Zimmer Biomet Holdings Inc.11.05.18
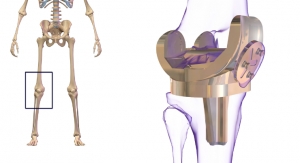
Zimmer Biomet Holdings Inc., a global leader in musculoskeletal healthcare, has announced U.S. Food and Drug Administration 510(k) clearance of the Persona Revision Knee System for revision knee replacement procedures. This revision system offers anatomic components designed to match a patient’s unique anatomy for a personalized fit. Available with a modern, intuitive instrumentation platform, the Persona Revision Knee System enables surgeons to take a personalized approach to addressing simple to complex revision procedures by offering the flexibility to utilize their preferred surgical approach.
“Given the complexity of revision procedures and the fact that every person’s anatomy is a little different, it is critical for a knee revision system to give surgeons the ability to personalize the treatment strategy in order to achieve the best outcome for each patient,” said Giles Scuderi, M.D., of New York, N.Y.
The Persona Revision Knee System utilizes Zimmer Biomet's proprietary technologies that are designed to enhance optimal fit and function:
“The clearance of the Persona Revision Knee System gives us the ability to provide surgeons with a full service portfolio for the continuum of knee arthroplasty care, from diagnostic tools, cement spacer technologies to re-implantation solutions,” said Todd Davis, Zimmer Biomet’s vice president and general manager of the global Knee business. “The Persona Revision Knee System gives surgeons the flexibility to truly tailor an implant solution based on each patient’s unique anatomy for a natural fit and function.”
Founded in 1927 and headquartered in Warsaw, Ind., Zimmer Biomet designs, manufactures, and markets orthopedic reconstructive products; sports medicine, biologics, extremities and trauma products; office-based technologies; spine, craniomaxillofacial and thoracic products; dental implants; and related surgical products. The company's products and solutions help treat patients suffering from disorders of, or injuries to, bones, joints or supporting soft tissues. Zimmer Biomet has operations in more than 25 countries around the world and sell products in more than 100 countries.
References
1. Bobyn, et al., Characterization of a New Porous Tantalum Biomaterial for Reconstructive Orthopaedics. 66th Annual AAOS 1999.
2. Zhang, et al., Interfacial Frictional Behavior: Cancellous Bone, Cortical Bone, and a Novel Porous Tantalum Biomaterial. Journal of Musculoskeletal Research. 3:4, 245-251, 1999.
3. Karageorgiou and Kaplan. Porosity of Biomaterial Scaffolds and Osteogenesis. Biomaterials. 26: 5474-91, 2005.
“Given the complexity of revision procedures and the fact that every person’s anatomy is a little different, it is critical for a knee revision system to give surgeons the ability to personalize the treatment strategy in order to achieve the best outcome for each patient,” said Giles Scuderi, M.D., of New York, N.Y.
The Persona Revision Knee System utilizes Zimmer Biomet's proprietary technologies that are designed to enhance optimal fit and function:
- Trabecular Metal Technology, the only tantalum-based porous material on the market with over 20 years of history making it one of the most clinically documented technologies.1-3
- Vivacit-E Highly Crosslinked Polyethylene (HXPE), a bearing surface designed with actively stabilized Vitamin E to protect against oxidation and maintain wear resistance and strength throughout the life of the implant.
“The clearance of the Persona Revision Knee System gives us the ability to provide surgeons with a full service portfolio for the continuum of knee arthroplasty care, from diagnostic tools, cement spacer technologies to re-implantation solutions,” said Todd Davis, Zimmer Biomet’s vice president and general manager of the global Knee business. “The Persona Revision Knee System gives surgeons the flexibility to truly tailor an implant solution based on each patient’s unique anatomy for a natural fit and function.”
Founded in 1927 and headquartered in Warsaw, Ind., Zimmer Biomet designs, manufactures, and markets orthopedic reconstructive products; sports medicine, biologics, extremities and trauma products; office-based technologies; spine, craniomaxillofacial and thoracic products; dental implants; and related surgical products. The company's products and solutions help treat patients suffering from disorders of, or injuries to, bones, joints or supporting soft tissues. Zimmer Biomet has operations in more than 25 countries around the world and sell products in more than 100 countries.
References
1. Bobyn, et al., Characterization of a New Porous Tantalum Biomaterial for Reconstructive Orthopaedics. 66th Annual AAOS 1999.
2. Zhang, et al., Interfacial Frictional Behavior: Cancellous Bone, Cortical Bone, and a Novel Porous Tantalum Biomaterial. Journal of Musculoskeletal Research. 3:4, 245-251, 1999.
3. Karageorgiou and Kaplan. Porosity of Biomaterial Scaffolds and Osteogenesis. Biomaterials. 26: 5474-91, 2005.