Managing Editor, Managing Editor03.22.13
The operating room wasn’t unusually warm that day, but Robert M. Campbell Jr., M.D., nevertheless began to sweat as he navigated around the tiny upper torso of a baby born without a chest wall. The 6-month-old boy needed a ventilator to breathe and was expected to die unless Mother Nature’s oversight somehow could be fixed.
As a professor, Campbell was fully versed in the birth defect that left the dark-haired tot without seven ribs or chest muscles on his right side. He knew the condition—rib agenesis—was relatively rare; he knew it could be accompanied by spina bifida, congenital vertebral fusion, meningocoele or other genetic anomalies; and he understood the syndrome eventually would lead to complications like shifting organs, abnormal lung growth and scoliosis (curvature of the spine).
Campbell also knew he was the child’s only hope for survival. His recruiter suspected it as well: Having heard of Campbell’s college engineering background, pediatric general surgeon Melvin Smith, M.D., flagged down Campbell in a hospital corridor and impulsively requested his help. Smith, then affiliated with CHRISTUS Santa Rosa Children’s Hospital in San Antonio, Texas, believed the boy’s condition was treatable due to his strong heart and potentially viable right lung that “just needed a place to work.”
Creating such an infrastructure, however, was particularly challenging. Besides lacking expertise in medical device design, manufacturing and production, the doctors had little or no knowledge of federal regulatory requirements or the investment channels available to startup companies and individual innovators. Perhaps the biggest obstacle, though, was the dearth of any existing device that could possibly have saved the boy’s life. The pair briefly considered the Toronto chest wall splint, but Campbell knew the device wouldn’t give the child enough room to aerate his lungs.
Thus, Campbell and Smith were forced to improvise a solution. During a delicate but radical nerve-racking procedure on Oct. 3, 1987, the pair built a picket fence-like chest wall of heavy gauge Steinmann fracture pins (a device invented by Swiss surgeon Fritz Steinmann in 1908 and used primarily in adults), inserting three 4-inch-long stainless steel rods vertically and bending those rods around the child’s five remaining ribs near his neck and waist. The rods, Campbell would later recall, were a nightmare to put in: “One slip and I would have torn the axillary artery or damaged the spinal cord…my shoes were filling up with sweat.”
Four days after inserting the metal fracture pins, the baby took his first ventilator-free breaths. Campbell and Smith initially were thrilled with the success of the revolutionary surgery, but they also knew their makeshift device was only a temporary fix. The rods they implanted in the infant were too rigid to accommodate future growth; without intervention, the boy’s lung would have no room to grow and his spine would begin to curve again. To truly help this child and others like him, the pair would have to build a better mousetrap—ideally, something that would be both easy to implant and expand through minor surgery.
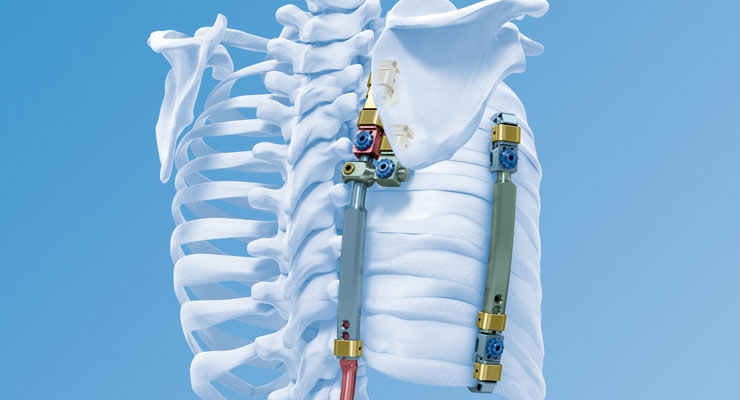
Campbell came up with the consummate design in 10 months, spending about 2,000 hours revising, refining and perfecting the blueprint for the Vertical Expandable Prosthetic Titanium Rib (VEPTR), a device akin to a curtain rod with two moving parts that works like a fracture pin but is easily implantable and expandable. The 12-inch rib is curved (as nature intended), and has three holes that enable surgeons to expand the device in outpatient surgery every six months. In many cases, the progressive lengthening of the VEPTR device can delay the necessity of performing a spinal fusion by several years or more.
The second-generation VEPTR II has the same basic design but features additional components such as rib hooks, transverse bars, an S rod and a proximal extension that provide orthopedic surgeons with more options to address juvenile chest wall and/or spinal defects. Comprising titanium (Campbell and his first manufacturing partner chose the material for its biocompatibility, light weight and non-interference with MRI scans), both versions can be implanted in children as young as six months through skeletal maturity, which typically occurs at age 14 for girls and 16 for boys.
