Mark Crawford , Contributing Writer03.22.13
When it comes to materials for orthopedic devices, OEMs have a lot to keep up with. Metal, once the biomaterial of choice, increasingly is being replaced with durable, high-performance plastics—not only because of metal-on-metal wear issues, but due to strength-to-weight ratios and corrosion resistance. In particular, polyetheretherketone (PEEK) is receiving more interest as a viable replacement for metal in joint implants.
Plastics manufacturers constantly innovate to meet evolving market demands. Design engineers are looking for plastics with specific combinations of performance characteristics, such as enhanced strength, flexibility and bioabsorbability. They also want to use materials that already are understood and approved by the U.S. Food and Drug Administration (FDA) to streamline approvals and keep costs down.
“Almost monthly it seems like a client is reacting to a market desire—sometimes existing, sometimes anticipated,” said Bing J. Carbone, president of Shelton, Conn.-based Modern Plastics Inc., a plastics distributor to the medical industry. “We stay on top of these developments so we can provide customers with up-to-the-minute advancements on new material options and services that we can offer.”
A tremendous amount of research is being conducted on creating new biocompatible compounds or improving existing materials by developing new blends. Although these new materials may have fantastic properties, many companies often are cautious about embracing them because it may mean gathering more data or conducting extensive trials. Instead, they combine materials that already have a proven history of effectiveness and FDA compliance to create blended materials that best meet the product’s performance specifications.
The FDA also has raised the regulatory bar in recent years, especially regarding component biocompatibility and supply chain management. OEMs increasingly expect their material providers to know the latest regulations and maintain top-level quality management systems that ideally include ISO 9001 and ISO 13485 certification. Material providers must constantly improve their processes, quality assurance methods and standard operating procedures to stay ahead of the curve and remain competitive. Because material lead times often are extended, expert and sometimes creative inventory management is required to meet short production deadlines.
What OEMs Want
OEMs want supply chain partners that can provide risk reduction (safety, reliability, track record of clinical success and security of supply), reliable and high-performance materials and the ability to access regulatory data from global markets. OEMs also hope their material providers are experts at supply chain efficiency, inventory management and serving emerging markets.
“Trends in material selection continue to be toward risk management and assessment with respect to the material being evaluated, as well as its biocompatibility,” said Mary Moynihan-Downes, director of the medical market for Rockaway, N.J.-based Titanium Industries Inc., a manufacturing distributor of metal. “Once those factors are satisfied, economics and availability become the key elements to be considered.”
There also are demands from the production and manufacturing side. “Almost every OEM engineer I talk with says there is pressure from management for smaller implants and instruments with tighter tolerances,” said Grant Niewinski, U.S. sales and marketing manager for Forecreu America Inc., a Chicago, Ill.-based producer of stainless steel and titanium cannulated bars for the orthopedics market. All these expectations challenge the design process, especially material selection.
As a result, design engineers are searching for more custom blends with specific features such as strength, hardness, flexibility, heat resistance, chemical resistance, etc.
“For example,” said Carbone, “barium sulfates, carbons, glass, anti-microbial and other fillers are being added to plastics to enhance characteristics such as strength, heat resistance, radiopacity and infection resistance. Although some of these fillers have been used for years, they are increasingly being combined in different ways to produce some novel products that should be introduced to the general market very soon.”
OEMs also want to launch their new product lines quickly—there is an ever-present push for speed, which also can impact the final material decision.
“Utilizing a material that is already associated with an ASTM [formerly the American Society for Testing and Materials] or ISO standard often influences a company's willingness to move forward,” commented Scott Shoppell, marketing manager for Fort Wayne, Ind.-based Fort Wayne Metals, a provider of research, development and production of fine grade medical wire, rod, cable, metal-on-metal composites and custom assemblies. “Some companies will not commit time and resources to developing materials that don’t already have an implant-grade specification. Companies with more knowledgeable and experienced personnel in their technical and materials groups tend to be less intimidated by the possibility of blazing trails with innovative materials.”
Material Trends
Popular materials in the orthopedic industry include high-grade polyester, polyethylene, metals and alloys such as stainless steel, titanium and cobalt chromium. For many years, the medical device industry’s mind-set has been to replace impaired tissue—for example, replacing cartilage or bone with synthetic materials.
