Mark Crawford, Contributing Writer08.14.19
Orthopedic design teams are under increasing pressure to deliver complex, high-quality products into the marketplace, as quickly as possible. National and international regulatory changes (such as the impending EU Medical Device Regulation) add to the challenge. As the medical device regulatory landscape becomes increasingly complicated, more OEMs are turning to their contract manufacturers (CM) for guidance and advice regarding maintaining and improving designs for their legacy products and new devices.
Additive manufacturing (AM) drives a number of orthopedic designs and product applications—some companies, in fact, only design products for AM. Top applications for AM include prototyping and instrument and implant production. AM continues to make impressive design strides, such as incorporating intricate lattice structures into devices that cannot be produced (or be practical or cost-effective) with traditional manufacturing methods. Perhaps the greatest overall benefit of AM to the orthopedic device industry is the rapid production of sophisticated prototypes that allow device designers to make quick, well-informed decisions that speed up production.
“The medical device market seeks faster release of products into the market, which is possible with more design iterations utilizing 3D-printed prototypes, in both plastic and metal,” said Dave Neal, president of STAT Design, a Lake Hopatcong, N.J.-based provider of design, development, and 3D printing services.
One of the hottest orthopedic markets is spine, especially over the last few years. Victoria Trafka, president and lead engineer for Engineering and Quality Solutions, a Colorado Springs, Colo.-based contract engineering and development company focused on orthopedic and spine implants and surgical instruments, sees no signs of the market slowing down anytime soon.
“The products being developed are quite varied, ranging from ‘me-too’ products to truly innovative devices that answer unmet needs or use new technology,” she said.
For example, one of these innovative devices is the expandable spinal cage. The latest designs include the ability to expand in height, adjust lordosis, and even expand the footprint. These cages contain very intricate mechanisms packed into small spaces, which must be durable enough to withstand the same rigorous strength testing as solid devices. Expandable cervical cages have also entered the market.
Whether it is a high-growth market like spinal cages, or updating and adding new capabilities to legacy products, OEMs increasingly seek advice from their CMs on how to improve their competitiveness. “This means we must constantly be on the lookout for ways to improve our design efficiencies, whether through automation or using improved design software tools or manufacturing and/or inspection techniques and equipment,” said Charlie Wing, engineering director for Orchid Design, a Shelton, Conn.-based contract product development company that specializes in orthopedic and spinal implants and instruments.
What OEMs Want
When it comes to design, OEMs want innovation, product differentiation, low cost, and speed to market. Requests range from help for redesigning legacy products to boost market share to designing new innovative products for highly specific markets. Other OEMs recognize they have gaps in their product portfolios that they want to fill in quickly. “These companies approach us and ask for not just a product concept or component of a project, but more of a complete manufactured final product family that can be launched and distributed by their sales force,” said Wing.
Implants are historically a strong segment of the market and OEMs are always looking for ways to improve implant function, osseointegration, and patient outcomes. For example, OEMs are interested in material properties, surface treatments, and coatings that improve performance and reduce the risk of infection. “An increased number of OEMs are focused on implant surface enhancements, such as nanoscale textures that improve bacterial resistance or coatings that improve tribological or boney in-growth properties,” said Wing.
OEMs are also eager to simplify surgical products and instruments and/or make them more intuitive, which reduces costs while also improving market share and brand awareness.
“This typically requires redesigning instruments and implants to work more smoothly and easily, sometimes with fewer features or options,” said Trafka. “A complex system with lots of options doesn’t always mean it’s better. OEMs are listening to surgeons who really want systems to work well and be easy to use—every time.”
Surgeons are very particular about the “feel” of the products and instruments they use, especially if ease of use speeds up surgical procedures and reduces their fatigue in the operating room. In response, more OEMs are paying closer attention to human factors engineering and end-user input during initial design studies.
“OEMs are asking for evidence-based designs and designs that are shaped and informed by human factors, which are subjected to iterative testing during each step of engineering and industrial design development,” said Bryce G. Rutter, CEO of Metaphase Design Group, a St. Louis, Mo.-based provider of ergonomically designed hand-intensive products and packaging.