The first titanium rib went to its fracture-pinned muse on April 19, 1989 (technically, the boy received the second VEPTR—Campbell accidentally broke the first one by repeatedly bending its loops with vice-grips). The success of the boy’s second surgery marked the start of a new chapter in Campbell’s quest to make the VEPTR globally available on a mass-market scale. He and Smith formed the Titanium Rib Project and developed five types of VEPTR procedures for children suffering from thoracic insufficiency syndrome (TIS), a rare condition in which the chest cannot support normal breathing or lung growth.
In the summer of 1990, the U.S. Food and Drug Administration (FDA) authorized a sole site feasibility study of the VEPTR device with Techmedica Co. (the original manufacturer) as the sponsor but 14 years passed before the agency gave its official blessing to Campbell’s invention through a humanitarian device exemption (HDE). Since its 1989 debut, the VEPTR has been used to treat hundreds of TIS-afflicted children in more than 25 countries worldwide. The device is now made by DePuy/Synthes.
“Back in 1987, there wasn’t much available to treat pediatric chest conditions,” said Campbell, a pediatric orthopedic surgeon and director of the Center for Thoracic Insufficiency Syndrome at the Children’s Hospital of Philadelphia. “There was no suitable fix at the time that would have allowed this child to breathe on his own. If the same situation came up today, there would still be only the VEPTR available to us because it’s the only FDA-approved device for TIS. There’s just not a lot of device choices for any particular condition in pediatrics."
Barriers to Innovation
Children have rarely been the beneficiary of medtech innovation. Comprising 30 percent of the general population, a mere 2 percent of deaths and 11 percent of all hospital discharges (excluding newborns), juveniles are, by and large, too small and too healthy a lot to attract much interest from investors or medical device manufacturers. Case in point: Roughly 294,000 Americans under the age of 18 are living with juvenile arthritis or other rheumatic condition, while an estimated 50 million adults (22 percent of the U.S. population) have been diagnosed with the disease, according to National Health Interview Survey data from 2007-2009. Such titanic market chasms (more than 170-fold, between young and older arthritis patients) indubitably influence new product development decision-making.
But sick and injured children aren’t that appealing to companies, either—their bodies are a microcosm of biodiversity, each containing its own growth rate, metabolism, heart rate, activity level, chemical composition, protein levels and blood pressure. The medley of physiological data varies according to age, making general treatments difficult, if not impossible, to develop. For instance, younger pediatric patients—neonates, infants and children up to 10 years of age—have higher heart rates than full-grown adults and those aged 11-17. Thus, some devices developed for and used by adults—such as cardiac valves—cannot be used in young children without major redesigns. Similarly, off-the-shelf hip and knee joints generally are too large for kids; even the smallest versions (designed mostly for women and Asian populations) would be inappropriate for young athletes with damaged knee cartilage or those suffering from juvenile arthritis.
Comparable differences exist among the various pediatric patient groups as well. Infant-specific devices may not be effective in toddlers and most likely would be useless in teenagers, even if all three groups share conditions or diseases. Such disparity requires medtech firms to develop and test a different product for each patient subpopulation, regardless of their mutual affliction.
The unique characteristics of children and the relatively diminutive size of the pediatric market are two of the biggest barriers to pediatric device innovation. “It takes about $30 million to bring a [medical] device to market,” explained Michael G. Vitale, M.D., M.P.H., a Columbia University professor and pediatric spinal surgeon at Morgan Stanley Children’s Hospital in New York, N.Y., who also is a board member of the Pediatric Orthopaedic Society of North America, a Rosemont, Ill.-based organization founded in 1971 to advance orthopedic care for children.
“In a market where 3,000 or 4,000 cases can occur each year, it would take a company far longer to recover its investment compared with the adult market,” he continued. “The math just doesn’t work for smaller [patient] populations. If you need $30 million to get your device to market and the size of the market is such that you’ll only make $1 million a year, no business other than a humanitarian group or philanthropic organization is going to be willing to spend $30 million to get back $1 million. There’s not enough of a financial incentive to support the development of pediatric devices so alternate mechanisms and strategies are necessary.”
Even when the financial incentive (and desire) exists, though, companies still encounter roadblocks en route to product commercialization. Many firms have trouble recruiting pediatric participants in clinical trials due to parental consent and the difficulty in performing experimental procedures on children. In addition, the various subpopulations can limit the number of young recruits and add to the overall trial cost. As Campbell noted: “For pediatric devices with a limited market, it’s hard to convince investors to spend millions of dollars on a clinical trial. With adult products, it’s different—those devices are used by a lot of people. The market size justifies the cost but with kids it’s a lot tougher to rationalize the expense of a clinical trial. It has become a serious problem.”
So has the regulatory process. The government’s product approval channels can be challenging for any company but startups and doctors (who often have the most innovative ideas) particularly are at a disadvantage because they lack the experience of larger firms. Campbell had no previous knowledge of the FDA’s requirements for device development before he sought approval for the VEPTR in 1990. He describes his 14-year odyssey with the FDA as a “trial and error experience” and contends the agency lacked a clear, transparent regulatory pathway for approval. And while he received grant funding for his device from the National Organization of Rare Disorders (NORD) and the FDA’s Orphan Products Division, he believes additional resources and mentors would have made the VEPTR approval process faster, easier and significantly more pleasant.
