Michael Barbella, Managing Editor05.17.21
The odds were stacked against “Bubba” well before his untimely world debut.
Born six weeks prematurely nearly three years ago, the neonate greeted this life without the ability to see or properly breathe. Doctors detected trouble with the boy’s bones while in-utero, but neither the source nor the extent of that trouble was evident until his birth.
Besides producing abnormally short arms and legs, Bubba’s gene mutation also beget an extremely narrow rib cage, which affected the boy’s respiration. As he grew, other genetic deficiencies emerged, most notably blindness and a weak heart.
Within three months, doctors diagnosed Bubba with Jeune syndrome, a form of congenital dwarfism characterized by a bell-shaped chest cavity, short ribs, and shortened arm and leg bones. Other skeletal abnormalities associated with the disorder include extra fingers and toes (polydactyly), unusually shaped clavicles and pelvic bones, and cone-shaped ends of the long arm and leg bones.
Jeune’s most distinctive feature can also be its most lethal: Left untreated, the condition (also known as asphyxiating thoracic dystrophy) dooms most babies to an abbreviated, tortuous lifespan as they slowly and progressively suffocate. Half of all Jeune syndrome victims die from respiratory failure before their sixth month, and many who survive that milestone eventually succumb to renal failure by age 3, clinical data show. However, normal chest development is possible for those with mild cases.
Bubba’s case was not mild, though—he constantly struggled to breathe, and that struggle often caused his heart to stop. “There were just so many things going wrong at once,” his mother Terasa recalled. “We were overwhelmed.”
So was Bubba. Predictably, his breathing worsened as he grew, leading to regular bouts of cardiac arrest and emergency life-saving CPR sessions. The sandy-haired tot also developed thoracic insufficiency syndrome (TIS), a rare condition typically related to congenital scoliosis. Fewer than 4,000 American children are born with the condition each year.
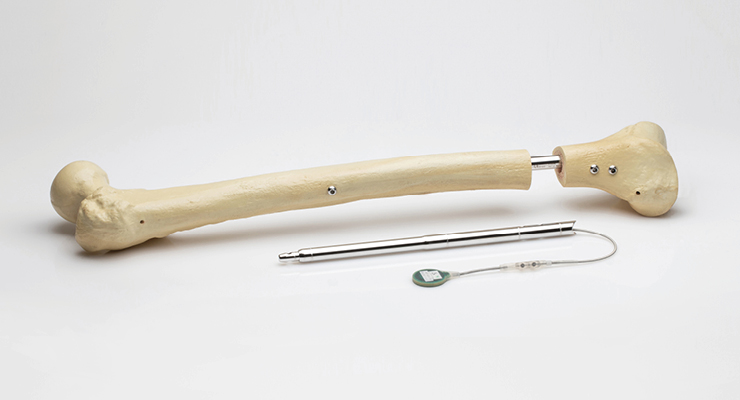
With time running out, Bubba’s doctors in Virginia transferred him to Children’s Hospital of Philadelphia for TIS treatment using a vertical expandable prosthetic titanium rib (VEPTR). Cleared by the U.S. Food and Drug Administration (FDA) under a Humanitarian Device Exemption in September 2004, the VEPTR is a curved metal rod with hooks on both ends that is surgically attached to a child’s ribs, spine, or pelvis. The rod has holes along its length so it can be adjusted as the patient grows.
The titanium rib helps straighten the pediatric spine and separate ribs, allowing for proper lung growth. It typically is implanted in infants as young as 6 months (like Bubba) and in teenagers until skeletal maturity, typically age 14 in girls and 16 in boys.
Surgeons first expanded Bubba’s rib cage two years ago in two separate (but related) procedures. He’s returned to Philadelphia every six months since then for VEPTR expansion surgery to help his lungs keep pace with his growth; and while he still faces various challenges—notably, feeding tube and ventilator dependency—Bubba can claim multiple small victories during his 28-month convalescence: some speech (“hey,” “Da-da”), independent movement (clapping, rolling over, attempting to chew his toes), and music preference (Michael Jackson). He is receiving physical therapy to sit unassisted and to stand with help, and above all else, no longer needs routine CPR.
After years of heartbreak, Bubba’s family finally has reason to hope for the future.
“...after Bubba had the VEPTR surgeries, his life—and ours—have changed so much,” Teresa said. “I’d love to see him off the vent, to talk and walk one day, to find something he enjoys doing with his life.”
Bubba is fortunate such opportunities exist. Before the VEPTR came along, children with TIS were victims of their genetic destinies, doomed by their misshapen skeletal structures to a lifetime of suffering.
The VEPTR, however, changed that fate, improving both longevity and life quality for TIS patients. Perhaps more importantly though, the device helped expose the flagrant disparity in orthopedic care between adults and children.