Integrating human factors engineering into orthopedic products makes them intuitive and easier to use. This is a simple and lower-cost way to add value and enhance functionality. The trend is, in part, driven by the FDA’s requirement to conduct usability research—both formative and summative usability studies—on all types of users who interact with a product’s design. Even with this directive, many companies wait until the later stages of development to consider human factors engineering, after significant investments in time and money have been made. This approach can easily backfire—for example, if validation studies result in the FDA ordering design changes that delay approval and product launch, it is quite possible these changes would not have been needed if human factors engineering had been used earlier in the design process.
New Trends and Technologies
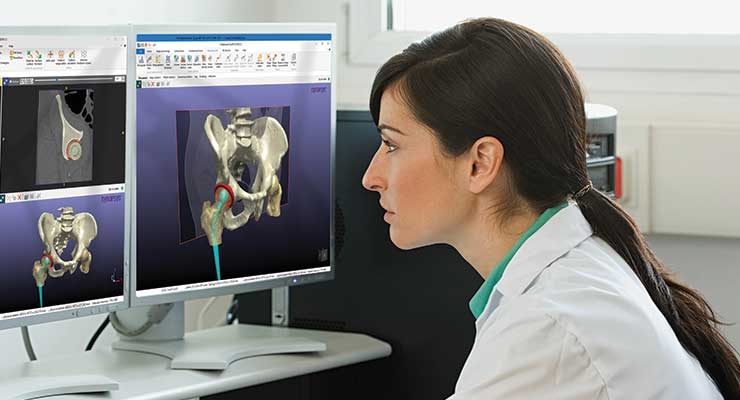
Computer simulation is rapidly becoming a preferred design optimization tool in orthopedics. Increasingly sophisticated computational simulation techniques will eventually limit the need for bench-top testing and clinical trials. Other useful modeling technologies are virtual reality (VR) and augmented reality (AR), which help designers and manufacturers visualize and interact with their designs in three dimensions without the need for fabricating expensive functional prototypes. In fact, computer simulation may eventually be approved by the FDA as a way to evaluate mechanical performance of devices that are submitted to the FDA.
“In the last few years, the ASTM committees that develop and maintain orthopedic testing standards have been working with FDA representatives to develop standards for carrying out modeling and simulation on devices,” said Trafka.
“We’re seeing increased use of radiological and 3D image-based computer simulation by orthopedic companies,” confirmed Philippe Young, engineering director for the Synopsys Simpleware Product Group, an Exeter, U.K.-based provider of software for 3D image data visualization, analysis, and model generation. “Simulation is being used both for custom patient-specific design for treating pathologies and new device designs to make sure they fit the target population. This kind of simulation from 3D images can look at anything from motion analysis to ensure a proper fit to more complex applications such as evaluating load-carrying behavior and the effect of electromagnetic energy on patients.”
The Internet of Things (IoT) also impacts medical device design. “Smart” devices that transmit real-time data, using embedded sensors that provide in-situ feedback, are in high demand. “Meeting these needs efficiently involves a cross-disciplinary approach, with software engineers working beside mechanical engineers and injection molding design professionals,” said Neal.
From the point of view of development and production, cloud-based platforms enable development teams to work on the same project and technical product documentation in a truly collaborative way, 24/7, from different global locations. IoT technologies also enable machines to “talk” to one another, resulting in less downtime during production. “Smarter development processes that are more user-friendly and less prone to user errors can achieve efficiency improvements with no extra expertise,” said Wing. “These IoT platforms require an open mindset, but usually only minimal training to save time and reduce costs.”
Additive manufacturing is producing increasingly complex product features, such as struts and pore size. Even though AM capabilities are rapidly advancing, most orthopedic devices still require secondary machining after being 3D-printed. This is because many have threads, holes, or recesses that are needed for instrument interface or connection of another device. For proper function, these features require a smooth surface finish or tight tolerances that cannot be achieved using AM. It is also critical that AM products are free of dust, chips, or debris that could compromise biocompatibility or the operation of embedded electronics or optics. With AM, any residual powders must be carefully removed. Unitedcoatings Group, for example, through its medical division, offers a process that removes residual metal powder and other contaminants from 3D-printed parts after secondary finishing operations are complete. Post-treatment cleaning can also be applied to porous structures.