Unpredictability and inconsistency from the FDA can make it difficult for companies to properly budget for new product development, too. Some manufacturers also have been discouraged by a lack of published data on pediatric devices but regulators are attempting to rectify the problem with a new regulation and related draft guidance that clarifies the kind of information companies must submit with pediatric device applications.
Under the proposal, published in a Feb. 19 Federal Register notice (78 Fed. Reg. 11,654), companies must submit “readily available” information about pediatric patients who might benefit from a device. Acceptable data includes publicly accessible material from medical journals, large surveys, patient registries, federal records, scientific research resources (MEDLINE and PubMed), online medical libraries (Cochrane and the Public Library of Science) and clinicaltrials.gov. The proposal bans information garnered from non-public sources such as internal marketing surveys, sales records, R&D reports, unpublished presentations and private consultations with clinicians.
In drafting the new rule, the FDA is attempting to better identify and track medical devices that could be used to treat children. The agency is requiring medtech firms to detail the ways in which a product will be used and estimate the number of U.S. pediatric patients who suffer from the disease the device treats or diagnoses. Companies also must provide a description of any affected pediatric subpopulations—either neonates (0-28 days), infants (younger than 2), children (aged 2-12) or adolescents (under 21).
The pediatric data rule applies to premarket approval applications, HDE requests and product development protocols, an alternative endorsement method that reverses the last two steps of FDA review and panel review. Manufacturers that fail to include pediatric information with their applications risk delaying the approval process. The FDA intends to use the data in these applications to report annually to Congress on the number of approved devices that could be used for a pediatric population as well as the number of approved devices that could be labeled for use in pediatric patients.
Gray’s Anatomy vs. Hamann-Todd
Campbell often jokes about the obsolescence of his operating room. “If a surgeon from the 1950s were to come into my operating room when I’m working on a patient, he would recognize many of the pediatric instruments and devices because there has been little progress since then,” he told Orthopedic Design & Technology. “I can’t tell you how many times I’ve been frustrated over the lack of modern medical devices for pediatric care. But you can’t hang it up when you don’t have what you need. You just make do with what you have.”
Orthopedic surgeons, however, haven’t really had much to work with in the last half-century or so. Most pediatric specialists have resorted to jury-rigging adult devices for kids or using products in an unapproved manner to help their patients. Pins normally reserved for the fixation of small bone fragments, for example, have been used as intramedullary rods in extremely small children. Iowa City, Iowa-based surgeon Stuart L. Weinstein, M.D., admits to using spinal rods in the legs of children because “they are the only thing that really fits.” He also has used adult arm implants in a child’s leg, specifically a humeral nail in the tibia. “Overall, we have to improvise quite frequently by using available products,” Weinstein disclosed to Orthopedics Today in 2010. “I have to use adult devices off-label because there is nothing else available.”
Campbell hasn’t always played by the rules, either. “You do what you have to do. Have I sawed devices in half? Sure. Have I used a device for another purpose? Sure. Does it work out 99 percent of the time? Yes, but it’s not ideal.”
Perfection, however, can be a difficult standard to achieve, especially in children. Their bodies are considerably smaller than adults, but their musculoskeletal structures grow and change as they mature. That growth, though, is not always uniform, so their devices must be able to accommodate the disjointed nature of their development (a prosthetic socket, for instance, might be outgrown but the pylon height may still be acceptable).
Accommodating for such variations in growth requires a comprehensive understanding of the juvenile anatomy, experts claim. The most successful pediatric device developers are connoisseurs of the juvenile physique, fully aware of the sensitivities of children’s growth plates (zones of cartilage at each end of the long bones), the hypermobility of their joints, the strength of their periosteum (the membrane lining the outer surface of all bones except large bone joints), the degree to which their femurs bow, and the number of additional bones they support upon their frames (94) compared with adults.
“It’s not necessarily the size that’s an issue, it’s knowing the anatomy of a child,” noted Nick Deeter, founder and board chairman of OrthoPediatrics Corp., a leading developer of juvenile orthopedic products based in Warsaw, Ind. “Kids are different. You have to account for growth plates which are not a factor in the adult market. When placing plates and screws on pediatric bones, it is critical to ensure the growth plates are avoided to prevent the potential for growth disturbances. Also, children’s bones have more bowing, like in the femur, therefore you cannot correct a deformity or fracture using a standard straight nail as in the adult market or it could damage the bone. There are a lot of variations in children that you don’t have in adults.”
OrthoPediatrics mastered all of those variations by securing exclusive rights to two of the world’s largest collections of pediatric skeletal data—the Hamann-Todd Human Osteological Collection and the Bolton-Brush Growth Study at Case Western Reserve University. The Hamann-Todd collection, now curated at the Cleveland Museum of Natural History, contains the records and bones of more than 3,000 people who died in the early 20th century. In most cases, the records show the identity of the donors along with their medical histories and in some cases, their photographs. Deeter said his company scanned all the pediatric skeletons in that collection to learn more about pediatric bone growth and unlock the mysteries of growth plates.
