Sam Brusco, Associate Editor07.05.22
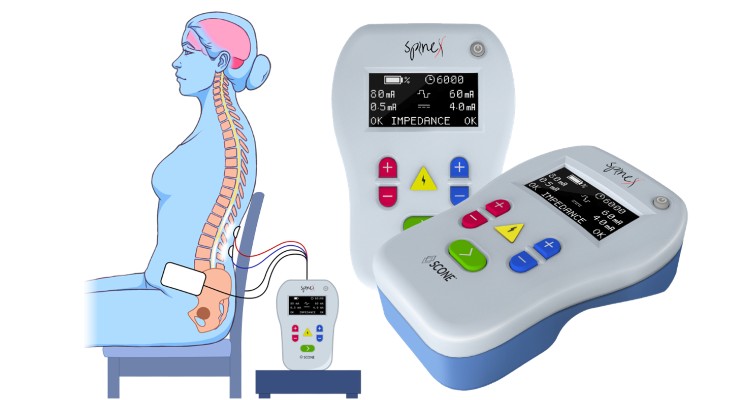
SpineX has begun a pivotal trial evaluating the SCONE non-invasive spinal neuromodulation device for neurogenic bladder treatment. The first patient was enrolled at Rancho Research Institute, the research arm of the prestigious and historic Rancho Los Amigos National Neurorehabilitation Center.
Neurogenic bladder is a common comorbidity that people suffer after paralysis. Surveys even suggest improving bladder, bowel, and sexual function as higher priority than restoration of ambulation and even chronic pain.
“For a person in a wheelchair, the inability to walk is the most obvious functional loss, but the impact on quality of life due to neurogenic bladder is unparalleled,” Dr. Evgeniy Kreydin, MD, co-founder of SpineX and assistant professor at University of Southern California told the press.
Neurogenic bladder patients deal with lost sensation of bladder fullness, low bladder capacity, frequent urination cycles during the day and night, and live with a constant fear of uncontrolled urine leakage.
“The initiation of the SCONE trial is an important milestone in bringing the world’s first non-invasive treatment modality for neurogenic bladder to market,” said Dr. Parag Gad, Ph.D., CEO of SpineX. “We are committed to transforming bladder management into a catheter free and leak free world.”
Neurogenic bladder is a common comorbidity that people suffer after paralysis. Surveys even suggest improving bladder, bowel, and sexual function as higher priority than restoration of ambulation and even chronic pain.
“For a person in a wheelchair, the inability to walk is the most obvious functional loss, but the impact on quality of life due to neurogenic bladder is unparalleled,” Dr. Evgeniy Kreydin, MD, co-founder of SpineX and assistant professor at University of Southern California told the press.
Neurogenic bladder patients deal with lost sensation of bladder fullness, low bladder capacity, frequent urination cycles during the day and night, and live with a constant fear of uncontrolled urine leakage.
“The initiation of the SCONE trial is an important milestone in bringing the world’s first non-invasive treatment modality for neurogenic bladder to market,” said Dr. Parag Gad, Ph.D., CEO of SpineX. “We are committed to transforming bladder management into a catheter free and leak free world.”