AM is used to print custom-made orthopedic implants using patient-specific measurements. “Image data from MRI and CT scans can accurately capture patient information that is then used to create custom-made implants,” said Young. “We have also observed considerable good work being done using additive manufacturing to add lattice structures to orthopedic devices at the design stage—for example, to reduce weight.”
By not having to invest in expensive castings or molds, additive manufacturing brings down the cost of low-quantity production runs. AM technology also makes it possible to “shell out” metal surgical instruments, making them lighter in weight without compromising their structural integrity. “Lighter-weight instruments provide other production benefits, such as cooling down faster after autoclave sterilization and lower costs for shipping loaner sets,” said Neal.
Spinal interbody implants and acetabular cups are becoming routine 3D-printed products. However, implants that bear loads other than pure compression, such as hip stems, remain challenging to date. “As 3D printing continues to evolve as a manufacturing solution, designers will have more ‘out-of-the-box’ opportunities to create cost savings and open up new possibilities for products that weren’t available or affordable before,” said Wing.
Improved Analytical Software
A growing array of solid model software and finite element analysis (FEA) software packages can be used to speed up product development and time to market. FEA is a sophisticated computer program that predicts how a proposed medical device will respond to real-world forces such as vibration, load, heat, fluid flow, electrostatics, and other physical effects. Finite element analysis will reveal a product’s design weaknesses and the most probable points of failure, which can then be fixed through redesign, increasing quality, durability, and reliability.
For complex parts, more medical device designers are utilizing geometric dimensioning and tolerancing (GD&T), which takes a 3D view of components rather than the traditional 2D (X-Y) coordinate approach. GD&T is a more accurate process for defining measurements for complex curvilinear, ergonomic shapes and the tolerances required for fitting complex and/or miniaturized parts together. As a result, these high-precision parts are more likely to meet performance specifications and pass inspection than parts generated from 2D drawings.
GD&T then leads to “model-based definition,” an increasingly popular approach in product design and manufacturing. Model-based definition (MBD) is essentially a 3D model annotated with all the necessary manufacturing information so a detailed drawing with all GD&T is no longer required to make the part. This way, designers can share their 3D models with all project personnel and be confident the MBD contains all the data and tolerances required to build the final product, thus saving time and reducing the risk of errors. “Continuous improvement initiatives are looking at MBD due to these potential cost and efficiency savings, as well as improving overall processing time,” said Wing.
Regulatory Challenges
Recent FDA guidance documents call for more clinical and non-clinical data for medical device designs. In Europe, the new Medical Device Regulation will also require additional technical documentation. This all creates more work for engineers and design teams, especially as some devices become reclassified to higher classes, while others previously considered exempt will fall under the new legislation. Even though the FDA still emphasizes a least-burdensome approach, the agency and other notified bodies have significantly increased the number and intensity of audits over the last 10 years because some companies have done a poor job of documentation in an effort to reduce costs.
“The challenge for design teams today is to make sure the regulatory changes do not negatively affect development costs, time to market, and product innovation,” said Wing. “Innovation will require clinical evidence of benefit. Clinical evidence requires time and money. Although positive for the overall economics of the healthcare system, it will be challenging to get funding to innovate based on longer-term return on investment.”
These challenges make human factors engineering and computer modeling and simulation even more attractive for streamlining innovation and controlling costs. In recognition of this, the FDA is in the process of developing guidance for establishing the credibility of computational models within the medical device clearance space. “This is definitely the future for designing orthopedic devices and getting them through the FDA process,” said Kerim Genc, business development manager for the Synopsys Simpleware Product Group and member of the American Society of Mechanical Engineers Validation and Verification (V&V) Committee, which is advising the FDA on this topic. “We are discussing how V&V guidance documents can be applied to patient-specific simulation workflows within the context of medical device clearance submissions. We hope that our input helps promote and accelerate the adoption of simulation-based design.”
“The FDA requirement for formative and summative studies is fundamentally changing how we do new product development,” Rutter added. “Because of the regulatory demands for iterative studies, human factors engineering is emerging as a core competency in many medical companies, where design decisions are based on user needs, behavior, emotion, and how the body works.”