The Bolton-Brush Growth Study is globally considered to be the most extensive source of longitudinal human growth data. The study combines information from two separate investigations—one that tracked participant’s mental and physical growth and another that charted growth and development of the face and teeth. More than 4,000 kids from Cleveland underwent head-to-toe X-rays of their bodies to contribute to the database on mental/physical growth. The youngest children were three months old when the study began; all participants were X-rayed every three months during the first two years of their life and then less frequently after that. The Bolton-Brush studies have become one of the longest-running examinations of normal human health and development.
Data from both osteological collections have helped OrthoPediatrics develop customized products for children. Its intramedullary femoral nail, for example, features a smaller diameter and three proximal locking hole options—oblique, transverse and recon. It also has an anterior bow to mimic the normal anatomy of kids ages 8 to 16, according to a product brochure. “When we developed the intramedullary nail, we made it so it goes in the correct way every time. It has so many bends in it, [the nail] looks like it was run over by a truck,” Deeter quipped.
OrthoPediatrics also modeled its PediFlex nail and PediPlates based on information it extracted from the bones of long-dead children. Its flexible nail is used to treat long bone fractures and is manufactured in both titanium (1.5 mm-4.5 mm) and stainless steel (1.5 mm-4.0 mm) in increments of 0.5 millimeters. The PediPlates are used to correct angular deformities.
“OrthoPediatrics is doing a great job designing plates and screws for the pediatric population,” said Andrew Bowe, M.D., a pediatric orthopedic surgeon affiliated with Robert Wood Johnson University Hospital in New Brunswick, N.J. “In general, what surgeons look for in a pediatric device are ease of use, and ease of removal. The hardware is usually removed in children. If they sustain another injury, a plate or some other kind of implant can significantly complicate their treatment. There are some studies that show rigid hardware around the joint can alter some of the mechanics of that joint over the course of many years. It’s always better to get hardware out of kids whenever possible.”
During its tenancy, however, an implant must accommodate a child’s natural growth. Many bone plates and rods are lengthened or modified through revision surgery, but some promising new technologies eliminate the need for multiple procedures, lowering the risk of complications and potential infections. Resorbable implants are becoming popular with specialists in pediatric craniofacial and orthopedic deformities. The implants are rigid enough to support the repair or reconstruction of a deformity but eventually disappear without interfering with a child’s growth.
Likewise, researchers are tinkering with human tissue to regrow skin, cartilage, bone and heart valves. Several years ago, engineers at the Massachusetts Institute of Technology in Cambridge built a new tissue scaffold that stimulated bone and cartilage growth when it was transplanted into damaged knees and other joints. The scaffold had two layers—one mirrored bone and the other emulated cartilage. When implanted into a joint, the scaffold stimulated mesenchymal stem cells in the bone marrow to produce both new bone and cartilage. Life sciences experts are hopeful that such breakthroughs could eventually eliminate or at the very least, limit the immunocompatibility and biocompatibility problems associated with the implant materials currently available.
Nurturing Invention
Campbell got lucky when he developed the VEPTR. He received support from manufacturers (Techmedica and Synthes Spine Company), non-profit groups (NORD) and to some extent, the FDA. Such support, however, traditionally has been rare for pediatric device inventors. “We were extremely lucky that we found companies that could be responsive to the needs of children in spite of the many obstacles,” Campbell told a U.S. Senate Committee on Health, Education, Labor and Pensions in 2007. “Small companies can be responsive to small pediatric projects, but do not have large budgets for device development or the regulatory resources to secure FDA approval. Large publicly owned companies have those resources, but can’t justify non-profit pediatric device projects to their stockholders.”
The government tackled that funding conundrum six years ago by passing the FDA Amendments Act, a bill that removed profit restrictions on pediatric devices approved under the agency’s HDE program and authorized $6 million annually through 2012 for projects that linked inventors of juvenile products with potential manufacturers. Congress renewed the legislation last summer through the Medical Device User Fee Act; children’s advocates lauded lawmakers for preserving the HDE pediatric profit incentive but lamented the bill’s failure to address inconsistent insurance coverage often associated with HDE-approved kids’ devices.
Besides extending the HDE pediatric profit incentive, the reauthorized bill also requires the FDA to hold a public meeting and issue a report on efforts to accelerate the development of products for rare juvenile diseases. In addition, the legislation renewed funding for the Pediatric Device Consortia Grant Program, an initiative that subsidizes non-profit consortia that develop medical devices for youngsters.
The five FDA-funded consortia have helped advance the development of roughly 135 proposed pediatric medical devices. The consortia currently are managing 80 projects, including a robo-implant to treat severe pediatric spinal deformities and a device that uses two rare earth magnets to slowly reconfigure the chest in children with hollowed torsos.
