith+Nephew, the skin substitute market is one of the highest growth and high potential wound management markets.
The company also prevailed in another battle in a patent dispute with Arthrex in August. A U.S. Court of Appeals panel rejected Arthrex’s attempt to overturn a ruling that two claims of an Arthrex surgical suture anchor patent are unpatentable. This loss was the second blow to Arthrex in a long-running dispute.
References
- https://pubmed.ncbi.nlm.nih.gov/30711371/
- https://pubmed.ncbi.nlm.nih.gov/29305445/
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3732879/
COVID-19 Consequences
Q2 2020 Revenue: $901 Million
Q2 2019 Revenue: $1.28 Billion
Percentage Change: -29.3%
“There remain many uncertainties as countries continue to battle COVID-19, but with our unique portfolio, proven strategy, strong balance sheet, and motivated workforce we are ready to take advantage as markets recover,” CEO Roland Diggelmann commented in the company’s press release detailing Q2 performance.
Smith+Nephew confirmed a contract with the UK government on April 1 to build OxVent ventilators, which are designed to allow for large scale production. OxVent ventilators are the result of joint collaboration between Smith+Nephew, the University of Oxford, and King’s College London. Government support was pulled from the project on May 4 due to actual clinical need and reviews of those put forward. The OxVent team is engaging with colleagues abroad from the U.K., where the need for ventilators is greater.