08.05.15
Tyber Medical LLC, a private-label manufacturer of spine and orthopedic implants, has acquired an exclusive licensing agreement for its BioTy antimicrobial surface coating technology.
The company offered few specifics of the deal, naming neither its licensing partner nor the applications best suited for the technology. In a news release Tyber executives only would identify the licensing partner as “a prestigious biomaterial research institution,” and said the technology would be used in “certain orthopedic applications.” The agreement also includes allowance for refining the manufacturing process, as well as ongoing funding for additional scientific studies.
Infection control is a critical concern in healthcare. Nowhere is that more evident than with surgical implants, where the body’s own defense mechanism will coat an implant with a “biofilm” layer. Biofilm is known to resist antibiotic therapy, the primary treatment option in infection control.
As a result, the cost impact of biofilm is significant. In fact, hospital-associated infections caused from biofilm on medical devices is estimated to cost the U.S. healthcare system $35.7 billion to $45 billion annually, according to the Centers for Disease Control and Prevention (CDC). Recent information released by the CDC shows that surgical site infection rates accounted for 31 percent of all healthcare-associated infections among hospitalized patients. In part, this is due to the widespread production, use and misuse of antibiotics, which have contributed to the next generation of drug resistant infectious organisms, commonly called “superbugs.”
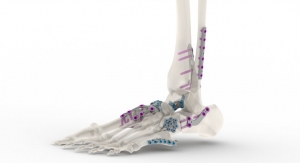
Tyber Medical’s BioTy Antimicrobial surface coating features technology that exhibits surface characteristics on a nanoscale that potentially can significantly reduce infections. Company executives said the BioTy process can be applied to various implant materials, including titanium, stainless steel, cobalt chrome and PEEK (polyether ether ketone).
“New research suggests that implant nanoscale features alone, without using pharmaceutical agents, can inhibit bacteria and virus functions to improve medical device performance,” said Thomas J. Webster, Ph.D., Art Zafiropoulo chairman and a chemical engineering professor at Northeastern University (Boston, Mass.), and a new advisory board member at Tyber Medical. “I’m pleased to be working with Tyber Medical to further develop the BioTy technology for use on a wide range of surgical implants.”
“We see three significant benefits resulting from our BioTy technology,” said Jeff Tyber, company founder and president. “Initially, it may enable us to be first to market with implants that exhibit exceptional infection control properties. Second, by tailoring implant surface characteristics, we can optimize certain qualities of the implant, such as bony ingrowth and attachment, to meet the needs of specific clinical applications. Lastly, the potential inherent antimicrobial properties of the BioTy process may negate the need and expense of pharmaceutical agents. As a result, cost effective infection control may be within our reach.”
Based in Morristown, N.J., Tyber Medical designs and develops Class II orthopedic systems. Its BioTy technology is the latest addition to a product portfolio in spine and orthopedics that includes headless compression trauma screws, an anterior cervical plating system, and a full line of spinal interbody spacers.
The company offered few specifics of the deal, naming neither its licensing partner nor the applications best suited for the technology. In a news release Tyber executives only would identify the licensing partner as “a prestigious biomaterial research institution,” and said the technology would be used in “certain orthopedic applications.” The agreement also includes allowance for refining the manufacturing process, as well as ongoing funding for additional scientific studies.
Infection control is a critical concern in healthcare. Nowhere is that more evident than with surgical implants, where the body’s own defense mechanism will coat an implant with a “biofilm” layer. Biofilm is known to resist antibiotic therapy, the primary treatment option in infection control.
As a result, the cost impact of biofilm is significant. In fact, hospital-associated infections caused from biofilm on medical devices is estimated to cost the U.S. healthcare system $35.7 billion to $45 billion annually, according to the Centers for Disease Control and Prevention (CDC). Recent information released by the CDC shows that surgical site infection rates accounted for 31 percent of all healthcare-associated infections among hospitalized patients. In part, this is due to the widespread production, use and misuse of antibiotics, which have contributed to the next generation of drug resistant infectious organisms, commonly called “superbugs.”
Tyber Medical’s BioTy Antimicrobial surface coating features technology that exhibits surface characteristics on a nanoscale that potentially can significantly reduce infections. Company executives said the BioTy process can be applied to various implant materials, including titanium, stainless steel, cobalt chrome and PEEK (polyether ether ketone).
“New research suggests that implant nanoscale features alone, without using pharmaceutical agents, can inhibit bacteria and virus functions to improve medical device performance,” said Thomas J. Webster, Ph.D., Art Zafiropoulo chairman and a chemical engineering professor at Northeastern University (Boston, Mass.), and a new advisory board member at Tyber Medical. “I’m pleased to be working with Tyber Medical to further develop the BioTy technology for use on a wide range of surgical implants.”
“We see three significant benefits resulting from our BioTy technology,” said Jeff Tyber, company founder and president. “Initially, it may enable us to be first to market with implants that exhibit exceptional infection control properties. Second, by tailoring implant surface characteristics, we can optimize certain qualities of the implant, such as bony ingrowth and attachment, to meet the needs of specific clinical applications. Lastly, the potential inherent antimicrobial properties of the BioTy process may negate the need and expense of pharmaceutical agents. As a result, cost effective infection control may be within our reach.”
Based in Morristown, N.J., Tyber Medical designs and develops Class II orthopedic systems. Its BioTy technology is the latest addition to a product portfolio in spine and orthopedics that includes headless compression trauma screws, an anterior cervical plating system, and a full line of spinal interbody spacers.