“Children deserve access to devices that are safe, effective, and made just for them. Yet many devices are not made with these considerations in mind, and some necessary devices are not made at all,” the late Robert M. Campbell Jr., M.D., VEPTR inventor, attested before a U.S. Senate committee in March 2007. “They are frequently denied access because there’s a relatively small market for pediatric devices, with little incentive for manufacturers to make them. I have been frustrated many times that the ‘shelves are bare’ when I need a modern device for [children’s] care. Physicians must commonly ‘jury-rig’ existing devices for children. The VEPTR was invented to replace such ‘jury-rigged’ devices.”
Clearly, Campbell’s invention succeeded in its mission: Since debuting more than 30 years ago, the VEPTR has become the standard of care worldwide for TIS treatment. Yet its success can largely be attributed to the very problem it was meant to address, and to some extent, Campbell’s tireless efforts to champion pediatric medical device development.
Besides helping identify, name, and define thoracic insufficiency syndrome, Campbell served for years as a scientific advisor and reviewer of research proposals for the National Organization for Rare Disorders. In addition, he was a main proponent of the 2007 Pediatric Medical Device Safety and Improvement Act, which established research priorities for pediatric devices and bolstered the FDA’s post-market surveillance of children’s products. The law also spawned the FDA’s Pediatric Device Consortia (PDC) grant program, an initiative designed to encourage and support pediatric device development among U.S. health systems and research centers.
“In recent years, the FDA has recognized that children have their own unique physiological needs and has taken major strides to help make a push to prioritize them,” said Nick A. Deeter, board chairman and CEO of WishBone Medical Inc., a pediatric orthopedic device firm based in Warsaw, Ind. “Pediatric orthopedics is still a very new segment within the industry; pediatric-focused orthopedic device companies have only existed in the last 15 years. This niche industry breeds developments to disrupt the status quo of using adult implants in children. Still to this day, adult products that are not cleared by the FDA for pediatric use are often altered on the spot in the O.R. to fit a child.”
There are many reasons for such impromptu modifications, the most obvious of which is size. Most adult orthopedic products are simply too big to be used inside a child. Case in point: Wishbone Medical makes a clavicle plate in two sizes, six-hole (74.5mm) and eight-hole (86.9mm), whereas Zimmer Biomet’s adult version ranges from 90mm to 140mm, depending on patient proportions.
Similarly, WishBone Medical’s anterior cervical plate is manufactured in one- or two-level lengths (20-32 mm and 34-52 mm respectively) with screw angulation up to 32 degrees, while NuVasive Inc. offers one- to five-level (adult) plate lengths with screw angulation up to 30 degrees.
The range of product sizes is different for children, too. Pediatric patients need a larger array of sizes to fit their growing bodies; the roughly 300 bones every baby is born with gradually is reduced to 206 by adulthood. Thus, younger patients will naturally require a larger pool of implants to repair their still maturing skeletal systems.
“There are really four factors involved in a pediatric device that adult products don’t have to deal with,” noted Matthew Oetgen, M.D., chief of Orthopaedic Surgery and Sports Medicine at Children’s National Hospital in Washington, D.C. “The first is size. They have to fit in kids. In adults you have to deal with size too, but there are relatively small differences within genders, going from small to large implants. In designing an [orthopedic] implant for children, you have to think about the range of sizes—from infant to skeletal maturity. That range is so much greater for kids than it is for adults, and that can be a barrier to [product] development.”
Bone structure is another potential barrier. There are significant anatomical differences in the bones of adults and children: A fully-developed adult bone is comprised of two regions, diaphysis and metaphysis, but a developing child’s bone has four areas—diaphysis, metaphysis, epiphysis, and physis.
The latter two regions are the most troublesome for pediatric device developers. The epiphysis is the rounded end of a long bone, yet its composition changes during a child’s development; it actually starts out as cartilage (at birth) and contributes to the joint, but eventually hardens and attaches to the bone at puberty. The epiphysis is a common site of injury in kids, with the area accounting for 6-15 percent of long bone fractures in children under 16, data show.
A more frequent bone injury site is the physis—a.k.a., growth plate—a translucent, cartilaginous disc separating the end and neck portions of long bones. Growth plate injuries are roughly twice as likely to occur than epiphysis site trauma, comprising 15-30 percent of all bone-related injuries. Damage to this area requires prompt attention, as the growth plate helps determine the future length and shape of mature bone. Improperly treated growth plate fractures can cause bones to heal crookedly or unevenly from their opposite counterparts.
“The shape of a pediatric bone is completely different than an adult one. In addition to size, the angles and presence of the growth plates must be taken into account,” explained Paul Gonsalves, global president, Orthofix Orthopedics. “For example, a standard surgical treatment for an adult fracture is the use of a rigid intramedullary rod. In a pediatric patient, this approach may affect the functionality of the growth plates. The shape of the rod must be designed to avoid the growth plates in order to minimize potential impact on the child’s growth. Additionally, the growth plates and the reduced weight of pediatric patients in general can be used as an advantage in developing specific innovative treatments that are not possible for adults. In 2004, Orthofix introduced the eight-Plate Guided Growth System, a minimally invasive device to correct knee and ankle deformities by ‘modulating’ the growth plate in pediatric patients. This innovation completely changed the approach to a common pediatric condition. Now the use of the eight-Plate system has become a standard of care.”