Indeed, recent financial incentives have helped encourage innovation (the number of juvenile-specific medical products has jumped five-fold since 2008), but it may take a paradigm shift in both attitude and business models for children to finally get the attention they deserve from the device industry.
OrthoPediatrics’ Deeter suggests companies consider the long-term economic benefit pediatric devices can contribute to society.
“Developing a medical device for the pediatric population is an investment, there’s no denying that,” Deeter said. “But these devices are economically viable. There’s a product that can fix bowed legs on a child in about six weeks…think about that. If a child goes through life with really bowed legs, he can get rheumatoid arthritis in his knee or hip and you’ll eventually have to replace those knees or hips.
But by straightening out those legs when they’re young, you’ve spent $1,000 on a procedure and avoided spending $100,000 in the future when that kid eventually needs new joints. It’s a lot easier to fix kids when they’re young…and when you do that, you’ll find they kind of fix themselves, too.”
As a professor, Campbell was fully versed in the birth defect that left the dark-haired tot without seven ribs or chest muscles on his right side. He knew the condition—rib agenesis—was relatively rare; he knew it could be accompanied by spina bifida, congenital vertebral fusion, meningocoele or other genetic anomalies; and he understood the syndrome eventually would lead to complications like shifting organs, abnormal lung growth and scoliosis (curvature of the spine).
Campbell also knew he was the child’s only hope for survival. His recruiter suspected it as well: Having heard of Campbell’s college engineering background, pediatric general surgeon Melvin Smith, M.D., flagged down Campbell in a hospital corridor and impulsively requested his help. Smith, then affiliated with CHRISTUS Santa Rosa Children’s Hospital in San Antonio, Texas, believed the boy’s condition was treatable due to his strong heart and potentially viable right lung that “just needed a place to work.”
Creating such an infrastructure, however, was particularly challenging. Besides lacking expertise in medical device design, manufacturing and production, the doctors had little or no knowledge of federal regulatory requirements or the investment channels available to startup companies and individual innovators. Perhaps the biggest obstacle, though, was the dearth of any existing device that could possibly have saved the boy’s life. The pair briefly considered the Toronto chest wall splint, but Campbell knew the device wouldn’t give the child enough room to aerate his lungs.
Thus, Campbell and Smith were forced to improvise a solution. During a delicate but radical nerve-racking procedure on Oct. 3, 1987, the pair built a picket fence-like chest wall of heavy gauge Steinmann fracture pins (a device invented by Swiss surgeon Fritz Steinmann in 1908 and used primarily in adults), inserting three 4-inch-long stainless steel rods vertically and bending those rods around the child’s five remaining ribs near his neck and waist. The rods, Campbell would later recall, were a nightmare to put in: “One slip and I would have torn the axillary artery or damaged the spinal cord…my shoes were filling up with sweat.”
Four days after inserting the metal fracture pins, the baby took his first ventilator-free breaths. Campbell and Smith initially were thrilled with the success of the revolutionary surgery, but they also knew their makeshift device was only a temporary fix. The rods they implanted in the infant were too rigid to accommodate future growth; without intervention, the boy’s lung would have no room to grow and his spine would begin to curve again. To truly help this child and others like him, the pair would have to build a better mousetrap—ideally, something that would be both easy to implant and expand through minor surgery.
Campbell came up with the consummate design in 10 months, spending about 2,000 hours revising, refining and perfecting the blueprint for the Vertical Expandable Prosthetic Titanium Rib (VEPTR), a device akin to a curtain rod with two moving parts that works like a fracture pin but is easily implantable and expandable. The 12-inch rib is curved (as nature intended), and has three holes that enable surgeons to expand the device in outpatient surgery every six months. In many cases, the progressive lengthening of the VEPTR device can delay the necessity of performing a spinal fusion by several years or more.
The second-generation VEPTR II has the same basic design but features additional components such as rib hooks, transverse bars, an S rod and a proximal extension that provide orthopedic surgeons with more options to address juvenile chest wall and/or spinal defects. Comprising titanium (Campbell and his first manufacturing partner chose the material for its biocompatibility, light weight and non-interference with MRI scans), both versions can be implanted in children as young as six months through skeletal maturity, which typically occurs at age 14 for girls and 16 for boys.
The first titanium rib went to its fracture-pinned muse on April 19, 1989 (technically, the boy received the second VEPTR—Campbell accidentally broke the first one by repeatedly bending its loops with vice-grips). The success of the boy’s second surgery marked the start of a new chapter in Campbell’s quest to make the VEPTR globally available on a mass-market scale. He and Smith formed the Titanium Rib Project and developed five types of VEPTR procedures for children suffering from thoracic insufficiency syndrome (TIS), a rare condition in which the chest cannot support normal breathing or lung growth.