Wing agreed.
“Too many engineers working at medical device companies today design products with little or no operating room experience,” he noted. “Designing products for surgeons or marketing professionals who relay what are supposedly the customer needs, results in products that miss the mark. There is no substitute for an engineer designer standing in the operating room and documenting the struggles that surgeon users face, but perhaps don’t even notice, as they operate.”
Trafka observed that she has seen multiple requests for design history file (DHF) remediations in recent years. Because DHF deficiencies are a major source of 483 letters in the medical device industry, FDA inspectors are scrutinizing DHFs more than ever before.
“In some cases,” said Trafka, “the deficiencies are related to products that have been on the market a long time and the DHF practices from many years ago just aren’t sufficient today. In other cases, documentation problems exist for newer products and companies just don’t have the expertise or procedures in place to fully comply with regulations.”
Because of this trend, companies are dedicating more resources to remediating DHFs for existing products, reviewing in-process development efforts more carefully, and revamping their design controls procedures to ensure their design files are compliant. This is happening at all sizes of organizations, from large, established OEMs all the way to smaller companies and startups.
“The internal effect of the DHF remediation effort is a slowing of new development as resources are shifted to improve the documentation of legacy products and answer corrective actions,” said Trafka. “However, the ultimate effect of stronger DHFs is a better product with fewer problems. Applying proper design controls helps ensure that the product is safe, does what it is intended to do with fewer problems, is more reliable and predictable, and meets market needs.”
On the Horizon
New technologies and advanced materials will continue to provide a greater variety of design options for medical devices. Additive manufacturing, sensor technologies, automation, robotics, VR and AR, and data analytics are taking hold in the orthopedic design space and offer fantastic design and manufacturing potential. Newer biomaterials such as silicon nitride and molybdenum rhenium are getting more attention—for example, molybdenum rhenium is being used in spinal applications and has potential in the foot and ankle markets.
Robotics is an exciting field in orthopedics, with considerable research and development being conducted regarding surgeon interfaces. For example, CARLO (Cold Ablation Robot-Guided Laser Osteotome) is a robot arm controlled by a surgeon that performs contactless laser bone ablation—the surgical removal of a section of diseased bone. Developed by Basel, Switzerland-based AOT AG, the device uses advanced navigation and control software to allow surgeons to perform bone operations in freely defined, curved, and functional sectional configurations, which cannot be accomplished with conventional tools such as saws, drills, or fraises.
“It is an exciting time to be a designer,” enthused Rutter. “Never have we used so much science to shape and inform design, especially with human factors engineering. Knowing how people think, feel, and behave provides fundamental insights into user needs and specific product performance requirements. And now we have some amazing technologies that allow us to capture the difference between what people say they do and how they really behave—the holy grail of world-class design.”
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.
Additive manufacturing (AM) drives a number of orthopedic designs and product applications—some companies, in fact, only design products for AM. Top applications for AM include prototyping and instrument and implant production. AM continues to make impressive design strides, such as incorporating intricate lattice structures into devices that cannot be produced (or be practical or cost-effective) with traditional manufacturing methods. Perhaps the greatest overall benefit of AM to the orthopedic device industry is the rapid production of sophisticated prototypes that allow device designers to make quick, well-informed decisions that speed up production.
“The medical device market seeks faster release of products into the market, which is possible with more design iterations utilizing 3D-printed prototypes, in both plastic and metal,” said Dave Neal, president of STAT Design, a Lake Hopatcong, N.J.-based provider of design, development, and 3D printing services.
One of the hottest orthopedic markets is spine, especially over the last few years. Victoria Trafka, president and lead engineer for Engineering and Quality Solutions, a Colorado Springs, Colo.-based contract engineering and development company focused on orthopedic and spine implants and surgical instruments, sees no signs of the market slowing down anytime soon.
“The products being developed are quite varied, ranging from ‘me-too’ products to truly innovative devices that answer unmet needs or use new technology,” she said.
For example, one of these innovative devices is the expandable spinal cage. The latest designs include the ability to expand in height, adjust lordosis, and even expand the footprint. These cages contain very intricate mechanisms packed into small spaces, which must be durable enough to withstand the same rigorous strength testing as solid devices. Expandable cervical cages have also entered the market.
