Kolon TissueGene Inc.01.16.19
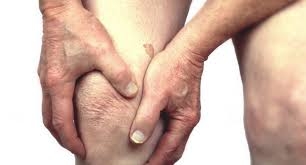
Kolon TissueGene Inc., a leader in advanced cell and gene therapies, has dosed its first patient in its pivotal U.S. Phase III clinical trials for Invossa, a cell and gene therapy in development for knee osteoarthritis (OA).
"This is an exciting step for Kolon TissueGene as we progress towards providing key clinical benefits for patients of this debilitating disease with this novel cell and gene therapy for knee osteoarthritis," stated Woosok Lee, president and CEO of Kolon TissueGene.
Kolon TissueGene is conducting pivotal Phase III trials for U.S. approval of Invossa for knee osteoarthritis. The pivotal phase III trials for U.S. approval of Invossa will enroll close to 1,020 patients in approximately 60 clinical sites across the United States. The trial investigators include orthopedic surgeons, rheumatologists and pain specialists. During the trial, the company will assess pain and function endpoints as well as magnetic resonance imaging, X-ray and liquid biomarkers.
In addition to demonstrating significant improvements in pain relief and function, the trials are designed to show structural benefits, including a delay in disease progression, and if successful could achieve a disease modifying osteoarthritis drug (DMOAD label claim). Such an indication by the U.S. Food and Drug Administration would be a first for any osteoarthritis drug approved in the United States.
Invossa is a first-in-class cell and gene therapy targeting OA of the knee through a single intra-articular injection. Clinical trials held in the United States and abroad have demonstrated pain relief and increased mobility, as well indicators towards decreased progression of OA and improvements in joint structure. The allogeneic (off-the-shelf) drug could provide an alternative to traditional treatment and surgery, or delay the progression of OA to minimize the need for multiple surgical interventions. In a concluded U.S. Phase II clinical trial, Kolon TissueGene demonstrated a two-year improvement of pain and function. The company seeks to continue to support these results through its planned national U.S. Phase III clinical trial. In July 2017, Kolon Life Science Inc., Kolon TissueGene's exclusive licensee for Asia, received marketing approval from the South Korea Ministry of Food & Drug Safety (MFDS) for Invossa-K Inj.
Kolon TissueGene Inc. is an advanced cell therapies company that has developed a first-in-class cell and gene therapy targeting OA of the knee. Kolon TissueGene's lead product, Invossa, is an allogeneic cell and gene therapy. The company is conducting Phase III clinical trials in the U.S. under a Special Protocol Assessment (SPA) agreement reached with the FDA.
"This is an exciting step for Kolon TissueGene as we progress towards providing key clinical benefits for patients of this debilitating disease with this novel cell and gene therapy for knee osteoarthritis," stated Woosok Lee, president and CEO of Kolon TissueGene.
Kolon TissueGene is conducting pivotal Phase III trials for U.S. approval of Invossa for knee osteoarthritis. The pivotal phase III trials for U.S. approval of Invossa will enroll close to 1,020 patients in approximately 60 clinical sites across the United States. The trial investigators include orthopedic surgeons, rheumatologists and pain specialists. During the trial, the company will assess pain and function endpoints as well as magnetic resonance imaging, X-ray and liquid biomarkers.
In addition to demonstrating significant improvements in pain relief and function, the trials are designed to show structural benefits, including a delay in disease progression, and if successful could achieve a disease modifying osteoarthritis drug (DMOAD label claim). Such an indication by the U.S. Food and Drug Administration would be a first for any osteoarthritis drug approved in the United States.
Invossa is a first-in-class cell and gene therapy targeting OA of the knee through a single intra-articular injection. Clinical trials held in the United States and abroad have demonstrated pain relief and increased mobility, as well indicators towards decreased progression of OA and improvements in joint structure. The allogeneic (off-the-shelf) drug could provide an alternative to traditional treatment and surgery, or delay the progression of OA to minimize the need for multiple surgical interventions. In a concluded U.S. Phase II clinical trial, Kolon TissueGene demonstrated a two-year improvement of pain and function. The company seeks to continue to support these results through its planned national U.S. Phase III clinical trial. In July 2017, Kolon Life Science Inc., Kolon TissueGene's exclusive licensee for Asia, received marketing approval from the South Korea Ministry of Food & Drug Safety (MFDS) for Invossa-K Inj.
Kolon TissueGene Inc. is an advanced cell therapies company that has developed a first-in-class cell and gene therapy targeting OA of the knee. Kolon TissueGene's lead product, Invossa, is an allogeneic cell and gene therapy. The company is conducting Phase III clinical trials in the U.S. under a Special Protocol Assessment (SPA) agreement reached with the FDA.