Maria Shepherd, President and Founder, Medi-Vantage08.14.19
With the continued surge of mergers and acquisitions, vertical integrations, and no clear U.S. policy or reform direction for our $3.6 trillion annual healthcare spend,1 the healthcare ecosystem remains in a phase of dramatic disruption.
What Is Keeping Hospital CEOs Awake at Night?
A study by the American College of Healthcare Executives (ACHE)2 involving community hospital CEOs (n=355) shows several common themes hospital CEOs grapple with today. Several of them, including the top issues outlined in Table 1, offer opportunities where medtech can help. At the top are Financial Challenges, followed by Government Mandates (reimbursement) and Patient Safety/Quality, which includes value-based care. The orthopedic medical device community can help with these top three issues.
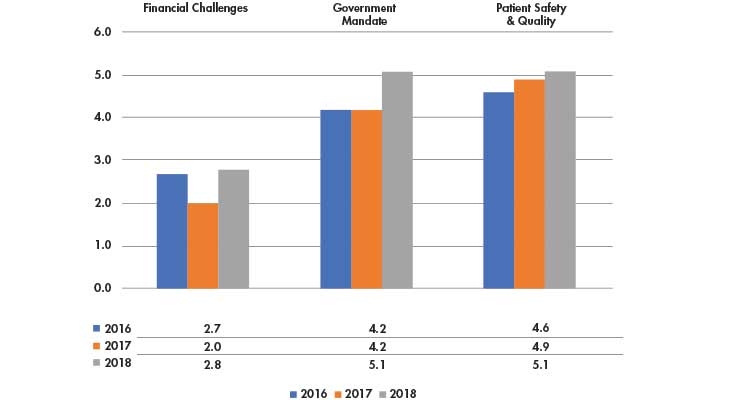
Table 1: Rank of CEOs’ top concerns over issues affecting their hospitals (on a scale of 1-10, with 1 representing the highest level of concern)2
The ACHE survey indicates hospital leadership is working to address the challenges of managing limited reimbursements against the rising costs of bringing talented staff (and retaining them) to provide the best possible care for their patients. Also indicated as concerning issues in the study were Personnel Shortages (#4), Behavioral Health/Addiction (#5), Patient Satisfaction (#6), Access to Care (#7), Physician-Hospital Relations (#8), Technology (#9), Population Health Management (#10) and Reorganization (#11).
Orthopedic Device Makers to the Rescue
There are many drivers in the healthcare system that apply cost pressure to hospital margins. Seventy percent of hospital CEOs are concerned that CMS regulations are a moving target.2 In addition to the obvious root causes, an AHA study3 predicts U.S. healthcare systems will lose $218 billion more in federal reimbursement payments by 2028, and private payers plan to follow the trend.
Medical devices that aid in reducing readmission rates help hospitals retain CMS reimbursement and address a major financial challenge. Penalties for readmissions are taxing on hospital budgets, and can drop CMS reimbursement by as much as 3 percent if those rates are too high. Private payers are also implementing penalty and credit models similar to those of CMS and hospitals are motivated to control readmission rates so they may achieve a superior negotiating position with payers to improve their rates or to become a Center of Excellence. Beyond concerns over reimbursement penalties, readmissions also drive up hospital costs, impacting overall profitability.
According to a study in Clinical Orthopaedics and Related Research,4 the all-cause orthopedics 30-day readmission rate was estimated at 4 percent. The unplanned portion of readmission rates for orthopedics was 3.4 percent. Longer length of stay correlated with increased 30-day readmissions and every additional day in the hospital increased that probability of readmission by 10 percent (Table 2).

Table 2: Unplanned readmission rates by orthopedic subspecialty4
One medical device company helping hospitals to reduce readmission rates is Avanos.5 Rather than use opioids in the OR, Avanos recommends helping patients manage pain in surgery using continuous peripheral nerve blocks. Once the surgery is over, the patient’s pain is managed through a postoperative pain pump, such as the ON-Q Pain Relief System. This combination of therapies offers patients pain relief without the side effects of traditional methods for surgical pain management, such as nausea, vomiting, grogginess, and the long-term potential for addiction.
Managing Value-Based Care
Since 2011, the number of accountable care organizations (ACOs) has grown from 61 to 923 in early 20176 (Table 3), but most healthcare organizations still rely on fee-for-service as their greatest source of income.
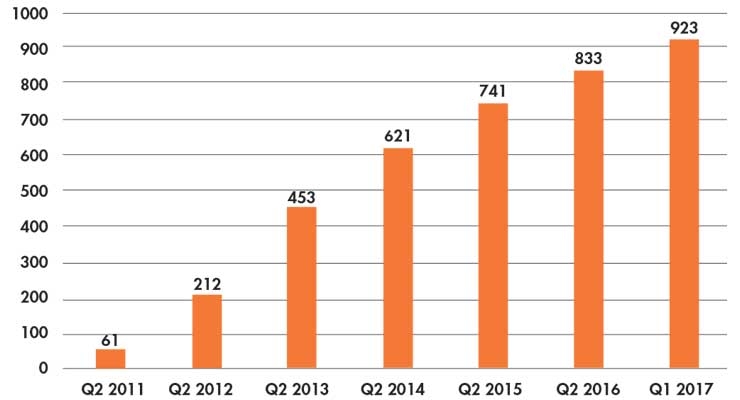
Table 3: Growth in number of ACOs6
Gather a group of orthopedic surgeons together and infection prevention in orthopedic medical device procedures will be at the forefront of their conversation. The challenge to improve the current standard of care and increase the probability that patients do not develop a surgically acquired infection is the level of rigor required to gain FDA approval for combination devices.
Entrepreneurs in this space are inventing new orthopedic devices coated with antimicrobial agents and antibiotic-eluting bone graft substitutes. Many agree there is a need for antimicrobial-based devices since implant-related infections remain among the leading root causes of implant failure. In addition, the cost to the healthcare system and patient is very high.7
The Medi-Vantage Perspective
Re-admission rates are still too high for orthopedic surgery. What tools are in your company’s bag that can be engineered to help address what’s keeping hospital CEOs up at night?
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.
What Is Keeping Hospital CEOs Awake at Night?
A study by the American College of Healthcare Executives (ACHE)2 involving community hospital CEOs (n=355) shows several common themes hospital CEOs grapple with today. Several of them, including the top issues outlined in Table 1, offer opportunities where medtech can help. At the top are Financial Challenges, followed by Government Mandates (reimbursement) and Patient Safety/Quality, which includes value-based care. The orthopedic medical device community can help with these top three issues.