Whether it is a high-growth market like spinal cages, or updating and adding new capabilities to legacy products, OEMs increasingly seek advice from their CMs on how to improve their competitiveness. “This means we must constantly be on the lookout for ways to improve our design efficiencies, whether through automation or using improved design software tools or manufacturing and/or inspection techniques and equipment,” said Charlie Wing, engineering director for Orchid Design, a Shelton, Conn.-based contract product development company that specializes in orthopedic and spinal implants and instruments.
What OEMs Want
When it comes to design, OEMs want innovation, product differentiation, low cost, and speed to market. Requests range from help for redesigning legacy products to boost market share to designing new innovative products for highly specific markets. Other OEMs recognize they have gaps in their product portfolios that they want to fill in quickly. “These companies approach us and ask for not just a product concept or component of a project, but more of a complete manufactured final product family that can be launched and distributed by their sales force,” said Wing.
Implants are historically a strong segment of the market and OEMs are always looking for ways to improve implant function, osseointegration, and patient outcomes. For example, OEMs are interested in material properties, surface treatments, and coatings that improve performance and reduce the risk of infection. “An increased number of OEMs are focused on implant surface enhancements, such as nanoscale textures that improve bacterial resistance or coatings that improve tribological or boney in-growth properties,” said Wing.
OEMs are also eager to simplify surgical products and instruments and/or make them more intuitive, which reduces costs while also improving market share and brand awareness.
“This typically requires redesigning instruments and implants to work more smoothly and easily, sometimes with fewer features or options,” said Trafka. “A complex system with lots of options doesn’t always mean it’s better. OEMs are listening to surgeons who really want systems to work well and be easy to use—every time.”
Surgeons are very particular about the “feel” of the products and instruments they use, especially if ease of use speeds up surgical procedures and reduces their fatigue in the operating room. In response, more OEMs are paying closer attention to human factors engineering and end-user input during initial design studies.
“OEMs are asking for evidence-based designs and designs that are shaped and informed by human factors, which are subjected to iterative testing during each step of engineering and industrial design development,” said Bryce G. Rutter, CEO of Metaphase Design Group, a St. Louis, Mo.-based provider of ergonomically designed hand-intensive products and packaging.
Integrating human factors engineering into orthopedic products makes them intuitive and easier to use. This is a simple and lower-cost way to add value and enhance functionality. The trend is, in part, driven by the FDA’s requirement to conduct usability research—both formative and summative usability studies—on all types of users who interact with a product’s design. Even with this directive, many companies wait until the later stages of development to consider human factors engineering, after significant investments in time and money have been made. This approach can easily backfire—for example, if validation studies result in the FDA ordering design changes that delay approval and product launch, it is quite possible these changes would not have been needed if human factors engineering had been used earlier in the design process.
New Trends and Technologies
Computer simulation is rapidly becoming a preferred design optimization tool in orthopedics. Increasingly sophisticated computational simulation techniques will eventually limit the need for bench-top testing and clinical trials. Other useful modeling technologies are virtual reality (VR) and augmented reality (AR), which help designers and manufacturers visualize and interact with their designs in three dimensions without the need for fabricating expensive functional prototypes. In fact, computer simulation may eventually be approved by the FDA as a way to evaluate mechanical performance of devices that are submitted to the FDA.
“In the last few years, the ASTM committees that develop and maintain orthopedic testing standards have been working with FDA representatives to develop standards for carrying out modeling and simulation on devices,” said Trafka.
“We’re seeing increased use of radiological and 3D image-based computer simulation by orthopedic companies,” confirmed Philippe Young, engineering director for the Synopsys Simpleware Product Group, an Exeter, U.K.-based provider of software for 3D image data visualization, analysis, and model generation. “Simulation is being used both for custom patient-specific design for treating pathologies and new device designs to make sure they fit the target population. This kind of simulation from 3D images can look at anything from motion analysis to ensure a proper fit to more complex applications such as evaluating load-carrying behavior and the effect of electromagnetic energy on patients.”
