Brian Ingraham, Senior R&D Director, Amcor Healthcare11.17.21
Although the orthopedic device market was affected in the short term by the pandemic, due to reduced road accidents and associated injuries, according to a report by Mordor Intelligence, it is expected to reach $51.4 billion in value by 2026 at a CAGR of approximately 4.9 percent.1 With this market growth comes the need for packaging that not only meets stringent FDA requirements, but also protects patients who rely on these critical devices.
At times, packaging orthopedic devices such as implants can prove to be challenging because of their size and sterility requirements. Thankfully, packaging materials can perform a vital service in protecting the device inside. Sealing technology can make a difference for the package in order to handle heavier and bulkier orthopedic items. Stronger seals in particular are necessary to ensure this safety, in addition to exhibiting optimum porosity for EtO sterilization. Although sourcing these materials may seem secondary to the materials of the devices themselves, without proper packaging, orthopedic devices are at risk.
Greater Device Protection
For orthopedic devices (especially implants), it is essential the packaging does not compromise the efficacy of the product and potentially endanger a patient. Seal strength plays a vital role in this protection—the strength of the seal can mean the difference between a compromised device or a secure one. Heat-seal coatings such as CR27 have been used to meet this critical need for package integrity. However, depending on the applications, orthopedic device manufactures can consider enhanced formulations that can increase seal strength.
Amcor’s ACT2100, for example, exceeds the 1 lb./inch strength across a broad range of sealing temperatures. Further, the coating is 25 percent stronger than other competitive coating technologies. For specialized orthopedic devices that require larger or heavier packages, greater seal strength is increasingly important since they are subject to higher sterilant gas volumes, which can place greater stress on seals within the packaging.
Moreover, manufacturers can face challenges with the application process of a seal. If a heat-seal coating cannot be applied consistently through contact coating processes, for instance, it could lead to integrity issues. Alternatively, an air-knife application of heat seals provides a more consistent coating for any surface topography.
Keeping Quality in Check
The packaging production process can run into unexpected issues if manufacturers do not ensure the necessary safety standards and quality controls at every step. Heat-seal coatings are no exception. Thus, it is critical for manufacturers to guarantee any suppliers are utilizing state-of-the-art mixing technologies in order to certify the correct particle size for the heat-seal coating.
Particle size is a key component to the seal’s breathability and porosity through sterilization, regardless of the intensity. With improved porosity, manufacturers can achieve more efficient EtO sterilization cycles and avoid any potential issues with residuals, as opposed to a less breathable packaging.
Further, quality control can be enforced with camera inspections of packaging materials throughout the manufacturing process in order to reduce the risk of contaminants or defects for customers. With in-line vision systems, manufacturers can more efficiently detect defects before the material is delivered.
Taking a Closer Look
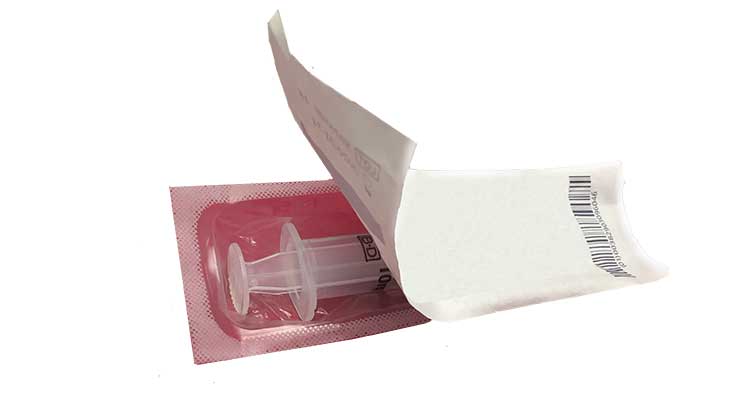
When it comes to medical device packaging, it’s vital that practitioners can easily determine with just a visual inspection whether or not the device has been compromised. By using a heat-seal coating with substantial seal evidence, medical practitioners will be able to easily confirm a package’s integrity. Heat-seal coatings that turn yellow or get brittle over time risk causing a lack of confidence in the package integrity.
Passing the “human eye” test is just as important as camera inspections for heat-seal coatings. Not only does clear peel evidence and a clean peel grant healthcare workers the ability to easily detect if the device is sterile and uncompromised, that peel also minimizes the risk of fiber tears while opening the package.
Ensure the Safety of Orthopedic Devices
It is critical for orthopedic device manufacturers to ensure their products remain sterile and uncompromised so they can protect patients. Heat-seal coatings are a key aspect of ensuring orthopedic medical packaging consistently meets the sterility requirements for these life-saving products.