In the summer of 1990, the U.S. Food and Drug Administration (FDA) authorized a sole site feasibility study of the VEPTR device with Techmedica Co. (the original manufacturer) as the sponsor but 14 years passed before the agency gave its official blessing to Campbell’s invention through a humanitarian device exemption (HDE). Since its 1989 debut, the VEPTR has been used to treat hundreds of TIS-afflicted children in more than 25 countries worldwide. The device is now made by DePuy/Synthes.
“Back in 1987, there wasn’t much available to treat pediatric chest conditions,” said Campbell, a pediatric orthopedic surgeon and director of the Center for Thoracic Insufficiency Syndrome at the Children’s Hospital of Philadelphia. “There was no suitable fix at the time that would have allowed this child to breathe on his own. If the same situation came up today, there would still be only the VEPTR available to us because it’s the only FDA-approved device for TIS. There’s just not a lot of device choices for any particular condition in pediatrics."
Barriers to Innovation
Children have rarely been the beneficiary of medtech innovation. Comprising 30 percent of the general population, a mere 2 percent of deaths and 11 percent of all hospital discharges (excluding newborns), juveniles are, by and large, too small and too healthy a lot to attract much interest from investors or medical device manufacturers. Case in point: Roughly 294,000 Americans under the age of 18 are living with juvenile arthritis or other rheumatic condition, while an estimated 50 million adults (22 percent of the U.S. population) have been diagnosed with the disease, according to National Health Interview Survey data from 2007-2009. Such titanic market chasms (more than 170-fold, between young and older arthritis patients) indubitably influence new product development decision-making.
But sick and injured children aren’t that appealing to companies, either—their bodies are a microcosm of biodiversity, each containing its own growth rate, metabolism, heart rate, activity level, chemical composition, protein levels and blood pressure. The medley of physiological data varies according to age, making general treatments difficult, if not impossible, to develop. For instance, younger pediatric patients—neonates, infants and children up to 10 years of age—have higher heart rates than full-grown adults and those aged 11-17. Thus, some devices developed for and used by adults—such as cardiac valves—cannot be used in young children without major redesigns. Similarly, off-the-shelf hip and knee joints generally are too large for kids; even the smallest versions (designed mostly for women and Asian populations) would be inappropriate for young athletes with damaged knee cartilage or those suffering from juvenile arthritis.
Comparable differences exist among the various pediatric patient groups as well. Infant-specific devices may not be effective in toddlers and most likely would be useless in teenagers, even if all three groups share conditions or diseases. Such disparity requires medtech firms to develop and test a different product for each patient subpopulation, regardless of their mutual affliction.
The unique characteristics of children and the relatively diminutive size of the pediatric market are two of the biggest barriers to pediatric device innovation. “It takes about $30 million to bring a [medical] device to market,” explained Michael G. Vitale, M.D., M.P.H., a Columbia University professor and pediatric spinal surgeon at Morgan Stanley Children’s Hospital in New York, N.Y., who also is a board member of the Pediatric Orthopaedic Society of North America, a Rosemont, Ill.-based organization founded in 1971 to advance orthopedic care for children.
“In a market where 3,000 or 4,000 cases can occur each year, it would take a company far longer to recover its investment compared with the adult market,” he continued. “The math just doesn’t work for smaller [patient] populations. If you need $30 million to get your device to market and the size of the market is such that you’ll only make $1 million a year, no business other than a humanitarian group or philanthropic organization is going to be willing to spend $30 million to get back $1 million. There’s not enough of a financial incentive to support the development of pediatric devices so alternate mechanisms and strategies are necessary.”
Even when the financial incentive (and desire) exists, though, companies still encounter roadblocks en route to product commercialization. Many firms have trouble recruiting pediatric participants in clinical trials due to parental consent and the difficulty in performing experimental procedures on children. In addition, the various subpopulations can limit the number of young recruits and add to the overall trial cost. As Campbell noted: “For pediatric devices with a limited market, it’s hard to convince investors to spend millions of dollars on a clinical trial. With adult products, it’s different—those devices are used by a lot of people. The market size justifies the cost but with kids it’s a lot tougher to rationalize the expense of a clinical trial. It has become a serious problem.”
|
Robert M. Campbell Jr., M.D., holding a model of the VEPTR II, which he developed. Photo courtesy of The Children’s Hospital of Philadelphia. |
Unpredictability and inconsistency from the FDA can make it difficult for companies to properly budget for new product development, too. Some manufacturers also have been discouraged by a lack of published data on pediatric devices but regulators are attempting to rectify the problem with a new regulation and related draft guidance that clarifies the kind of information companies must submit with pediatric device applications.
Under the proposal, published in a Feb. 19 Federal Register notice (78 Fed. Reg. 11,654), companies must submit “readily available” information about pediatric patients who might benefit from a device. Acceptable data includes publicly accessible material from medical journals, large surveys, patient registries, federal records, scientific research resources (MEDLINE and PubMed), online medical libraries (Cochrane and the Public Library of Science) and clinicaltrials.gov. The proposal bans information garnered from non-public sources such as internal marketing surveys, sales records, R&D reports, unpublished presentations and private consultations with clinicians.