More recently, researchers and medical companies have been seeking alternative materials that also can induce regeneration and restoration of the impaired tissue. Bioresorbable materials, including polyglycolic acid, polydioxanone, polylactic acid and polycaprolactone, are more compatible with surrounding tissues and the body’s own natural healing processes.
“Bioresorbable materials can be used as a temporary scaffold until the tissue’s intended function is restored,” indicated Swati Amin, advanced biomaterials engineer for Secant Medical Inc., a Perkasie, Pa.-based designer and manufacturer of biomedical textiles and material structures for medical devices. “For example, these materials can be engineered and processed to achieve a desired degradation time that matches the time it takes for the native tissue to heal itself.”
One of the biggest challenges in manufacturing these types of materials, noted Amin, is the stability of the polymer. “Oxygen, heat, or light can alter these sensitive materials and propagate a cascade of oxidation, causing unwanted degradation prior to implantation,” she added. “Knowing this during polymer processing is essential.”
The ongoing problems with metal-on-metal hip implants are driving more interest in metal-free alternatives for these implant devices that have just as much strength and durability as metal—especially PEEK.
“Given that PEEK polymers are stronger than many metals on a per-mass basis, these materials are increasingly selected as an alternative to metal,” stated John Devine, marketing and technology director for Invibio Biomaterial Solutions, a provider of PEEK-based biomaterials based in Thornton Cleveleys, in the United Kingdom. “The manufacturing efficiency gains for metals are also reaching a plateau; in order to reduce costs, device manufacturers are looking to alternative processes such as injection molding.”
An advanced form of PEEK is Invibio’s PEEK-Optima Ultra Reinforced, which offers semi-rigid fixation with improved fatigue performance and radiolucency. “It combines the high-performance material properties of PEEK-Optima with additional strength imparted by carbon fiber, providing an alternative to metal biomaterials traditionally used in internal fracture fixation procedures,” said Devine.
Carbone indicated that more OEMs are considering PEEK for a broader range of applications, including newer spine, orthopedic, dental, small-bone and cranial applications, as well as newer devices and markets including implantable pump systems, drug delivery, neurological, pain management and sleep apnea—products that previously were not leading candidates for PEEK.
The trend toward minimally invasive surgery also offers great opportunities for combining unique materials in innovative ways. For example, the Kiva VCF treatment system, manufactured by Santa Clara, Calif.-based Benvenue Medical Inc., which is used to treat painful vertebral compression fractures that result from osteoporosis, uses both polyetheretherketone and nitinol, a nickel-titanium alloy that exhibits shape memory and superelasticity.
“The Kiva device combines the benefits of the shape memory of nitinol and the stress shielding and strength of PEEK-Optima,” said Devine.
Staying Ahead of the Curve
Secant Medical has integrated more biocompatible material chemistry into its development processes and applications.
“Knowing polymers at a molecular level, and understanding the complicated interaction between the implant and biological structure, are essential for both material selection and material structure,” said Amin. “Our new analytical characterization lab provides us with a deeper understanding of materials from both physical and chemical perspectives, which directly facilitates how we conduct material selection.”
Ulbrich Stainless Steels and Special Metals Inc., with plants in Wallingford, Conn., and Westminster, S.C., has expanded its product offerings for nitinol by manufacturing continuous length strip, complex-shaped cross-sections and fine wire down to less than .001-inch diameters on a daily basis.
“Other alloys we produce include niobium, tantalum, L605 (chromium-nickel-tungsten-cobalt), kovar (nickel-cobalt-iron) and many copper alloys that OEMs have been coating for medical applications,” said medical products manager Todd Hall. “While typically taboo in the medical industry, copper has become popular in a few applications when it can be coated and isolated from the body in non-implant applications.”
Last year, Fort Wayne Metals added melting capabilities to its portfolio of in-house processes. The company believes this is the first melt facility dedicated to serving the medical device industry—thereby providing more integrated supply options for select materials. The initial alloy development will focus on nitinol, followed by reactive materials such as titanium and boutique melting for niche alloys.
“Some large mills require minimum heat sizes of up to 40,000 pounds,” said Shoppell. “Our melt facility is designed to support heat quantities that are more compatible to the medical industry—in the range of 500 to 2,000 pounds. This will help reduce the amount of dollars needed to fund new alloy development.”
Fort Wayne Metals also is developing a platform of bioabsorbable metallic filaments with relatively high fatigue strength and elasticity compared to currently available materials (for example, annealed iron, magnesium and magnesium alloys) that can be used in a variety of applications.
