SpineGuard02.09.17
SpineGuard, a developer and marketer of disposable medical devices to improve spine surgery safety, has been granted a patent by the U.S. Patent Office for the application of its Dynamic Surgical Guidance technology for a new application: bone quality measurement.
“Because of population aging, orthopedists and neurosurgeons are treating an increasing number of osteoporotic patients, and they are expressing the need to precisely measure the bone quality. A widely shared opinion amongst surgeons is that the existing standard of care—known as a DEXA scan—only brings an uncertain (unreliable) answer to this growing need. This grant from the U.S. Patent Office follows previous patent grants in China and Japan. It confirms the potential of SpineGuard’s DSG technology for this new application,” said Pierre Jérôme, CEO and co-founder of SpineGuard.
Osteoporosis represents a serious and growing healthcare issue due to the world's aging population. Most patients with a fragility fracture are neither evaluated, nor treated for osteoporosis. For patients being diagnosed with osteoporosis, several complications are associated with spine surgeries, and the quality of the purchase of the pedicle screws is directly linked with the bone mineral density.
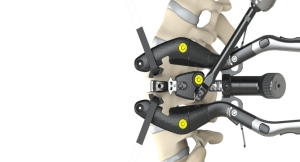
“We believe that the DSG technology can allow surgeons facing these skeletal pathologies to evaluate the bone density of their patients intraoperatively in a much more precise anatomical area and in doing so, to fine-tune their surgical strategy. For spine surgeries, this will ease the choice of the implants, their size, their diameter, their location and if cement should be used or not,” said Stéphane Bette, co-founder, chief technology officer and U.S. general manager of SpineGuard.
Co-founded in 2009 in France and the United States, SpineGuard’s mission is to make spine surgery safer by bringing real-time digital technology into the operating room. Its primary objective is to establish its proprietary DSG (Dynamic Surgical Guidance) technology as the global standard of surgical care, starting with safer screw placement in spine surgery and then in other surgeries. PediGuard, the first device designed using DSG, was co-invented by Maurice Bourlion, Ph.D., Ciaran Bolger, M.D., Ph.D., and Alain Vanquaethem, biomedical engineer. It is the world’s first and only handheld device capable of alerting surgeons to potential pedicular or vertebral breaches. More than 50,000 surgical procedures have been performed worldwide with DSG enabled devices. Numerous studies published in peer-reviewed medical and scientific journals have demonstrated the multiple benefits that PediGuard delivers to patients, surgical staff and hospitals. SpineGuard is expanding the scope of its DSG platform through strategic partnerships with medical device companies and the development of smart instruments and implants. SpineGuard has offices in San Francisco, Calof., and Paris, France.
Sources :
Bouxsein ML, Kaufman J, Tosi L, Cummings S, Lane J, Johnell O. Recommendations for optimal care of the fragility fracture patient to reduce the risk of future fracture. J Am Acad Orthop Surg. 2004 Nov-Dec;12(6):385-95.
Lehman RA Jr, Kang DG, Wagner SC. Management of osteoporosis in spine surgery. J Am Acad Orthop Surg. 2015 Apr.23 (4):253-63. doi: 10.5435/JAAOS-D-14-00042.
More information on the DSG™ technology, its new applications and surgeons’ testimonials here.
“Because of population aging, orthopedists and neurosurgeons are treating an increasing number of osteoporotic patients, and they are expressing the need to precisely measure the bone quality. A widely shared opinion amongst surgeons is that the existing standard of care—known as a DEXA scan—only brings an uncertain (unreliable) answer to this growing need. This grant from the U.S. Patent Office follows previous patent grants in China and Japan. It confirms the potential of SpineGuard’s DSG technology for this new application,” said Pierre Jérôme, CEO and co-founder of SpineGuard.
Osteoporosis represents a serious and growing healthcare issue due to the world's aging population. Most patients with a fragility fracture are neither evaluated, nor treated for osteoporosis. For patients being diagnosed with osteoporosis, several complications are associated with spine surgeries, and the quality of the purchase of the pedicle screws is directly linked with the bone mineral density.
“We believe that the DSG technology can allow surgeons facing these skeletal pathologies to evaluate the bone density of their patients intraoperatively in a much more precise anatomical area and in doing so, to fine-tune their surgical strategy. For spine surgeries, this will ease the choice of the implants, their size, their diameter, their location and if cement should be used or not,” said Stéphane Bette, co-founder, chief technology officer and U.S. general manager of SpineGuard.
Co-founded in 2009 in France and the United States, SpineGuard’s mission is to make spine surgery safer by bringing real-time digital technology into the operating room. Its primary objective is to establish its proprietary DSG (Dynamic Surgical Guidance) technology as the global standard of surgical care, starting with safer screw placement in spine surgery and then in other surgeries. PediGuard, the first device designed using DSG, was co-invented by Maurice Bourlion, Ph.D., Ciaran Bolger, M.D., Ph.D., and Alain Vanquaethem, biomedical engineer. It is the world’s first and only handheld device capable of alerting surgeons to potential pedicular or vertebral breaches. More than 50,000 surgical procedures have been performed worldwide with DSG enabled devices. Numerous studies published in peer-reviewed medical and scientific journals have demonstrated the multiple benefits that PediGuard delivers to patients, surgical staff and hospitals. SpineGuard is expanding the scope of its DSG platform through strategic partnerships with medical device companies and the development of smart instruments and implants. SpineGuard has offices in San Francisco, Calof., and Paris, France.
Sources :
Bouxsein ML, Kaufman J, Tosi L, Cummings S, Lane J, Johnell O. Recommendations for optimal care of the fragility fracture patient to reduce the risk of future fracture. J Am Acad Orthop Surg. 2004 Nov-Dec;12(6):385-95.
Lehman RA Jr, Kang DG, Wagner SC. Management of osteoporosis in spine surgery. J Am Acad Orthop Surg. 2015 Apr.23 (4):253-63. doi: 10.5435/JAAOS-D-14-00042.
More information on the DSG™ technology, its new applications and surgeons’ testimonials here.