Orthofix’s eight-Plate system is a figure-eight-shaped device that corrects limb misalignment through natural growth. The paper clip-sized product is attached to one side of a bone’s physis to restrict growth, leaving the opposite side free to expand. As the unrestricted side grows, the screws fastened to the plated side diverge, effectively serving as a hinge, thus allowing the bone to realign.
The eight-Plate system is one of several pediatric deformity-correcting solutions Orthofix offers under its JuniOrtho product brand. Other devices include the Paediatric LRS system, AGILE Nail, TL Hex TruLok Hexapod System, and the JuniOrtho Plating System. The latter solution won FDA 510(k) clearance and European CE mark approval last July for treating advanced lower extremities deformities and trauma reconstruction.
The JuniOrtho System features pre-operative planning software that streamlines implant selection before a surgical procedure, enabling clinicians to visualize the implant and accurately plan the surgical position. The tool helps physicians choose an implant size that best fits the patient’s body.
Manufactured in various plate sizes and lengths, the JuniOrtho Plating System accommodates both locking and non-locking screws that correspond to the appropriate plate size. Additionally, the system incorporates sterile implants and single-use tools to reduce contamination risk and optimize procedural efficiency.
Orthofix further augmented its pediatric deformity correction portfolio this past winter with the U.S. and European market launch of the FITBONE intramedullary lengthening system, a solution it acquired from German developer Wittenstein SE in February 2020. The FITBONE intramedullary lengthening nail is a fully implantable system for correcting leg length and both femur and tibia deformities. Implanted through a minimally invasive procedure, the system consists of the intramedullary nail, a subcutaneously implanted receiver, and an external control set that lets patients manage the discretion phase at home.
Preoperative planning enables the FITBONE system to achieve axial and torsional bone alignment intraoperatively. The device has been used in more than 3,500 realignment cases in 15 countries since its development.
“One additional area that is extremely important is the preoperative planning phase for pediatric patients. Our recently developed OrthoNet digital platforms enable surgeons to accurately plan the surgery and position templating devices on the radiological images in order to visualize the implant in relation to the anatomy,” Gonsalves told Orthopedic Design & Technology. “This software is designed to streamline the selection of the precise size of device to ensure the best fit and optimal positioning for the patient’s body prior to the surgical procedure. In addition to new software technologies, remote patient management through telemedicine or dedicated patient apps and custom-made implants are becoming more and more important. These technologies offer pediatric patients the best services in order to achieve the best outcomes. At Orthofix, we believe this will be one of the biggest differentiators among companies in the future and that is why we are focusing our research in this area.”
Indeed, preoperative planning and customized implants increasingly are being incorporated into pediatric product offerings, though they still (comparatively) lag behind their adult counterparts. Zimmer Biomet, for instance, provides preoperative planning tools for its knee and shoulder implants but not for its idiopathic scoliosis treatment systems.
WishBone Medical, conversely, developed proprietary planning software for its FDA-cleared Smart Correction External Fixation System, a hexapod external fixator for stabilizing and maintaining alignment of complicated fractures, soft tissues, and/or congenital deformity repairs. The System is designed to treat open and closed fractures, arthrodesis, and long bone pseudoarthrosis as well as limb lengthening, deformity, and angular correction, boney or soft tissue defect correction, and malunions.
WishBone Medical rival OrthoPediatrics Corp. provides pre-surgical deformity correction planning too. The Warsaw, Ind.-based company’s external fixation and hexapod system, Orthex, consists of rings, struts, implants, hardware accessories, and software for correcting deformities in the upper and lower extremities. The patented “Point & Click” software eliminates the need for surgeons to measure any mounting parameters; in a step-by-step process, users complete Center of Rotation of Angulation (CORA) deformity planning to determine (via a computer program) the necessary angles and translations to align the affected bones.
OrthoPediatrics also offers preoperative planning with its RESPONSE Spine System, which frequently uses FIREFLY Pedicle Screw Navigation Guides from Englewood, Colo.-based medtech incubator Mighty Oak Medical. OrthoPediatrics distributes the FIREFLY technology in U.S. children’s hospitals.
FIREFLY uses artificial intelligence-driven preoperative planning for optimizing pedicle screw size and trajectory during complex spinal surgeries. The FIREFLY technology incorporates 3D printing and computed tomography data to create navigation guides for each vertebral level and a biocompatible bone model that are adapted to a patient’s anatomy with 99.7 percent accuracy, according to the company. Screw trajectories determined by the presurgical planning process are integrated into the virtual model for real-time surgeon reference intraoperatively. The technology’s benefits include screw placement accuracy, improved OR efficiency, reduced intraoperative radiation, and fewer revision surgeries.
