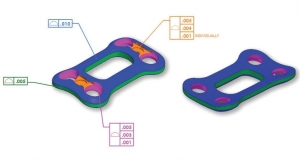
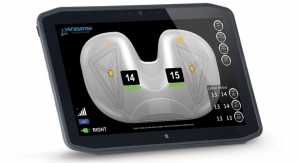
Sean Fenske, Editor-in-Chief01.07.19
It’s been said that “Necessity is the mother of invention.” Should a need present, someone somewhere will eventually create a solution.
In a very short time, the use of smart phones and other personal electronic devices has led to an unnatural position for the human head as people view their smartphones. This results in discomfort and, eventually, chronic neck pain caused by “text neck.” “Text neck,” or forward head posture, has grown into an epidemic, often resulting in neck pain, cervical arthritis, disc herniation, headache, arm and shoulder pain, lower lumbar disc and joint changes, among other effects.
Kenneth Hansraj, M.D., chief of spine surgery at New York Spine Surgery & Rehabilitation Medicine, offers a more clinical explanation of the condition. Using Cosmosworks, a finite element assessment package, Dr. Hansraj created a model to demonstrate the stresses being experienced by the neck when the head tilts forward by as little as 15 degrees.
“The weight seen by the spine dramatically increases when flexing the head forward at varying degrees,” explained Dr. Hansraj on the New York Spine Surgery website. “An adult head weighs 10 to 12 pounds in the neutral position. As the head tilts forward, the forces seen by the neck surges to 27 pounds at 15 degrees, 40 pounds at 30 degrees, 49 pounds at 45 degrees, and 60 pounds at 60 degrees.”1
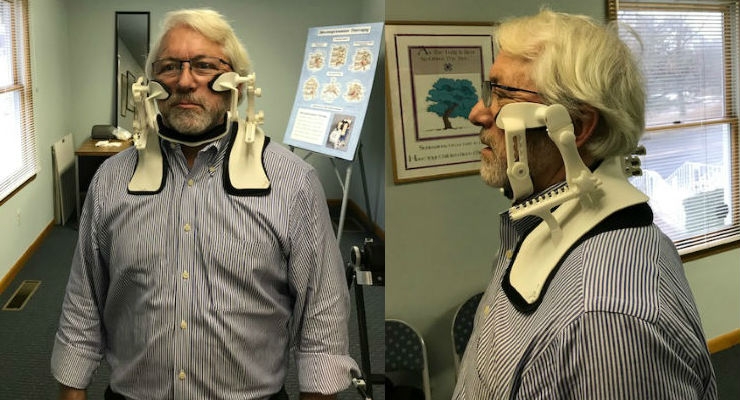
Recognizing this need, Dr. Ronald P. Dellanno invented the Cervigard Forward Head Posture Neck Collar. The device is worn by the patient for 20 minutes per day, but could be used for several hours at a time. The collar affixes to the neck and shoulders, and is equipped with cheek pads and a chin support. The device is adjusted until the wearer’s head is over the neck and shoulders. This positioning can be achieved over a period of weeks to accommodate for severe pain starting points as the head is moved backwards into proper position. The collar is intended to help patients address the issues experience from “text neck” by realigning the head to a more natural position.
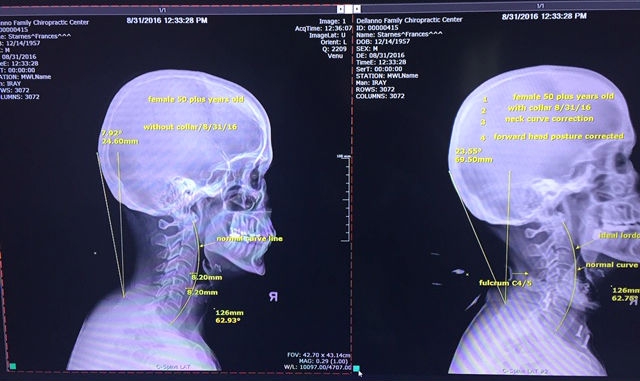
At left, the X-ray displays the painful neck alignment prior to treatment. At right, the X-ray shows the collar correction of both the neck curve and the forward head posture.
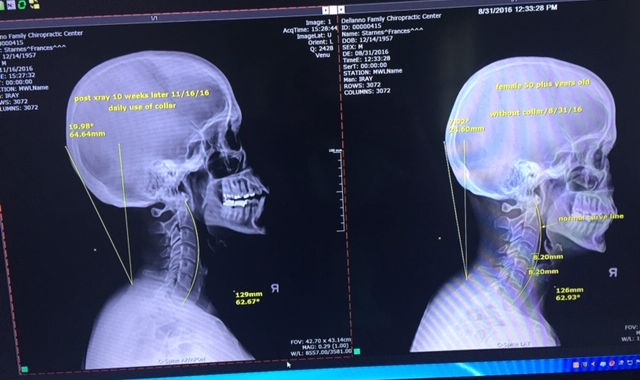
At left, the corrected spine alignment is displayed after 10 weeks of daily use of the Cervigard for a 20-minute duration. This is a permanent correction. At right, the original X-ray showing the patient’s alignment.
Curious about the technology and the processes taken to develop the device, Orthopedic Design and Technology spoke with Dr. Dellanno regarding his collar.
Sean Fenske: What has been the response from users/patients?
Dr. Ronald P. Dellanno: The response has been universally positive. Patients experience a reduction in pain (on a subjective measurement scale) and the clinicians see improvement in neck posture as shown on x-ray. Feedback by patients includes a video testimonial by an ob/gyn physician who was willing to endorse the product.
Fenske: How was it developed/manufactured? Was it done “roughly," such as in a home workshop, or was a contract manufacturing firm used?
Dr. Dellanno: The product has gone through a series of prototype phases using design engineering and manufacturing firms. The most recent phase was by a large-volume manufacturer who completed a pilot production run for product verification. The product is now in the full production phase.
Fenske: Did the final version come about as a result of several rounds of prototypes? If so, what were some of the changes to the design along the way?
Dr. Dellanno: Yes, the final version is the result of an iterative process, as noted previously. The most noticeable change was how the ratchet mechanism works. This took a few iterations to get it so that it had the proper length of travel and the gears matched properly.
Fenske: What was the inspiration for the development of the device?
Dr. Dellanno: The challenges of forward head posture—also called “text neck”—are well understood among chiropractors and physiatrists. Current products and methods fall short of providing relief from pain and correction of the curvature (or lordosis) of the cervical spine. It was the experiences of patients in my clinic that led me to develop the device, in collaboration with a designer who has done other work in orthotic devices and a physiatrist who runs a clinic to address orthopedic injury.
Fenske: To your knowledge, are there any current, non-surgical technologies on the market that address this issue?
Dr. Dellanno: There are other devices (i.e., a variety of neck braces) that seek to address the problem of “text neck.” None of them, however, actively position the head over the shoulders in a way that provides relief to the pain that patients suffer.
Fenske: Is development still ongoing or is the current version envisioned as the final design?
Dr. Dellanno: The current version of the collar is envisioned as the final design for the present time. It has been used extensively for patients suffering from neck pain and design iterations have resulted in a product that is well-tested and effective. That said, we will conduct comparison studies of other devices and methods to demonstrate the broader effectiveness of the Cervigard collar and actively monitor feedback on the use of the device.
As we gain experience with an ever-wider range of patients, there may be versions that would be better suited for larger patients or patients with anatomical variations who could benefit from other form factors in the device. At the present time, we believe the current version will serve the vast majority of patients but we want to be able to serve all patients who suffer from neck pain.
Fenske: Has the device gone through any types of clinical trials?
Dr. Dellanno: While no clinical trials are needed for regulatory or reimbursement purposes, there has been broad use of the product in a variety of patients who suffer from cervical neck pain. Data from these patients have been captured and show effectiveness (qualitative results). Quantitative studies to demonstrate effectiveness of the product are planned.
Fenske: Have you secured or have you applied for a patent for the device?
Dr. Dellanno: U.S. Patent US 8,038,635 has been granted for the Forward Head Position Correction Collar and PCT patents have been applied.

