BONESUPPORT08.07.18
BONESUPPORT, an emerging leader in orthobiologics for the management of bone voids, has signed an agreement with MTF Biologics, the world’s largest tissue bank, to extend and strengthen its U.S. product offering.
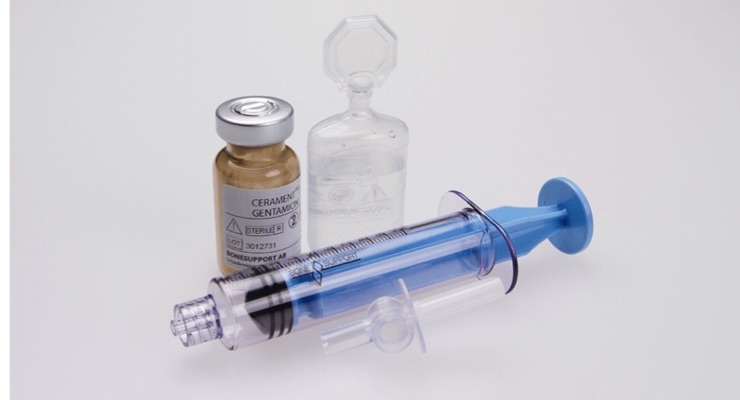
The agreement will give BONESUPPORT U.S. rights to two forms of bone graft materials comprised of 100 percent demineralized bone matrix (DBM), which offer improved osteoinductivity when compared to many other DBM products. These products, which are both osteoinductive and osteoconductive, complement CERAMENT BVF (osteoconductive) and the products gained from the company’s recent strategic agreement with Collagen Matrix Inc. (osteoinductive and osteogenic)
BONESUPPORT plans to launch the products supplied by MTF Biologics under the BONESUPPORT brand name and through its own U.S. distribution network.
MTF Biologics is a non-profit service organization dedicated to providing clinically sound, safe allograft tissue. MTF Biologics is comprised of a national consortium of academic medical institutions, organ procurement organizations and tissue recovery organizations. It is based in Edison, N.J.
Emil Billbäck, CEO of BONESUPPORT said, “Today’s deal with MTF Biologics is a further important step in creating the broad and complementary U.S. product offering that will address the needs of orthopedic surgeons managing bone voids. We are pleased that MTF Biologics has chosen to partner with BONESUPPORT, and we look forward to marketing products based on their demineralized bone matrix. With our expanded product portfolio and new broad U.S. distribution channel, that we are on track to have in place in late October, we are well placed to significantly improve our competitive position and rapidly grow sales ahead of the planned U.S. launch of CERAMENT G in 2021.”
“MTF Biologics is pleased to be partnering with BONESUPPORT on the promotion of our demineralized bone matrix fiber technology. We are excited for this opportunity to work together to deliver biologic solutions that serve the needs of surgeons and their patients.” said Tom Shaffer, MTF Biologics executive vice president of Global Sales and Marketing.
MTF Biologics is a nonprofit national consortium comprised of leading organ procurement organizations, tissue recovery organization and academic medical institutions, and governed by a board of surgeons who are leading experts in tissue transplantation. As the world's largest tissue bank, MTF Biologics saves and heals lives by honoring donated gifts, serving patients and advancing science. Since its inception in 1987, the organization has received tissue from more than 120,000 donors and distributed more than 7.5 million allografts for transplantation. Through its IIAM subsidiary, it has placed more than 55,000 non-transplantable organs for research. Through its Statline subsidiary, it has managed more than 10 million donor referrals.
BONESUPPORT is a rapidly growing commercial stage orthobiologics company, based in Lund, Sweden. The company develops and commercializes injectable bio-ceramic bone graft substitutes that remodel to the patient’s own bone and have the capability of eluting drugs directly into the bone void. BONESUPPORT’s bio-ceramic bone graft substitutes CERAMENT BONE VOID FILLER (BVF), CERAMENT G* and CERAMENT V* are all based on the company’s proprietary technology platform. The company’s products are targeting a large addressable market opportunity across trauma, chronic osteomyelitis (bone infection), revision arthroplasty (replacement of a joint prosthesis) and infected diabetic foot. BONESUPPORT’s total sales increased from SEK 62 million in 2015 to SEK 129 million in 2017, representing a compound annual growth rate of 45 percent.