“With the advancement of instruments and implants specifically for kids, surgeons today have more options to surgically treat pediatric trauma, limb deformities, and scoliosis, which enables faster growth than the adult market,” said OrthoPediatrics president David Bailey and COO/CFO Fred Hite. “The challenges facing orthopedic surgeons are extremely complicated and wide-ranging; adult products often do not work for these children. We focus on the largest procedure areas or where we feel we can make the biggest impact on the lives of children. So far this has been in the areas of pediatric trauma, limb deformity, and scoliosis, and we still have work to do to meet unmet needs in these focus areas.”
Maybe so, but the product market in these focus areas has become considerably more diversified in the last decade or two. OrthoPediatrics has developed a plethora of devices for treating trauma, deformities, and scoliosis, including: the DFOS system, Locking Cannulated Blade plate system (with instruments), Locking Proximal Femur plate system, Pediatric Nailing Platform | Femur, PediFoot Deformity Correction System, PediLoc Tibia Locking Plate System, Pedi Nail Intramedullary Nail, Pedi Plates, and Trans-Physeal Locking Screws, among others.
NuVasive expanded its line of pediatric deformity-correcting implants last year with the global launch of the Reline 3D system for adolescent idiopathic scoliosis treatment, and the limited release of its Precice Plate for limb lengthening and reconstruction procedures.
The Reline 3D system deploys intricate surgical techniques to address three-dimensional spine deformities. The system consolidates current deformity techniques engaging multi-step and single-lane correction into one integrated procedure to help address current OR inefficiencies.
Reline 3D is considered an extension of NuVasive’s fixation portfolio of Reline systems, including Reline Open, Reline MAS, Reline Trauma, and Reline Small Stature.
The company’s Precice Plate aims to address pediatric applications or patients with small anatomies who cannot accommodate a lengthening device that traditionally is implanted within the bone. With locking plate technology, the plate affixes to the outside of a bone to avoid potential damage to the physis. A programmable external remote controller magnetically couples with the plate through the skin to perform micro adjustments.
Over several months from home, the patient follows a custom lengthening schedule to gradually distract the plate, stretching newly-forming bone to a targeted length. once the desired length is achieved, the plate is removed.
“By expanding the application of our remote-controlled implants, NSO [NuVasive Specialized Orthopedics] has delivered a treatment option that elevates the standard of care for pediatric patients with limb length discrepancy,” Rob Martin, general manager of NSO, said at the Precice Plate’s September 2020 unveiling. “With a less invasive treatment protocol and at-home precision lengthening, the availability of the Precice Plate directly reflects NuVasive’s commitment to grow its proprietary technology in previously underserved markets and deliver better patient outcomes.”
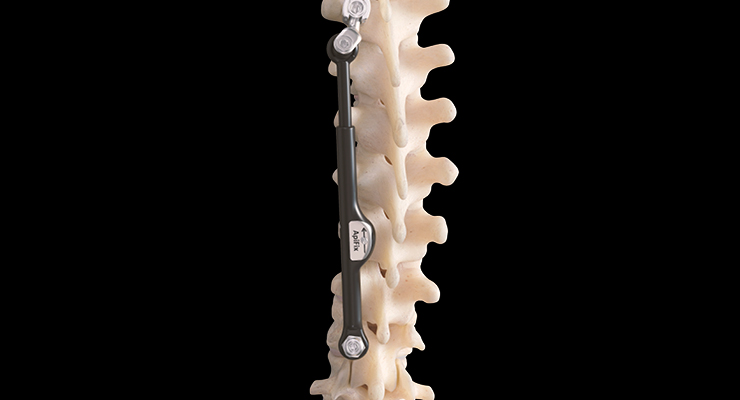
Stryker Corp. has exhibited a similar commitment in the pediatric spine sector, working with the Scoliosis Research Society and the Pediatric Orthopedic Society of North America to identify underserved pockets of the overall pediatric market and develop corresponding products. Consequently, the company has devised treatments for adolescent idiopathic scoliosis, neuromuscular scoliosis, congenital scoliosis, and early-onset scoliosis.
“We have developed a portfolio that can address the skeletal needs of the pediatric patient. Our pediatric deformity portfolio is comprised of established products such as Nile, Mesa, Xia 3, and Xia 4.5,” explained Eric Major, president of Stryker’s Spine Division. “The Nile Alternative Fixation and Nile Proximal Fixation Spinal Systems, which include implants intended for use in orthopedic surgery to provide temporary stabilization as a bone anchor during the development of solid bony fusion and to aid in the repairs of bone fractures. The Mesa, Xia 3, and Xia 4.5 Spinal Systems are indicated as a adjunct to fusion to treat idiopathic scoliosis, congenital scoliosis, and neuromuscular scoliosis when used for posterior non-cervical fixation in pediatric patients. We’ve also received FDA clearance for our Xia Growth Rod Conversion set for patients with potential for additional spinal growth under 10 years of age who require surgical treatment to obtain and maintain correction of severe spinal deformities. There was a large unmet need in the pediatric orthopedic device space for years, but we are starting to see more products available for surgeons.”