In drafting the new rule, the FDA is attempting to better identify and track medical devices that could be used to treat children. The agency is requiring medtech firms to detail the ways in which a product will be used and estimate the number of U.S. pediatric patients who suffer from the disease the device treats or diagnoses. Companies also must provide a description of any affected pediatric subpopulations—either neonates (0-28 days), infants (younger than 2), children (aged 2-12) or adolescents (under 21).
The pediatric data rule applies to premarket approval applications, HDE requests and product development protocols, an alternative endorsement method that reverses the last two steps of FDA review and panel review. Manufacturers that fail to include pediatric information with their applications risk delaying the approval process. The FDA intends to use the data in these applications to report annually to Congress on the number of approved devices that could be used for a pediatric population as well as the number of approved devices that could be labeled for use in pediatric patients.
Gray’s Anatomy vs. Hamann-Todd
Campbell often jokes about the obsolescence of his operating room. “If a surgeon from the 1950s were to come into my operating room when I’m working on a patient, he would recognize many of the pediatric instruments and devices because there has been little progress since then,” he told Orthopedic Design & Technology. “I can’t tell you how many times I’ve been frustrated over the lack of modern medical devices for pediatric care. But you can’t hang it up when you don’t have what you need. You just make do with what you have.”
Orthopedic surgeons, however, haven’t really had much to work with in the last half-century or so. Most pediatric specialists have resorted to jury-rigging adult devices for kids or using products in an unapproved manner to help their patients. Pins normally reserved for the fixation of small bone fragments, for example, have been used as intramedullary rods in extremely small children. Iowa City, Iowa-based surgeon Stuart L. Weinstein, M.D., admits to using spinal rods in the legs of children because “they are the only thing that really fits.” He also has used adult arm implants in a child’s leg, specifically a humeral nail in the tibia. “Overall, we have to improvise quite frequently by using available products,” Weinstein disclosed to Orthopedics Today in 2010. “I have to use adult devices off-label because there is nothing else available.”
Campbell hasn’t always played by the rules, either. “You do what you have to do. Have I sawed devices in half? Sure. Have I used a device for another purpose? Sure. Does it work out 99 percent of the time? Yes, but it’s not ideal.”
Perfection, however, can be a difficult standard to achieve, especially in children. Their bodies are considerably smaller than adults, but their musculoskeletal structures grow and change as they mature. That growth, though, is not always uniform, so their devices must be able to accommodate the disjointed nature of their development (a prosthetic socket, for instance, might be outgrown but the pylon height may still be acceptable).
Accommodating for such variations in growth requires a comprehensive understanding of the juvenile anatomy, experts claim. The most successful pediatric device developers are connoisseurs of the juvenile physique, fully aware of the sensitivities of children’s growth plates (zones of cartilage at each end of the long bones), the hypermobility of their joints, the strength of their periosteum (the membrane lining the outer surface of all bones except large bone joints), the degree to which their femurs bow, and the number of additional bones they support upon their frames (94) compared with adults.
“It’s not necessarily the size that’s an issue, it’s knowing the anatomy of a child,” noted Nick Deeter, founder and board chairman of OrthoPediatrics Corp., a leading developer of juvenile orthopedic products based in Warsaw, Ind. “Kids are different. You have to account for growth plates which are not a factor in the adult market. When placing plates and screws on pediatric bones, it is critical to ensure the growth plates are avoided to prevent the potential for growth disturbances. Also, children’s bones have more bowing, like in the femur, therefore you cannot correct a deformity or fracture using a standard straight nail as in the adult market or it could damage the bone. There are a lot of variations in children that you don’t have in adults.”
OrthoPediatrics mastered all of those variations by securing exclusive rights to two of the world’s largest collections of pediatric skeletal data—the Hamann-Todd Human Osteological Collection and the Bolton-Brush Growth Study at Case Western Reserve University. The Hamann-Todd collection, now curated at the Cleveland Museum of Natural History, contains the records and bones of more than 3,000 people who died in the early 20th century. In most cases, the records show the identity of the donors along with their medical histories and in some cases, their photographs. Deeter said his company scanned all the pediatric skeletons in that collection to learn more about pediatric bone growth and unlock the mysteries of growth plates.
The Bolton-Brush Growth Study is globally considered to be the most extensive source of longitudinal human growth data. The study combines information from two separate investigations—one that tracked participant’s mental and physical growth and another that charted growth and development of the face and teeth. More than 4,000 kids from Cleveland underwent head-to-toe X-rays of their bodies to contribute to the database on mental/physical growth. The youngest children were three months old when the study began; all participants were X-rayed every three months during the first two years of their life and then less frequently after that. The Bolton-Brush studies have become one of the longest-running examinations of normal human health and development.