The company’s research and development is focused on the continuing development and refinement of its CERAMENT technology to extend its use into additional indications by the elution of drugs and therapeutic agents. BONESUPPORT currently has a pipeline of pre-clinical product candidates that have been designed to promote bone growth. In addition, BONESUPPORT is looking to expand its product offering in the U.S. and has entered into strategic agreements with Collagen Matrix Inc. and MTF Biologics to market and distribute products that are complementary to CERAMENT BVF.
*CERAMENT G: Not available in the United States, for investigational use only.
CERAMENT V: Not available in the United States.
The agreement will give BONESUPPORT U.S. rights to two forms of bone graft materials comprised of 100 percent demineralized bone matrix (DBM), which offer improved osteoinductivity when compared to many other DBM products. These products, which are both osteoinductive and osteoconductive, complement CERAMENT BVF (osteoconductive) and the products gained from the company’s recent strategic agreement with Collagen Matrix Inc. (osteoinductive and osteogenic)
BONESUPPORT plans to launch the products supplied by MTF Biologics under the BONESUPPORT brand name and through its own U.S. distribution network.
MTF Biologics is a non-profit service organization dedicated to providing clinically sound, safe allograft tissue. MTF Biologics is comprised of a national consortium of academic medical institutions, organ procurement organizations and tissue recovery organizations. It is based in Edison, N.J.
Emil Billbäck, CEO of BONESUPPORT said, “Today’s deal with MTF Biologics is a further important step in creating the broad and complementary U.S. product offering that will address the needs of orthopedic surgeons managing bone voids. We are pleased that MTF Biologics has chosen to partner with BONESUPPORT, and we look forward to marketing products based on their demineralized bone matrix. With our expanded product portfolio and new broad U.S. distribution channel, that we are on track to have in place in late October, we are well placed to significantly improve our competitive position and rapidly grow sales ahead of the planned U.S. launch of CERAMENT G in 2021.”
“MTF Biologics is pleased to be partnering with BONESUPPORT on the promotion of our demineralized bone matrix fiber technology. We are excited for this opportunity to work together to deliver biologic solutions that serve the needs of surgeons and their patients.” said Tom Shaffer, MTF Biologics executive vice president of Global Sales and Marketing.
MTF Biologics is a nonprofit national consortium comprised of leading organ procurement organizations, tissue recovery organization and academic medical institutions, and governed by a board of surgeons who are leading experts in tissue transplantation. As the world's largest tissue bank, MTF Biologics saves and heals lives by honoring donated gifts, serving patients and advancing science. Since its inception in 1987, the organization has received tissue from more than 120,000 donors and distributed more than 7.5 million allografts for transplantation. Through its IIAM subsidiary, it has placed more than 55,000 non-transplantable organs for research. Through its Statline subsidiary, it has managed more than 10 million donor referrals.
BONESUPPORT is a rapidly growing commercial stage orthobiologics company, based in Lund, Sweden. The company develops and commercializes injectable bio-ceramic bone graft substitutes that remodel to the patient’s own bone and have the capability of eluting drugs directly into the bone void. BONESUPPORT’s bio-ceramic bone graft substitutes CERAMENT BONE VOID FILLER (BVF), CERAMENT G* and CERAMENT V* are all based on the company’s proprietary technology platform. The company’s products are targeting a large addressable market opportunity across trauma, chronic osteomyelitis (bone infection), revision arthroplasty (replacement of a joint prosthesis) and infected diabetic foot. BONESUPPORT’s total sales increased from SEK 62 million in 2015 to SEK 129 million in 2017, representing a compound annual growth rate of 45 percent.
The company’s research and development is focused on the continuing development and refinement of its CERAMENT technology to extend its use into additional indications by the elution of drugs and therapeutic agents. BONESUPPORT currently has a pipeline of pre-clinical product candidates that have been designed to promote bone growth. In addition, BONESUPPORT is looking to expand its product offering in the U.S. and has entered into strategic agreements with Collagen Matrix Inc. and MTF Biologics to market and distribute products that are complementary to CERAMENT BVF.
*CERAMENT G: Not available in the United States, for investigational use only.
CERAMENT V: Not available in the United States.