Many of those products, however, tend to favor the pediatric population’s oldest patients. FDA data show that device approvals for kids reached a 10-year high in 2017, but nearly 40 percent of those authorizations went to products indicated for individuals ages 18 and older (the agency defines “pediatric” patients as those between birth and 21 years of age).
Clearly, the medtech market for kids continues to languish in the shadow of adults. And perhaps it always will: The barriers to innovation in this sector are inherent and seemingly insurmountable—complex biologies, callow mentalities, and an overall small patient population (both in stature and numbers).
Nevertheless, the market is slowly demanding more attention. “The market has already come so far,” Oetgen said. “The National Capital Consortium for Pediatric Device Innovation (NCC-PDI) has loosened the innovation process and has helped companies enter this field. The pediatric device market has long been neglected but now it’s attracting some support. I’m really quite high on the future of this market. We’re starting to see paid dividends and new innovations commercialized. We’re seeing better devices and better outcomes for kids, and that’s what we’re all in this game for. Kids are not just little adults.”
Born six weeks prematurely nearly three years ago, the neonate greeted this life without the ability to see or properly breathe. Doctors detected trouble with the boy’s bones while in-utero, but neither the source nor the extent of that trouble was evident until his birth.
Besides producing abnormally short arms and legs, Bubba’s gene mutation also beget an extremely narrow rib cage, which affected the boy’s respiration. As he grew, other genetic deficiencies emerged, most notably blindness and a weak heart.
Within three months, doctors diagnosed Bubba with Jeune syndrome, a form of congenital dwarfism characterized by a bell-shaped chest cavity, short ribs, and shortened arm and leg bones. Other skeletal abnormalities associated with the disorder include extra fingers and toes (polydactyly), unusually shaped clavicles and pelvic bones, and cone-shaped ends of the long arm and leg bones.
Jeune’s most distinctive feature can also be its most lethal: Left untreated, the condition (also known as asphyxiating thoracic dystrophy) dooms most babies to an abbreviated, tortuous lifespan as they slowly and progressively suffocate. Half of all Jeune syndrome victims die from respiratory failure before their sixth month, and many who survive that milestone eventually succumb to renal failure by age 3, clinical data show. However, normal chest development is possible for those with mild cases.
Bubba’s case was not mild, though—he constantly struggled to breathe, and that struggle often caused his heart to stop. “There were just so many things going wrong at once,” his mother Terasa recalled. “We were overwhelmed.”
So was Bubba. Predictably, his breathing worsened as he grew, leading to regular bouts of cardiac arrest and emergency life-saving CPR sessions. The sandy-haired tot also developed thoracic insufficiency syndrome (TIS), a rare condition typically related to congenital scoliosis. Fewer than 4,000 American children are born with the condition each year.
With time running out, Bubba’s doctors in Virginia transferred him to Children’s Hospital of Philadelphia for TIS treatment using a vertical expandable prosthetic titanium rib (VEPTR). Cleared by the U.S. Food and Drug Administration (FDA) under a Humanitarian Device Exemption in September 2004, the VEPTR is a curved metal rod with hooks on both ends that is surgically attached to a child’s ribs, spine, or pelvis. The rod has holes along its length so it can be adjusted as the patient grows.
The titanium rib helps straighten the pediatric spine and separate ribs, allowing for proper lung growth. It typically is implanted in infants as young as 6 months (like Bubba) and in teenagers until skeletal maturity, typically age 14 in girls and 16 in boys.
Surgeons first expanded Bubba’s rib cage two years ago in two separate (but related) procedures. He’s returned to Philadelphia every six months since then for VEPTR expansion surgery to help his lungs keep pace with his growth; and while he still faces various challenges—notably, feeding tube and ventilator dependency—Bubba can claim multiple small victories during his 28-month convalescence: some speech (“hey,” “Da-da”), independent movement (clapping, rolling over, attempting to chew his toes), and music preference (Michael Jackson). He is receiving physical therapy to sit unassisted and to stand with help, and above all else, no longer needs routine CPR.
After years of heartbreak, Bubba’s family finally has reason to hope for the future.
“...after Bubba had the VEPTR surgeries, his life—and ours—have changed so much,” Teresa said. “I’d love to see him off the vent, to talk and walk one day, to find something he enjoys doing with his life.”
Bubba is fortunate such opportunities exist. Before the VEPTR came along, children with TIS were victims of their genetic destinies, doomed by their misshapen skeletal structures to a lifetime of suffering.
The VEPTR, however, changed that fate, improving both longevity and life quality for TIS patients. Perhaps more importantly though, the device helped expose the flagrant disparity in orthopedic care between adults and children.