Meeting Market Demand
For material suppliers, a critical factor in meeting market demand is knowing what customers want and planning for these needs ahead of time. OEMs expect their vendors to have deep knowledge of material behavior, production technology and FDA compliance and validation.
“Most OEMs have a keen understanding of what they need and what information and requirements the FDA will need for their products to move forward,” said Hall. “What they ask us to do is to push the limits on most of the physical properties into new ranges—characteristics such as tensile strength, surface finish, elongation, thinness and higher aspect ratios.”
Customers occasionally have material needs that require the development of a new metal alloy or plastic blend. Some companies such as Modern Plastics maintain direct relationships with firms that are conducting current research and development—ranging from resin manufacturers to extruders to compression molders—and sometimes are aware of potential solutions that their clients don’t even know exist.
To maximize customer service and minimize surprises, material suppliers must maintain constant communication with their supply chain partners in order to anticipate upcoming shortages, property changes, pricing and other factors that may affect material options.
Traditionally, the main focus by OEMs has been on the physical requirements of the final device. However, as FDA requirements become more stringent, “clients expect us to know the chemistry of the material, including chemical components, material properties throughout the development process, degradation of the material once implanted and its biocompatibility,” said Amin.
Devine noted that successful adoption of materials is based on providing a real clinical benefit that delivers economic benefits. Invibio teams devote substantial time with customers and surgeons to understand their individual requirements.
“Our aim is to ensure that we understand the performance requirements and invest the needed time and research and development resources to demonstrate these requirements,” said Devine.
For example, Invibio maintains a drug and device master file at the FDA that is available to customers, along with specific test data required for both China and Japan. The data is used by the regulatory authorities and provide verification of the biocompatibility and biostability of the company’s products. After material suppliers have identified what their customers need, they must expertly develop and manage their supply chains to meet those needs and timelines—which quickly can change.
“Customers expect us to be able optimize the materials we work with and recommend,” said Shoppell. “They also expect us to help manage the raw material supply stream and mitigate risks by having a robust supply chain, as well deliver materials at a competitive price and lead time.”
“It’s important to be certain that the material we provide to our customers is 100-percent compliant with their approved supplier lists,” added Moynihan-Downes. “We continually audit our global supply chain to confirm that our supply base is in compliance with our requirements.”
A big challenge for any material supplier is having enough material in all different grades and sizes available at all times for its domestic and global customers.
“Most contract manufacturers do not have a forecast at their disposal, which is really dependent on OEM needs,” said Niewinski. “Since 90 percent of the OEMs consist of only 10-12 companies, they control the market regarding the grades and sizes used in the selection of material.”
Forecreu America has focused sharply on inventory management by implementing new material usage programs for all its customers, including securing and holding materials for 12-24 months. The company maintains a series of standard sizes and grades for its medical customers. “Most companies have a four- to six-week window to get a prototype approved and manufactured,” said Niewinski.
“Forecreu America expedites this process by providing a wide arrange of grades and sizes that design engineers and R&D [research and development] departments can select from and have shipped the same day.”
To meet specific client needs for customized advanced materials, Secant Medical recently established an analytical lab that increases its capabilities for working with advanced materials. Customization can include yarn size in terms of cross-sectional profile, ratio of co-polymers blended together to achieve desired physical and chemical integrity and percentage of radiopaque material to achieve a desired radiopacity, depending on the location of the implant in the body.
Fort Wayne Metals also works with material suppliers to create new materials, such as its 35N LT (ASTM F562) alloy (35 percent cobalt, 35 percent nickel, 20 percent chromium and 10 percent molybdenum) which it co-patented with ATI Allvac, one of its raw material suppliers.
“In this case, the market was demanding a material with improved fatigue life,” said Shoppell. “We collaborated with Allvac to develop a melt practice that enhanced the standard version of ASTM F562, which has been utilized in medical devices for more than two decades. This alloy development work helped provide a solution for many of our customers and has also been used in multiple orthopedic and spinal applications.”
Get Involved Early
Material selection is one of the most important parts of designing an orthopedic implant; making these decisions early in the design process, with the input of an experienced material supplier, is critical for successful and cost-efficient product development. OEM design engineers won’t know it all—bringing in the material supplier to help make the best material choice will have a huge impact on lead time and price.
