PR Newswire11.06.18
United Orthopedic Corporation, an international designer, manufacturer, and distributor of orthopedic implants and instruments, has announced that the UTS (United Tri-tapered Short Stem) hip stem and an extension of the U-Motion II Acetabular System are now available to U.S. surgeons.
"The UTS hip stem and U-Motion II extension reflect our commitment to addressing some of the challenges in today's hip replacement market—reducing dislocations and providing solutions that support rapid recovery from surgery," said Calvin Lin, president of United Orthopedic Corporation USA. "Since the launch of these products at this year's American Academy of Orthopaedic Surgeons Annual Meeting, we have received positive feedback from orthopedic surgeons, citing easier insertion of the hip stem, greater range of motion and improved stability."
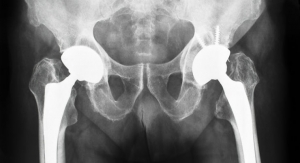
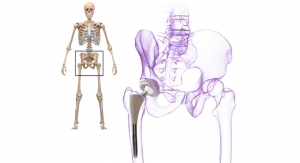
The UTS hip stem is a tapered wedge short stem suitable for less invasive primary hip replacement. It is designed for easier insertion through small incisions and requires simpler femoral preparation, enabling rapid recovery. Shorter stems preserve bone, which allows for more favorable conditions in future revisions.1 The UTS hip stem is available in 14 refined proximal sizes. Its features include standard and high offset options for restoring joint biomechanics, and a reduced lateral shoulder designed to conserve bone in the greater trochanter.
"The shortened stem length and plasma coating coupled with the unique shape and properties of the UTS stem afford greater stability at the time of implantation." said Richard Steinfeld, M.D., a practicing orthopedic surgeon from Orthopaedic Center of Vero Beach, Fla. "This provides for greater bone conservation and affords patients the ability to fully weight bear immediately following their surgical procedure."
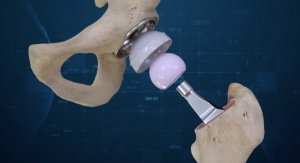
The extension of the U-Motion II Acetabular System for total hip replacement surgery includes a 50mm acetabular cup and cup liner, which is compatible with the 36mm BIOLOX delta ceramic and CoCrMo metal femoral heads. The 36mm femoral head with the new U-Motion II 50mm Acetabular cup is designed to provide patients a greater range of motion,2 increase jumping distance, improve stability, and lower the risk of dislocation.
"The UTS hip stem offers some unique advantages over other hip stems on the market. The proximal porous ingrowth surface and tapered design allows for early weight bearing while preventing subsidence. Multiple offset options are available, allowing fine tuning of the soft tissue balance and stability during surgery," said Ronald Hillock, M.D., an orthopedic surgeon at the Nevada Orthopedic & Spine Center, in Las Vegas, Nev. "The instruments are well thought out and streamlined. I have been pleased with my initial UTS experience."
The demand for primary total hip arthroplasties is estimated to grow by 174 percent to 572,000 by 2030, while the demand for hip revisions is projected to double by 2026.3 Dislocation continues to be one of the most common causes of failure after primary and revision total hip arthroplasty. Clinical studies show large femoral heads decrease the incidence of dislocation after total hip arthroplasty.4
United Orthopedic Corporation is an international designer, manufacturer, and distributor of regulatorily compliant orthopedic implants and instrument sets. The company offers solutions used in primary and revision total hip/knee replacement in addition to oncology applications. The company operates Quality Management Systems compliant with ISO 13485, U.S. Food and Drug Administration and CE requirements.
References
1. Molli RG,et al. A Short Tapered Stem Reduces Intraoperative Complications in Primary Total Hip Arthroplasty. Clin Orthop Relat Res. 2012 Feb;470(2):450-61.
2. Guyen O. Constrained liners, dual mobility or large diameter heads to avoid dislocation in THA. EFORT Open Reviews. 2016;1(5):197-204. doi:10.1302/2058-5241.1.000054.