“Children deserve access to devices that are safe, effective, and made just for them. Yet many devices are not made with these considerations in mind, and some necessary devices are not made at all,” the late Robert M. Campbell Jr., M.D., VEPTR inventor, attested before a U.S. Senate committee in March 2007. “They are frequently denied access because there’s a relatively small market for pediatric devices, with little incentive for manufacturers to make them. I have been frustrated many times that the ‘shelves are bare’ when I need a modern device for [children’s] care. Physicians must commonly ‘jury-rig’ existing devices for children. The VEPTR was invented to replace such ‘jury-rigged’ devices.”
Clearly, Campbell’s invention succeeded in its mission: Since debuting more than 30 years ago, the VEPTR has become the standard of care worldwide for TIS treatment. Yet its success can largely be attributed to the very problem it was meant to address, and to some extent, Campbell’s tireless efforts to champion pediatric medical device development.
Besides helping identify, name, and define thoracic insufficiency syndrome, Campbell served for years as a scientific advisor and reviewer of research proposals for the National Organization for Rare Disorders. In addition, he was a main proponent of the 2007 Pediatric Medical Device Safety and Improvement Act, which established research priorities for pediatric devices and bolstered the FDA’s post-market surveillance of children’s products. The law also spawned the FDA’s Pediatric Device Consortia (PDC) grant program, an initiative designed to encourage and support pediatric device development among U.S. health systems and research centers.
“In recent years, the FDA has recognized that children have their own unique physiological needs and has taken major strides to help make a push to prioritize them,” said Nick A. Deeter, board chairman and CEO of WishBone Medical Inc., a pediatric orthopedic device firm based in Warsaw, Ind. “Pediatric orthopedics is still a very new segment within the industry; pediatric-focused orthopedic device companies have only existed in the last 15 years. This niche industry breeds developments to disrupt the status quo of using adult implants in children. Still to this day, adult products that are not cleared by the FDA for pediatric use are often altered on the spot in the O.R. to fit a child.”
There are many reasons for such impromptu modifications, the most obvious of which is size. Most adult orthopedic products are simply too big to be used inside a child. Case in point: Wishbone Medical makes a clavicle plate in two sizes, six-hole (74.5mm) and eight-hole (86.9mm), whereas Zimmer Biomet’s adult version ranges from 90mm to 140mm, depending on patient proportions.
Similarly, WishBone Medical’s anterior cervical plate is manufactured in one- or two-level lengths (20-32 mm and 34-52 mm respectively) with screw angulation up to 32 degrees, while NuVasive Inc. offers one- to five-level (adult) plate lengths with screw angulation up to 30 degrees.
The range of product sizes is different for children, too. Pediatric patients need a larger array of sizes to fit their growing bodies; the roughly 300 bones every baby is born with gradually is reduced to 206 by adulthood. Thus, younger patients will naturally require a larger pool of implants to repair their still maturing skeletal systems.
“There are really four factors involved in a pediatric device that adult products don’t have to deal with,” noted Matthew Oetgen, M.D., chief of Orthopaedic Surgery and Sports Medicine at Children’s National Hospital in Washington, D.C. “The first is size. They have to fit in kids. In adults you have to deal with size too, but there are relatively small differences within genders, going from small to large implants. In designing an [orthopedic] implant for children, you have to think about the range of sizes—from infant to skeletal maturity. That range is so much greater for kids than it is for adults, and that can be a barrier to [product] development.”
Bone structure is another potential barrier. There are significant anatomical differences in the bones of adults and children: A fully-developed adult bone is comprised of two regions, diaphysis and metaphysis, but a developing child’s bone has four areas—diaphysis, metaphysis, epiphysis, and physis.
The latter two regions are the most troublesome for pediatric device developers. The epiphysis is the rounded end of a long bone, yet its composition changes during a child’s development; it actually starts out as cartilage (at birth) and contributes to the joint, but eventually hardens and attaches to the bone at puberty. The epiphysis is a common site of injury in kids, with the area accounting for 6-15 percent of long bone fractures in children under 16, data show.
A more frequent bone injury site is the physis—a.k.a., growth plate—a translucent, cartilaginous disc separating the end and neck portions of long bones. Growth plate injuries are roughly twice as likely to occur than epiphysis site trauma, comprising 15-30 percent of all bone-related injuries. Damage to this area requires prompt attention, as the growth plate helps determine the future length and shape of mature bone. Improperly treated growth plate fractures can cause bones to heal crookedly or unevenly from their opposite counterparts.
“The shape of a pediatric bone is completely different than an adult one. In addition to size, the angles and presence of the growth plates must be taken into account,” explained Paul Gonsalves, global president, Orthofix Orthopedics. “For example, a standard surgical treatment for an adult fracture is the use of a rigid intramedullary rod. In a pediatric patient, this approach may affect the functionality of the growth plates. The shape of the rod must be designed to avoid the growth plates in order to minimize potential impact on the child’s growth. Additionally, the growth plates and the reduced weight of pediatric patients in general can be used as an advantage in developing specific innovative treatments that are not possible for adults. In 2004, Orthofix introduced the eight-Plate Guided Growth System, a minimally invasive device to correct knee and ankle deformities by ‘modulating’ the growth plate in pediatric patients. This innovation completely changed the approach to a common pediatric condition. Now the use of the eight-Plate system has become a standard of care.”