Lead time issues often are related to material availability. Does the material need to be melted from scratch, or is it available in stock? Most designers simply cannot wait three to six months for a material to be melted and instead decide to utilize materials that are readily available.
“Sometimes price can be a limiting factor as well, especially if a material is too exotic for a certain application,” added Shoppell. “An engineer may recognize that an alternative material can provide added benefits to the device, but if the end price point doesn’t support the selection, a substitute will be used.”
A circular relationship involving all concerned parties—OEM, distributor, machining house, resin manufacturer, stock shape extruder and injection molder—should be established at the start of the project. Otherwise, complications and contradictions may occur during product development, slowing down production and causing missed deadlines.
“For example,” noted Carbone, “an engineer can design a product using specifications he or she finds from a book on polymers that looks perfect for the desired outcome. The device may be ultimately injection molded. However, when it comes time to produce prototypical quantities for qualification and machine the first articles, complications may arise that impede the initial success of development. It may be that the resin selected is not available in a stock shape, so machining a prototype becomes difficult. Or perhaps a substitution of the grade of plastic is made, resulting in slightly different performance from the prototype to the actual molded part.”
Another reason to consider device design during the material selection process is that certain features may only be possible to create through a particular manufacturing process; if that process cannot be used for the part, additional manufacturing steps may need to be added (more cost).
“For example, cycle time to machine the part is one hour per part,” said Devine. “To injection-mold the same size part would take less than one minute. Although faster production will reduce the cost per part, production of design via the injection molding route may be limited, making it necessary to include a design modification or secondary step to achieve the same design features. Typically within medical device industry, every step requires validation—therefore the fewer the steps, the simpler the validation process.”
“Understanding how a material impacts the overall cost of a project/product is always important and there is no better time to run through the cost/price exercise than the early development phase,” concluded Shoppell. “After all, nobody wants to be surprised in regards to cost/price after the primary development phase has been completed.”
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. He can be reached at mark.crawford@charter.net.
Plastics manufacturers constantly innovate to meet evolving market demands. Design engineers are looking for plastics with specific combinations of performance characteristics, such as enhanced strength, flexibility and bioabsorbability. They also want to use materials that already are understood and approved by the U.S. Food and Drug Administration (FDA) to streamline approvals and keep costs down.
“Almost monthly it seems like a client is reacting to a market desire—sometimes existing, sometimes anticipated,” said Bing J. Carbone, president of Shelton, Conn.-based Modern Plastics Inc., a plastics distributor to the medical industry. “We stay on top of these developments so we can provide customers with up-to-the-minute advancements on new material options and services that we can offer.”
A tremendous amount of research is being conducted on creating new biocompatible compounds or improving existing materials by developing new blends. Although these new materials may have fantastic properties, many companies often are cautious about embracing them because it may mean gathering more data or conducting extensive trials. Instead, they combine materials that already have a proven history of effectiveness and FDA compliance to create blended materials that best meet the product’s performance specifications.
The FDA also has raised the regulatory bar in recent years, especially regarding component biocompatibility and supply chain management. OEMs increasingly expect their material providers to know the latest regulations and maintain top-level quality management systems that ideally include ISO 9001 and ISO 13485 certification. Material providers must constantly improve their processes, quality assurance methods and standard operating procedures to stay ahead of the curve and remain competitive. Because material lead times often are extended, expert and sometimes creative inventory management is required to meet short production deadlines.
What OEMs Want
OEMs want supply chain partners that can provide risk reduction (safety, reliability, track record of clinical success and security of supply), reliable and high-performance materials and the ability to access regulatory data from global markets. OEMs also hope their material providers are experts at supply chain efficiency, inventory management and serving emerging markets.
“Trends in material selection continue to be toward risk management and assessment with respect to the material being evaluated, as well as its biocompatibility,” said Mary Moynihan-Downes, director of the medical market for Rockaway, N.J.-based Titanium Industries Inc., a manufacturing distributor of metal. “Once those factors are satisfied, economics and availability become the key elements to be considered.”
There also are demands from the production and manufacturing side. “Almost every OEM engineer I talk with says there is pressure from management for smaller implants and instruments with tighter tolerances,” said Grant Niewinski, U.S. sales and marketing manager for Forecreu America Inc., a Chicago, Ill.-based producer of stainless steel and titanium cannulated bars for the orthopedics market. All these expectations challenge the design process, especially material selection.