With greater strength, improved porosity throughout sterilization, and consistency, high-quality seals can provide greater assurance throughout the lifecycle of orthopedic devices.
Reference
Brian Ingraham is senior director of R&D healthcare at Amcor Healthcare. He has 26 years of experience in the flexible packaging field, 19 of which have been with Amcor with increasing responsibility and leadership roles across pharmaceuticals, medical, food, home and personal care, and technical specialties markets. Ingraham has four patents and five FPA Innovation awards, where he was the lead developer. He received his BS in chemical engineering at the University of Wisconsin Madison and his MS in engineer management at Milwaukee School of Engineering.
At times, packaging orthopedic devices such as implants can prove to be challenging because of their size and sterility requirements. Thankfully, packaging materials can perform a vital service in protecting the device inside. Sealing technology can make a difference for the package in order to handle heavier and bulkier orthopedic items. Stronger seals in particular are necessary to ensure this safety, in addition to exhibiting optimum porosity for EtO sterilization. Although sourcing these materials may seem secondary to the materials of the devices themselves, without proper packaging, orthopedic devices are at risk.
Greater Device Protection
For orthopedic devices (especially implants), it is essential the packaging does not compromise the efficacy of the product and potentially endanger a patient. Seal strength plays a vital role in this protection—the strength of the seal can mean the difference between a compromised device or a secure one. Heat-seal coatings such as CR27 have been used to meet this critical need for package integrity. However, depending on the applications, orthopedic device manufactures can consider enhanced formulations that can increase seal strength.
Amcor’s ACT2100, for example, exceeds the 1 lb./inch strength across a broad range of sealing temperatures. Further, the coating is 25 percent stronger than other competitive coating technologies. For specialized orthopedic devices that require larger or heavier packages, greater seal strength is increasingly important since they are subject to higher sterilant gas volumes, which can place greater stress on seals within the packaging.
Moreover, manufacturers can face challenges with the application process of a seal. If a heat-seal coating cannot be applied consistently through contact coating processes, for instance, it could lead to integrity issues. Alternatively, an air-knife application of heat seals provides a more consistent coating for any surface topography.
Keeping Quality in Check
The packaging production process can run into unexpected issues if manufacturers do not ensure the necessary safety standards and quality controls at every step. Heat-seal coatings are no exception. Thus, it is critical for manufacturers to guarantee any suppliers are utilizing state-of-the-art mixing technologies in order to certify the correct particle size for the heat-seal coating.
Particle size is a key component to the seal’s breathability and porosity through sterilization, regardless of the intensity. With improved porosity, manufacturers can achieve more efficient EtO sterilization cycles and avoid any potential issues with residuals, as opposed to a less breathable packaging.
Further, quality control can be enforced with camera inspections of packaging materials throughout the manufacturing process in order to reduce the risk of contaminants or defects for customers. With in-line vision systems, manufacturers can more efficiently detect defects before the material is delivered.
Taking a Closer Look
When it comes to medical device packaging, it’s vital that practitioners can easily determine with just a visual inspection whether or not the device has been compromised. By using a heat-seal coating with substantial seal evidence, medical practitioners will be able to easily confirm a package’s integrity. Heat-seal coatings that turn yellow or get brittle over time risk causing a lack of confidence in the package integrity.
Passing the “human eye” test is just as important as camera inspections for heat-seal coatings. Not only does clear peel evidence and a clean peel grant healthcare workers the ability to easily detect if the device is sterile and uncompromised, that peel also minimizes the risk of fiber tears while opening the package.
Ensure the Safety of Orthopedic Devices
It is critical for orthopedic device manufacturers to ensure their products remain sterile and uncompromised so they can protect patients. Heat-seal coatings are a key aspect of ensuring orthopedic medical packaging consistently meets the sterility requirements for these life-saving products.
With greater strength, improved porosity throughout sterilization, and consistency, high-quality seals can provide greater assurance throughout the lifecycle of orthopedic devices.
Reference
Brian Ingraham is senior director of R&D healthcare at Amcor Healthcare. He has 26 years of experience in the flexible packaging field, 19 of which have been with Amcor with increasing responsibility and leadership roles across pharmaceuticals, medical, food, home and personal care, and technical specialties markets. Ingraham has four patents and five FPA Innovation awards, where he was the lead developer. He received his BS in chemical engineering at the University of Wisconsin Madison and his MS in engineer management at Milwaukee School of Engineering.