Orthofix’s eight-Plate system is a figure-eight-shaped device that corrects limb misalignment through natural growth. The paper clip-sized product is attached to one side of a bone’s physis to restrict growth, leaving the opposite side free to expand. As the unrestricted side grows, the screws fastened to the plated side diverge, effectively serving as a hinge, thus allowing the bone to realign.
The eight-Plate system is one of several pediatric deformity-correcting solutions Orthofix offers under its JuniOrtho product brand. Other devices include the Paediatric LRS system, AGILE Nail, TL Hex TruLok Hexapod System, and the JuniOrtho Plating System. The latter solution won FDA 510(k) clearance and European CE mark approval last July for treating advanced lower extremities deformities and trauma reconstruction.
The JuniOrtho System features pre-operative planning software that streamlines implant selection before a surgical procedure, enabling clinicians to visualize the implant and accurately plan the surgical position. The tool helps physicians choose an implant size that best fits the patient’s body.
Manufactured in various plate sizes and lengths, the JuniOrtho Plating System accommodates both locking and non-locking screws that correspond to the appropriate plate size. Additionally, the system incorporates sterile implants and single-use tools to reduce contamination risk and optimize procedural efficiency.
Orthofix further augmented its pediatric deformity correction portfolio this past winter with the U.S. and European market launch of the FITBONE intramedullary lengthening system, a solution it acquired from German developer Wittenstein SE in February 2020. The FITBONE intramedullary lengthening nail is a fully implantable system for correcting leg length and both femur and tibia deformities. Implanted through a minimally invasive procedure, the system consists of the intramedullary nail, a subcutaneously implanted receiver, and an external control set that lets patients manage the discretion phase at home.
Preoperative planning enables the FITBONE system to achieve axial and torsional bone alignment intraoperatively. The device has been used in more than 3,500 realignment cases in 15 countries since its development.
“One additional area that is extremely important is the preoperative planning phase for pediatric patients. Our recently developed OrthoNet digital platforms enable surgeons to accurately plan the surgery and position templating devices on the radiological images in order to visualize the implant in relation to the anatomy,” Gonsalves told Orthopedic Design & Technology. “This software is designed to streamline the selection of the precise size of device to ensure the best fit and optimal positioning for the patient’s body prior to the surgical procedure. In addition to new software technologies, remote patient management through telemedicine or dedicated patient apps and custom-made implants are becoming more and more important. These technologies offer pediatric patients the best services in order to achieve the best outcomes. At Orthofix, we believe this will be one of the biggest differentiators among companies in the future and that is why we are focusing our research in this area.”
Indeed, preoperative planning and customized implants increasingly are being incorporated into pediatric product offerings, though they still (comparatively) lag behind their adult counterparts. Zimmer Biomet, for instance, provides preoperative planning tools for its knee and shoulder implants but not for its idiopathic scoliosis treatment systems.
WishBone Medical, conversely, developed proprietary planning software for its FDA-cleared Smart Correction External Fixation System, a hexapod external fixator for stabilizing and maintaining alignment of complicated fractures, soft tissues, and/or congenital deformity repairs. The System is designed to treat open and closed fractures, arthrodesis, and long bone pseudoarthrosis as well as limb lengthening, deformity, and angular correction, boney or soft tissue defect correction, and malunions.
WishBone Medical rival OrthoPediatrics Corp. provides pre-surgical deformity correction planning too. The Warsaw, Ind.-based company’s external fixation and hexapod system, Orthex, consists of rings, struts, implants, hardware accessories, and software for correcting deformities in the upper and lower extremities. The patented “Point & Click” software eliminates the need for surgeons to measure any mounting parameters; in a step-by-step process, users complete Center of Rotation of Angulation (CORA) deformity planning to determine (via a computer program) the necessary angles and translations to align the affected bones.
OrthoPediatrics also offers preoperative planning with its RESPONSE Spine System, which frequently uses FIREFLY Pedicle Screw Navigation Guides from Englewood, Colo.-based medtech incubator Mighty Oak Medical. OrthoPediatrics distributes the FIREFLY technology in U.S. children’s hospitals.
FIREFLY uses artificial intelligence-driven preoperative planning for optimizing pedicle screw size and trajectory during complex spinal surgeries. The FIREFLY technology incorporates 3D printing and computed tomography data to create navigation guides for each vertebral level and a biocompatible bone model that are adapted to a patient’s anatomy with 99.7 percent accuracy, according to the company. Screw trajectories determined by the presurgical planning process are integrated into the virtual model for real-time surgeon reference intraoperatively. The technology’s benefits include screw placement accuracy, improved OR efficiency, reduced intraoperative radiation, and fewer revision surgeries.