As a result, design engineers are searching for more custom blends with specific features such as strength, hardness, flexibility, heat resistance, chemical resistance, etc.
“For example,” said Carbone, “barium sulfates, carbons, glass, anti-microbial and other fillers are being added to plastics to enhance characteristics such as strength, heat resistance, radiopacity and infection resistance. Although some of these fillers have been used for years, they are increasingly being combined in different ways to produce some novel products that should be introduced to the general market very soon.”
OEMs also want to launch their new product lines quickly—there is an ever-present push for speed, which also can impact the final material decision.
“Utilizing a material that is already associated with an ASTM [formerly the American Society for Testing and Materials] or ISO standard often influences a company's willingness to move forward,” commented Scott Shoppell, marketing manager for Fort Wayne, Ind.-based Fort Wayne Metals, a provider of research, development and production of fine grade medical wire, rod, cable, metal-on-metal composites and custom assemblies. “Some companies will not commit time and resources to developing materials that don’t already have an implant-grade specification. Companies with more knowledgeable and experienced personnel in their technical and materials groups tend to be less intimidated by the possibility of blazing trails with innovative materials.”
Material Trends
Popular materials in the orthopedic industry include high-grade polyester, polyethylene, metals and alloys such as stainless steel, titanium and cobalt chromium. For many years, the medical device industry’s mind-set has been to replace impaired tissue—for example, replacing cartilage or bone with synthetic materials.
More recently, researchers and medical companies have been seeking alternative materials that also can induce regeneration and restoration of the impaired tissue. Bioresorbable materials, including polyglycolic acid, polydioxanone, polylactic acid and polycaprolactone, are more compatible with surrounding tissues and the body’s own natural healing processes.
“Bioresorbable materials can be used as a temporary scaffold until the tissue’s intended function is restored,” indicated Swati Amin, advanced biomaterials engineer for Secant Medical Inc., a Perkasie, Pa.-based designer and manufacturer of biomedical textiles and material structures for medical devices. “For example, these materials can be engineered and processed to achieve a desired degradation time that matches the time it takes for the native tissue to heal itself.”
One of the biggest challenges in manufacturing these types of materials, noted Amin, is the stability of the polymer. “Oxygen, heat, or light can alter these sensitive materials and propagate a cascade of oxidation, causing unwanted degradation prior to implantation,” she added. “Knowing this during polymer processing is essential.”
The ongoing problems with metal-on-metal hip implants are driving more interest in metal-free alternatives for these implant devices that have just as much strength and durability as metal—especially PEEK.
“Given that PEEK polymers are stronger than many metals on a per-mass basis, these materials are increasingly selected as an alternative to metal,” stated John Devine, marketing and technology director for Invibio Biomaterial Solutions, a provider of PEEK-based biomaterials based in Thornton Cleveleys, in the United Kingdom. “The manufacturing efficiency gains for metals are also reaching a plateau; in order to reduce costs, device manufacturers are looking to alternative processes such as injection molding.”
An advanced form of PEEK is Invibio’s PEEK-Optima Ultra Reinforced, which offers semi-rigid fixation with improved fatigue performance and radiolucency. “It combines the high-performance material properties of PEEK-Optima with additional strength imparted by carbon fiber, providing an alternative to metal biomaterials traditionally used in internal fracture fixation procedures,” said Devine.
Carbone indicated that more OEMs are considering PEEK for a broader range of applications, including newer spine, orthopedic, dental, small-bone and cranial applications, as well as newer devices and markets including implantable pump systems, drug delivery, neurological, pain management and sleep apnea—products that previously were not leading candidates for PEEK.
The trend toward minimally invasive surgery also offers great opportunities for combining unique materials in innovative ways. For example, the Kiva VCF treatment system, manufactured by Santa Clara, Calif.-based Benvenue Medical Inc., which is used to treat painful vertebral compression fractures that result from osteoporosis, uses both polyetheretherketone and nitinol, a nickel-titanium alloy that exhibits shape memory and superelasticity.
“The Kiva device combines the benefits of the shape memory of nitinol and the stress shielding and strength of PEEK-Optima,” said Devine.
Staying Ahead of the Curve
Secant Medical has integrated more biocompatible material chemistry into its development processes and applications.
“Knowing polymers at a molecular level, and understanding the complicated interaction between the implant and biological structure, are essential for both material selection and material structure,” said Amin. “Our new analytical characterization lab provides us with a deeper understanding of materials from both physical and chemical perspectives, which directly facilitates how we conduct material selection.”