The Internet of Things (IoT) also impacts medical device design. “Smart” devices that transmit real-time data, using embedded sensors that provide in-situ feedback, are in high demand. “Meeting these needs efficiently involves a cross-disciplinary approach, with software engineers working beside mechanical engineers and injection molding design professionals,” said Neal.
From the point of view of development and production, cloud-based platforms enable development teams to work on the same project and technical product documentation in a truly collaborative way, 24/7, from different global locations. IoT technologies also enable machines to “talk” to one another, resulting in less downtime during production. “Smarter development processes that are more user-friendly and less prone to user errors can achieve efficiency improvements with no extra expertise,” said Wing. “These IoT platforms require an open mindset, but usually only minimal training to save time and reduce costs.”
Additive manufacturing is producing increasingly complex product features, such as struts and pore size. Even though AM capabilities are rapidly advancing, most orthopedic devices still require secondary machining after being 3D-printed. This is because many have threads, holes, or recesses that are needed for instrument interface or connection of another device. For proper function, these features require a smooth surface finish or tight tolerances that cannot be achieved using AM. It is also critical that AM products are free of dust, chips, or debris that could compromise biocompatibility or the operation of embedded electronics or optics. With AM, any residual powders must be carefully removed. Unitedcoatings Group, for example, through its medical division, offers a process that removes residual metal powder and other contaminants from 3D-printed parts after secondary finishing operations are complete. Post-treatment cleaning can also be applied to porous structures.
AM is used to print custom-made orthopedic implants using patient-specific measurements. “Image data from MRI and CT scans can accurately capture patient information that is then used to create custom-made implants,” said Young. “We have also observed considerable good work being done using additive manufacturing to add lattice structures to orthopedic devices at the design stage—for example, to reduce weight.”
By not having to invest in expensive castings or molds, additive manufacturing brings down the cost of low-quantity production runs. AM technology also makes it possible to “shell out” metal surgical instruments, making them lighter in weight without compromising their structural integrity. “Lighter-weight instruments provide other production benefits, such as cooling down faster after autoclave sterilization and lower costs for shipping loaner sets,” said Neal.
Spinal interbody implants and acetabular cups are becoming routine 3D-printed products. However, implants that bear loads other than pure compression, such as hip stems, remain challenging to date. “As 3D printing continues to evolve as a manufacturing solution, designers will have more ‘out-of-the-box’ opportunities to create cost savings and open up new possibilities for products that weren’t available or affordable before,” said Wing.
Improved Analytical Software
A growing array of solid model software and finite element analysis (FEA) software packages can be used to speed up product development and time to market. FEA is a sophisticated computer program that predicts how a proposed medical device will respond to real-world forces such as vibration, load, heat, fluid flow, electrostatics, and other physical effects. Finite element analysis will reveal a product’s design weaknesses and the most probable points of failure, which can then be fixed through redesign, increasing quality, durability, and reliability.
For complex parts, more medical device designers are utilizing geometric dimensioning and tolerancing (GD&T), which takes a 3D view of components rather than the traditional 2D (X-Y) coordinate approach. GD&T is a more accurate process for defining measurements for complex curvilinear, ergonomic shapes and the tolerances required for fitting complex and/or miniaturized parts together. As a result, these high-precision parts are more likely to meet performance specifications and pass inspection than parts generated from 2D drawings.
GD&T then leads to “model-based definition,” an increasingly popular approach in product design and manufacturing. Model-based definition (MBD) is essentially a 3D model annotated with all the necessary manufacturing information so a detailed drawing with all GD&T is no longer required to make the part. This way, designers can share their 3D models with all project personnel and be confident the MBD contains all the data and tolerances required to build the final product, thus saving time and reducing the risk of errors. “Continuous improvement initiatives are looking at MBD due to these potential cost and efficiency savings, as well as improving overall processing time,” said Wing.
Regulatory Challenges
Recent FDA guidance documents call for more clinical and non-clinical data for medical device designs. In Europe, the new Medical Device Regulation will also require additional technical documentation. This all creates more work for engineers and design teams, especially as some devices become reclassified to higher classes, while others previously considered exempt will fall under the new legislation. Even though the FDA still emphasizes a least-burdensome approach, the agency and other notified bodies have significantly increased the number and intensity of audits over the last 10 years because some companies have done a poor job of documentation in an effort to reduce costs.