Table 1: Rank of CEOs’ top concerns over issues affecting their hospitals (on a scale of 1-10, with 1 representing the highest level of concern)2
The ACHE survey indicates hospital leadership is working to address the challenges of managing limited reimbursements against the rising costs of bringing talented staff (and retaining them) to provide the best possible care for their patients. Also indicated as concerning issues in the study were Personnel Shortages (#4), Behavioral Health/Addiction (#5), Patient Satisfaction (#6), Access to Care (#7), Physician-Hospital Relations (#8), Technology (#9), Population Health Management (#10) and Reorganization (#11).
Orthopedic Device Makers to the Rescue
There are many drivers in the healthcare system that apply cost pressure to hospital margins. Seventy percent of hospital CEOs are concerned that CMS regulations are a moving target.2 In addition to the obvious root causes, an AHA study3 predicts U.S. healthcare systems will lose $218 billion more in federal reimbursement payments by 2028, and private payers plan to follow the trend.
Medical devices that aid in reducing readmission rates help hospitals retain CMS reimbursement and address a major financial challenge. Penalties for readmissions are taxing on hospital budgets, and can drop CMS reimbursement by as much as 3 percent if those rates are too high. Private payers are also implementing penalty and credit models similar to those of CMS and hospitals are motivated to control readmission rates so they may achieve a superior negotiating position with payers to improve their rates or to become a Center of Excellence. Beyond concerns over reimbursement penalties, readmissions also drive up hospital costs, impacting overall profitability.
According to a study in Clinical Orthopaedics and Related Research,4 the all-cause orthopedics 30-day readmission rate was estimated at 4 percent. The unplanned portion of readmission rates for orthopedics was 3.4 percent. Longer length of stay correlated with increased 30-day readmissions and every additional day in the hospital increased that probability of readmission by 10 percent (Table 2).

Table 2: Unplanned readmission rates by orthopedic subspecialty4
One medical device company helping hospitals to reduce readmission rates is Avanos.5 Rather than use opioids in the OR, Avanos recommends helping patients manage pain in surgery using continuous peripheral nerve blocks. Once the surgery is over, the patient’s pain is managed through a postoperative pain pump, such as the ON-Q Pain Relief System. This combination of therapies offers patients pain relief without the side effects of traditional methods for surgical pain management, such as nausea, vomiting, grogginess, and the long-term potential for addiction.
Managing Value-Based Care
Since 2011, the number of accountable care organizations (ACOs) has grown from 61 to 923 in early 20176 (Table 3), but most healthcare organizations still rely on fee-for-service as their greatest source of income.

Table 3: Growth in number of ACOs6
Gather a group of orthopedic surgeons together and infection prevention in orthopedic medical device procedures will be at the forefront of their conversation. The challenge to improve the current standard of care and increase the probability that patients do not develop a surgically acquired infection is the level of rigor required to gain FDA approval for combination devices.
Entrepreneurs in this space are inventing new orthopedic devices coated with antimicrobial agents and antibiotic-eluting bone graft substitutes. Many agree there is a need for antimicrobial-based devices since implant-related infections remain among the leading root causes of implant failure. In addition, the cost to the healthcare system and patient is very high.7
The Medi-Vantage Perspective
Re-admission rates are still too high for orthopedic surgery. What tools are in your company’s bag that can be engineered to help address what’s keeping hospital CEOs up at night?
References
- http://bit.ly/odt190701
- http://bit.ly/odt190702
- http://bit.ly/odt190703
- http://bit.ly/odt190704
- http://bit.ly/odt190705
- http://bit.ly/odt190706
- http://bit.ly/odt190707
Maria Shepherd has more than 20 years of leadership experience in medical device/life science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.