Ulbrich Stainless Steels and Special Metals Inc., with plants in Wallingford, Conn., and Westminster, S.C., has expanded its product offerings for nitinol by manufacturing continuous length strip, complex-shaped cross-sections and fine wire down to less than .001-inch diameters on a daily basis.
“Other alloys we produce include niobium, tantalum, L605 (chromium-nickel-tungsten-cobalt), kovar (nickel-cobalt-iron) and many copper alloys that OEMs have been coating for medical applications,” said medical products manager Todd Hall. “While typically taboo in the medical industry, copper has become popular in a few applications when it can be coated and isolated from the body in non-implant applications.”
Last year, Fort Wayne Metals added melting capabilities to its portfolio of in-house processes. The company believes this is the first melt facility dedicated to serving the medical device industry—thereby providing more integrated supply options for select materials. The initial alloy development will focus on nitinol, followed by reactive materials such as titanium and boutique melting for niche alloys.
“Some large mills require minimum heat sizes of up to 40,000 pounds,” said Shoppell. “Our melt facility is designed to support heat quantities that are more compatible to the medical industry—in the range of 500 to 2,000 pounds. This will help reduce the amount of dollars needed to fund new alloy development.”
Fort Wayne Metals also is developing a platform of bioabsorbable metallic filaments with relatively high fatigue strength and elasticity compared to currently available materials (for example, annealed iron, magnesium and magnesium alloys) that can be used in a variety of applications.
Meeting Market Demand
For material suppliers, a critical factor in meeting market demand is knowing what customers want and planning for these needs ahead of time. OEMs expect their vendors to have deep knowledge of material behavior, production technology and FDA compliance and validation.
“Most OEMs have a keen understanding of what they need and what information and requirements the FDA will need for their products to move forward,” said Hall. “What they ask us to do is to push the limits on most of the physical properties into new ranges—characteristics such as tensile strength, surface finish, elongation, thinness and higher aspect ratios.”
Customers occasionally have material needs that require the development of a new metal alloy or plastic blend. Some companies such as Modern Plastics maintain direct relationships with firms that are conducting current research and development—ranging from resin manufacturers to extruders to compression molders—and sometimes are aware of potential solutions that their clients don’t even know exist.
To maximize customer service and minimize surprises, material suppliers must maintain constant communication with their supply chain partners in order to anticipate upcoming shortages, property changes, pricing and other factors that may affect material options.
Traditionally, the main focus by OEMs has been on the physical requirements of the final device. However, as FDA requirements become more stringent, “clients expect us to know the chemistry of the material, including chemical components, material properties throughout the development process, degradation of the material once implanted and its biocompatibility,” said Amin.
Devine noted that successful adoption of materials is based on providing a real clinical benefit that delivers economic benefits. Invibio teams devote substantial time with customers and surgeons to understand their individual requirements.
“Our aim is to ensure that we understand the performance requirements and invest the needed time and research and development resources to demonstrate these requirements,” said Devine.
For example, Invibio maintains a drug and device master file at the FDA that is available to customers, along with specific test data required for both China and Japan. The data is used by the regulatory authorities and provide verification of the biocompatibility and biostability of the company’s products. After material suppliers have identified what their customers need, they must expertly develop and manage their supply chains to meet those needs and timelines—which quickly can change.
“Customers expect us to be able optimize the materials we work with and recommend,” said Shoppell. “They also expect us to help manage the raw material supply stream and mitigate risks by having a robust supply chain, as well deliver materials at a competitive price and lead time.”
“It’s important to be certain that the material we provide to our customers is 100-percent compliant with their approved supplier lists,” added Moynihan-Downes. “We continually audit our global supply chain to confirm that our supply base is in compliance with our requirements.”
A big challenge for any material supplier is having enough material in all different grades and sizes available at all times for its domestic and global customers.