“The challenge for design teams today is to make sure the regulatory changes do not negatively affect development costs, time to market, and product innovation,” said Wing. “Innovation will require clinical evidence of benefit. Clinical evidence requires time and money. Although positive for the overall economics of the healthcare system, it will be challenging to get funding to innovate based on longer-term return on investment.”
These challenges make human factors engineering and computer modeling and simulation even more attractive for streamlining innovation and controlling costs. In recognition of this, the FDA is in the process of developing guidance for establishing the credibility of computational models within the medical device clearance space. “This is definitely the future for designing orthopedic devices and getting them through the FDA process,” said Kerim Genc, business development manager for the Synopsys Simpleware Product Group and member of the American Society of Mechanical Engineers Validation and Verification (V&V) Committee, which is advising the FDA on this topic. “We are discussing how V&V guidance documents can be applied to patient-specific simulation workflows within the context of medical device clearance submissions. We hope that our input helps promote and accelerate the adoption of simulation-based design.”
“The FDA requirement for formative and summative studies is fundamentally changing how we do new product development,” Rutter added. “Because of the regulatory demands for iterative studies, human factors engineering is emerging as a core competency in many medical companies, where design decisions are based on user needs, behavior, emotion, and how the body works.”
Wing agreed.
“Too many engineers working at medical device companies today design products with little or no operating room experience,” he noted. “Designing products for surgeons or marketing professionals who relay what are supposedly the customer needs, results in products that miss the mark. There is no substitute for an engineer designer standing in the operating room and documenting the struggles that surgeon users face, but perhaps don’t even notice, as they operate.”
Trafka observed that she has seen multiple requests for design history file (DHF) remediations in recent years. Because DHF deficiencies are a major source of 483 letters in the medical device industry, FDA inspectors are scrutinizing DHFs more than ever before.
“In some cases,” said Trafka, “the deficiencies are related to products that have been on the market a long time and the DHF practices from many years ago just aren’t sufficient today. In other cases, documentation problems exist for newer products and companies just don’t have the expertise or procedures in place to fully comply with regulations.”
Because of this trend, companies are dedicating more resources to remediating DHFs for existing products, reviewing in-process development efforts more carefully, and revamping their design controls procedures to ensure their design files are compliant. This is happening at all sizes of organizations, from large, established OEMs all the way to smaller companies and startups.
“The internal effect of the DHF remediation effort is a slowing of new development as resources are shifted to improve the documentation of legacy products and answer corrective actions,” said Trafka. “However, the ultimate effect of stronger DHFs is a better product with fewer problems. Applying proper design controls helps ensure that the product is safe, does what it is intended to do with fewer problems, is more reliable and predictable, and meets market needs.”
On the Horizon
New technologies and advanced materials will continue to provide a greater variety of design options for medical devices. Additive manufacturing, sensor technologies, automation, robotics, VR and AR, and data analytics are taking hold in the orthopedic design space and offer fantastic design and manufacturing potential. Newer biomaterials such as silicon nitride and molybdenum rhenium are getting more attention—for example, molybdenum rhenium is being used in spinal applications and has potential in the foot and ankle markets.
Robotics is an exciting field in orthopedics, with considerable research and development being conducted regarding surgeon interfaces. For example, CARLO (Cold Ablation Robot-Guided Laser Osteotome) is a robot arm controlled by a surgeon that performs contactless laser bone ablation—the surgical removal of a section of diseased bone. Developed by Basel, Switzerland-based AOT AG, the device uses advanced navigation and control software to allow surgeons to perform bone operations in freely defined, curved, and functional sectional configurations, which cannot be accomplished with conventional tools such as saws, drills, or fraises.
“It is an exciting time to be a designer,” enthused Rutter. “Never have we used so much science to shape and inform design, especially with human factors engineering. Knowing how people think, feel, and behave provides fundamental insights into user needs and specific product performance requirements. And now we have some amazing technologies that allow us to capture the difference between what people say they do and how they really behave—the holy grail of world-class design.”
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.