“With the advancement of instruments and implants specifically for kids, surgeons today have more options to surgically treat pediatric trauma, limb deformities, and scoliosis, which enables faster growth than the adult market,” said OrthoPediatrics president David Bailey and COO/CFO Fred Hite. “The challenges facing orthopedic surgeons are extremely complicated and wide-ranging; adult products often do not work for these children. We focus on the largest procedure areas or where we feel we can make the biggest impact on the lives of children. So far this has been in the areas of pediatric trauma, limb deformity, and scoliosis, and we still have work to do to meet unmet needs in these focus areas.”
Maybe so, but the product market in these focus areas has become considerably more diversified in the last decade or two. OrthoPediatrics has developed a plethora of devices for treating trauma, deformities, and scoliosis, including: the DFOS system, Locking Cannulated Blade plate system (with instruments), Locking Proximal Femur plate system, Pediatric Nailing Platform | Femur, PediFoot Deformity Correction System, PediLoc Tibia Locking Plate System, Pedi Nail Intramedullary Nail, Pedi Plates, and Trans-Physeal Locking Screws, among others.
NuVasive expanded its line of pediatric deformity-correcting implants last year with the global launch of the Reline 3D system for adolescent idiopathic scoliosis treatment, and the limited release of its Precice Plate for limb lengthening and reconstruction procedures.
The Reline 3D system deploys intricate surgical techniques to address three-dimensional spine deformities. The system consolidates current deformity techniques engaging multi-step and single-lane correction into one integrated procedure to help address current OR inefficiencies.
Reline 3D is considered an extension of NuVasive’s fixation portfolio of Reline systems, including Reline Open, Reline MAS, Reline Trauma, and Reline Small Stature.
The company’s Precice Plate aims to address pediatric applications or patients with small anatomies who cannot accommodate a lengthening device that traditionally is implanted within the bone. With locking plate technology, the plate affixes to the outside of a bone to avoid potential damage to the physis. A programmable external remote controller magnetically couples with the plate through the skin to perform micro adjustments.
Over several months from home, the patient follows a custom lengthening schedule to gradually distract the plate, stretching newly-forming bone to a targeted length. once the desired length is achieved, the plate is removed.
“By expanding the application of our remote-controlled implants, NSO [NuVasive Specialized Orthopedics] has delivered a treatment option that elevates the standard of care for pediatric patients with limb length discrepancy,” Rob Martin, general manager of NSO, said at the Precice Plate’s September 2020 unveiling. “With a less invasive treatment protocol and at-home precision lengthening, the availability of the Precice Plate directly reflects NuVasive’s commitment to grow its proprietary technology in previously underserved markets and deliver better patient outcomes.”
Stryker Corp. has exhibited a similar commitment in the pediatric spine sector, working with the Scoliosis Research Society and the Pediatric Orthopedic Society of North America to identify underserved pockets of the overall pediatric market and develop corresponding products. Consequently, the company has devised treatments for adolescent idiopathic scoliosis, neuromuscular scoliosis, congenital scoliosis, and early-onset scoliosis.
“We have developed a portfolio that can address the skeletal needs of the pediatric patient. Our pediatric deformity portfolio is comprised of established products such as Nile, Mesa, Xia 3, and Xia 4.5,” explained Eric Major, president of Stryker’s Spine Division. “The Nile Alternative Fixation and Nile Proximal Fixation Spinal Systems, which include implants intended for use in orthopedic surgery to provide temporary stabilization as a bone anchor during the development of solid bony fusion and to aid in the repairs of bone fractures. The Mesa, Xia 3, and Xia 4.5 Spinal Systems are indicated as a adjunct to fusion to treat idiopathic scoliosis, congenital scoliosis, and neuromuscular scoliosis when used for posterior non-cervical fixation in pediatric patients. We’ve also received FDA clearance for our Xia Growth Rod Conversion set for patients with potential for additional spinal growth under 10 years of age who require surgical treatment to obtain and maintain correction of severe spinal deformities. There was a large unmet need in the pediatric orthopedic device space for years, but we are starting to see more products available for surgeons.”
Many of those products, however, tend to favor the pediatric population’s oldest patients. FDA data show that device approvals for kids reached a 10-year high in 2017, but nearly 40 percent of those authorizations went to products indicated for individuals ages 18 and older (the agency defines “pediatric” patients as those between birth and 21 years of age).
Clearly, the medtech market for kids continues to languish in the shadow of adults. And perhaps it always will: The barriers to innovation in this sector are inherent and seemingly insurmountable—complex biologies, callow mentalities, and an overall small patient population (both in stature and numbers).
Nevertheless, the market is slowly demanding more attention. “The market has already come so far,” Oetgen said. “The National Capital Consortium for Pediatric Device Innovation (NCC-PDI) has loosened the innovation process and has helped companies enter this field. The pediatric device market has long been neglected but now it’s attracting some support. I’m really quite high on the future of this market. We’re starting to see paid dividends and new innovations commercialized. We’re seeing better devices and better outcomes for kids, and that’s what we’re all in this game for. Kids are not just little adults.”