3. Kurtz, et al. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007 Apr;89(4):780-5.
4. Howie, et al. Large femoral heads decrease the incidence of dislocation after total hip arthroplasty: a randomized controlled trial. J Bone Joint Surg Am. 2012 Jun 20;94(12):1095-102. doi: 10.2106/JBJS.K.00570.
"The UTS hip stem and U-Motion II extension reflect our commitment to addressing some of the challenges in today's hip replacement market—reducing dislocations and providing solutions that support rapid recovery from surgery," said Calvin Lin, president of United Orthopedic Corporation USA. "Since the launch of these products at this year's American Academy of Orthopaedic Surgeons Annual Meeting, we have received positive feedback from orthopedic surgeons, citing easier insertion of the hip stem, greater range of motion and improved stability."
The UTS hip stem is a tapered wedge short stem suitable for less invasive primary hip replacement. It is designed for easier insertion through small incisions and requires simpler femoral preparation, enabling rapid recovery. Shorter stems preserve bone, which allows for more favorable conditions in future revisions.1 The UTS hip stem is available in 14 refined proximal sizes. Its features include standard and high offset options for restoring joint biomechanics, and a reduced lateral shoulder designed to conserve bone in the greater trochanter.
"The shortened stem length and plasma coating coupled with the unique shape and properties of the UTS stem afford greater stability at the time of implantation." said Richard Steinfeld, M.D., a practicing orthopedic surgeon from Orthopaedic Center of Vero Beach, Fla. "This provides for greater bone conservation and affords patients the ability to fully weight bear immediately following their surgical procedure."
The extension of the U-Motion II Acetabular System for total hip replacement surgery includes a 50mm acetabular cup and cup liner, which is compatible with the 36mm BIOLOX delta ceramic and CoCrMo metal femoral heads. The 36mm femoral head with the new U-Motion II 50mm Acetabular cup is designed to provide patients a greater range of motion,2 increase jumping distance, improve stability, and lower the risk of dislocation.
"The UTS hip stem offers some unique advantages over other hip stems on the market. The proximal porous ingrowth surface and tapered design allows for early weight bearing while preventing subsidence. Multiple offset options are available, allowing fine tuning of the soft tissue balance and stability during surgery," said Ronald Hillock, M.D., an orthopedic surgeon at the Nevada Orthopedic & Spine Center, in Las Vegas, Nev. "The instruments are well thought out and streamlined. I have been pleased with my initial UTS experience."
The demand for primary total hip arthroplasties is estimated to grow by 174 percent to 572,000 by 2030, while the demand for hip revisions is projected to double by 2026.3 Dislocation continues to be one of the most common causes of failure after primary and revision total hip arthroplasty. Clinical studies show large femoral heads decrease the incidence of dislocation after total hip arthroplasty.4
United Orthopedic Corporation is an international designer, manufacturer, and distributor of regulatorily compliant orthopedic implants and instrument sets. The company offers solutions used in primary and revision total hip/knee replacement in addition to oncology applications. The company operates Quality Management Systems compliant with ISO 13485, U.S. Food and Drug Administration and CE requirements.
References
1. Molli RG,et al. A Short Tapered Stem Reduces Intraoperative Complications in Primary Total Hip Arthroplasty. Clin Orthop Relat Res. 2012 Feb;470(2):450-61.
2. Guyen O. Constrained liners, dual mobility or large diameter heads to avoid dislocation in THA. EFORT Open Reviews. 2016;1(5):197-204. doi:10.1302/2058-5241.1.000054.
3. Kurtz, et al. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007 Apr;89(4):780-5.
4. Howie, et al. Large femoral heads decrease the incidence of dislocation after total hip arthroplasty: a randomized controlled trial. J Bone Joint Surg Am. 2012 Jun 20;94(12):1095-102. doi: 10.2106/JBJS.K.00570.