“Most contract manufacturers do not have a forecast at their disposal, which is really dependent on OEM needs,” said Niewinski. “Since 90 percent of the OEMs consist of only 10-12 companies, they control the market regarding the grades and sizes used in the selection of material.”
|
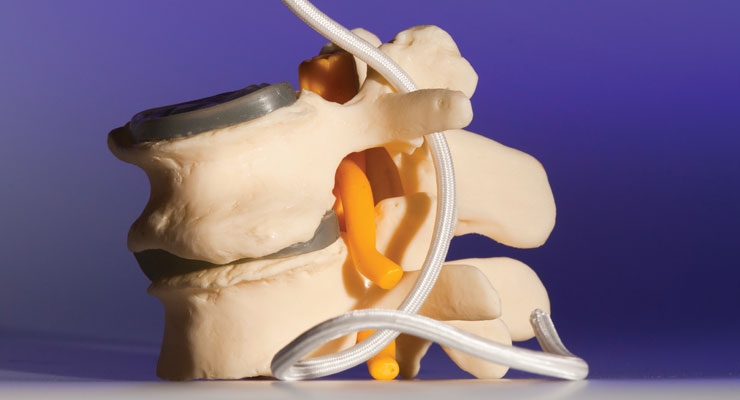
The ongoing problems with metal-on-metal hip implants are driving more interest in metal-free alternatives for orthopedic devices that have just as much strength and durability as metal—especially polyetheretherketone (PEEK, samples pictured above). Photo courtesy of Invibio Biomaterial Solutions. |
“Forecreu America expedites this process by providing a wide arrange of grades and sizes that design engineers and R&D [research and development] departments can select from and have shipped the same day.”
To meet specific client needs for customized advanced materials, Secant Medical recently established an analytical lab that increases its capabilities for working with advanced materials. Customization can include yarn size in terms of cross-sectional profile, ratio of co-polymers blended together to achieve desired physical and chemical integrity and percentage of radiopaque material to achieve a desired radiopacity, depending on the location of the implant in the body.
Fort Wayne Metals also works with material suppliers to create new materials, such as its 35N LT (ASTM F562) alloy (35 percent cobalt, 35 percent nickel, 20 percent chromium and 10 percent molybdenum) which it co-patented with ATI Allvac, one of its raw material suppliers.
“In this case, the market was demanding a material with improved fatigue life,” said Shoppell. “We collaborated with Allvac to develop a melt practice that enhanced the standard version of ASTM F562, which has been utilized in medical devices for more than two decades. This alloy development work helped provide a solution for many of our customers and has also been used in multiple orthopedic and spinal applications.”
Get Involved Early
Material selection is one of the most important parts of designing an orthopedic implant; making these decisions early in the design process, with the input of an experienced material supplier, is critical for successful and cost-efficient product development. OEM design engineers won’t know it all—bringing in the material supplier to help make the best material choice will have a huge impact on lead time and price.
Lead time issues often are related to material availability. Does the material need to be melted from scratch, or is it available in stock? Most designers simply cannot wait three to six months for a material to be melted and instead decide to utilize materials that are readily available.
“Sometimes price can be a limiting factor as well, especially if a material is too exotic for a certain application,” added Shoppell. “An engineer may recognize that an alternative material can provide added benefits to the device, but if the end price point doesn’t support the selection, a substitute will be used.”
A circular relationship involving all concerned parties—OEM, distributor, machining house, resin manufacturer, stock shape extruder and injection molder—should be established at the start of the project. Otherwise, complications and contradictions may occur during product development, slowing down production and causing missed deadlines.
“For example,” noted Carbone, “an engineer can design a product using specifications he or she finds from a book on polymers that looks perfect for the desired outcome. The device may be ultimately injection molded. However, when it comes time to produce prototypical quantities for qualification and machine the first articles, complications may arise that impede the initial success of development. It may be that the resin selected is not available in a stock shape, so machining a prototype becomes difficult. Or perhaps a substitution of the grade of plastic is made, resulting in slightly different performance from the prototype to the actual molded part.”
Another reason to consider device design during the material selection process is that certain features may only be possible to create through a particular manufacturing process; if that process cannot be used for the part, additional manufacturing steps may need to be added (more cost).
“For example, cycle time to machine the part is one hour per part,” said Devine. “To injection-mold the same size part would take less than one minute. Although faster production will reduce the cost per part, production of design via the injection molding route may be limited, making it necessary to include a design modification or secondary step to achieve the same design features. Typically within medical device industry, every step requires validation—therefore the fewer the steps, the simpler the validation process.”
“Understanding how a material impacts the overall cost of a project/product is always important and there is no better time to run through the cost/price exercise than the early development phase,” concluded Shoppell. “After all, nobody wants to be surprised in regards to cost/price after the primary development phase has been completed.”
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. He can be reached at mark.crawford@charter.net.