Data from both osteological collections have helped OrthoPediatrics develop customized products for children. Its intramedullary femoral nail, for example, features a smaller diameter and three proximal locking hole options—oblique, transverse and recon. It also has an anterior bow to mimic the normal anatomy of kids ages 8 to 16, according to a product brochure. “When we developed the intramedullary nail, we made it so it goes in the correct way every time. It has so many bends in it, [the nail] looks like it was run over by a truck,” Deeter quipped.
OrthoPediatrics also modeled its PediFlex nail and PediPlates based on information it extracted from the bones of long-dead children. Its flexible nail is used to treat long bone fractures and is manufactured in both titanium (1.5 mm-4.5 mm) and stainless steel (1.5 mm-4.0 mm) in increments of 0.5 millimeters. The PediPlates are used to correct angular deformities.
“OrthoPediatrics is doing a great job designing plates and screws for the pediatric population,” said Andrew Bowe, M.D., a pediatric orthopedic surgeon affiliated with Robert Wood Johnson University Hospital in New Brunswick, N.J. “In general, what surgeons look for in a pediatric device are ease of use, and ease of removal. The hardware is usually removed in children. If they sustain another injury, a plate or some other kind of implant can significantly complicate their treatment. There are some studies that show rigid hardware around the joint can alter some of the mechanics of that joint over the course of many years. It’s always better to get hardware out of kids whenever possible.”
During its tenancy, however, an implant must accommodate a child’s natural growth. Many bone plates and rods are lengthened or modified through revision surgery, but some promising new technologies eliminate the need for multiple procedures, lowering the risk of complications and potential infections. Resorbable implants are becoming popular with specialists in pediatric craniofacial and orthopedic deformities. The implants are rigid enough to support the repair or reconstruction of a deformity but eventually disappear without interfering with a child’s growth.
Likewise, researchers are tinkering with human tissue to regrow skin, cartilage, bone and heart valves. Several years ago, engineers at the Massachusetts Institute of Technology in Cambridge built a new tissue scaffold that stimulated bone and cartilage growth when it was transplanted into damaged knees and other joints. The scaffold had two layers—one mirrored bone and the other emulated cartilage. When implanted into a joint, the scaffold stimulated mesenchymal stem cells in the bone marrow to produce both new bone and cartilage. Life sciences experts are hopeful that such breakthroughs could eventually eliminate or at the very least, limit the immunocompatibility and biocompatibility problems associated with the implant materials currently available.
Nurturing Invention
Campbell got lucky when he developed the VEPTR. He received support from manufacturers (Techmedica and Synthes Spine Company), non-profit groups (NORD) and to some extent, the FDA. Such support, however, traditionally has been rare for pediatric device inventors. “We were extremely lucky that we found companies that could be responsive to the needs of children in spite of the many obstacles,” Campbell told a U.S. Senate Committee on Health, Education, Labor and Pensions in 2007. “Small companies can be responsive to small pediatric projects, but do not have large budgets for device development or the regulatory resources to secure FDA approval. Large publicly owned companies have those resources, but can’t justify non-profit pediatric device projects to their stockholders.”
The government tackled that funding conundrum six years ago by passing the FDA Amendments Act, a bill that removed profit restrictions on pediatric devices approved under the agency’s HDE program and authorized $6 million annually through 2012 for projects that linked inventors of juvenile products with potential manufacturers. Congress renewed the legislation last summer through the Medical Device User Fee Act; children’s advocates lauded lawmakers for preserving the HDE pediatric profit incentive but lamented the bill’s failure to address inconsistent insurance coverage often associated with HDE-approved kids’ devices.
Besides extending the HDE pediatric profit incentive, the reauthorized bill also requires the FDA to hold a public meeting and issue a report on efforts to accelerate the development of products for rare juvenile diseases. In addition, the legislation renewed funding for the Pediatric Device Consortia Grant Program, an initiative that subsidizes non-profit consortia that develop medical devices for youngsters.
The five FDA-funded consortia have helped advance the development of roughly 135 proposed pediatric medical devices. The consortia currently are managing 80 projects, including a robo-implant to treat severe pediatric spinal deformities and a device that uses two rare earth magnets to slowly reconfigure the chest in children with hollowed torsos.
Indeed, recent financial incentives have helped encourage innovation (the number of juvenile-specific medical products has jumped five-fold since 2008), but it may take a paradigm shift in both attitude and business models for children to finally get the attention they deserve from the device industry.
OrthoPediatrics’ Deeter suggests companies consider the long-term economic benefit pediatric devices can contribute to society.
“Developing a medical device for the pediatric population is an investment, there’s no denying that,” Deeter said. “But these devices are economically viable. There’s a product that can fix bowed legs on a child in about six weeks…think about that. If a child goes through life with really bowed legs, he can get rheumatoid arthritis in his knee or hip and you’ll eventually have to replace those knees or hips.
But by straightening out those legs when they’re young, you’ve spent $1,000 on a procedure and avoided spending $100,000 in the future when that kid eventually needs new joints. It’s a lot easier to fix kids when they’re young…and when you do that, you’ll find they kind of fix themselves, too.”