The Cervigard can be placed and adjusted by the patient.
Fenske: Can a patient “get into” the device on his/her own or does it require another person to assist?
Dr. Dellanno: The collar can be placed and adjusted by the patient. Initial settings are made by and with a clinician. Once these fine adjustments have been made in the practice office, the collar is taken by the patient to use at home for a prescribed period of time.
Fenske: What’s your next step for the device? Going to market? Pitching to companies for purchase or distribution?
Dr. Dellanno: The product is scheduled to be available for general use in the spring. An important part of our business is regulatory compliance and we are registered in good standing with the FDA. Initial sales are planned through selected practices of chiropractors and physiatrists. Some training on the use of all features of the collar for practitioners is needed and training certification is in place. The product will be available through the company’s web site (cervigard.com) as well as by phone.
The company ARMAC is the first distributor we have engaged; they are a sports medicine distributor of orthotic and other devices, as well as experts in fitting devices, supporting practitioners, and conducting medical billing. Several on-going discussions are addressing the availability of the collar through networks of rehabilitation clinics.
Reference
1 https://realspinesurgery.com/text-neck/
In a very short time, the use of smart phones and other personal electronic devices has led to an unnatural position for the human head as people view their smartphones. This results in discomfort and, eventually, chronic neck pain caused by “text neck.” “Text neck,” or forward head posture, has grown into an epidemic, often resulting in neck pain, cervical arthritis, disc herniation, headache, arm and shoulder pain, lower lumbar disc and joint changes, among other effects.
Kenneth Hansraj, M.D., chief of spine surgery at New York Spine Surgery & Rehabilitation Medicine, offers a more clinical explanation of the condition. Using Cosmosworks, a finite element assessment package, Dr. Hansraj created a model to demonstrate the stresses being experienced by the neck when the head tilts forward by as little as 15 degrees.
“The weight seen by the spine dramatically increases when flexing the head forward at varying degrees,” explained Dr. Hansraj on the New York Spine Surgery website. “An adult head weighs 10 to 12 pounds in the neutral position. As the head tilts forward, the forces seen by the neck surges to 27 pounds at 15 degrees, 40 pounds at 30 degrees, 49 pounds at 45 degrees, and 60 pounds at 60 degrees.”1
Recognizing this need, Dr. Ronald P. Dellanno invented the Cervigard Forward Head Posture Neck Collar. The device is worn by the patient for 20 minutes per day, but could be used for several hours at a time. The collar affixes to the neck and shoulders, and is equipped with cheek pads and a chin support. The device is adjusted until the wearer’s head is over the neck and shoulders. This positioning can be achieved over a period of weeks to accommodate for severe pain starting points as the head is moved backwards into proper position. The collar is intended to help patients address the issues experience from “text neck” by realigning the head to a more natural position.

At left, the X-ray displays the painful neck alignment prior to treatment. At right, the X-ray shows the collar correction of both the neck curve and the forward head posture.

At left, the corrected spine alignment is displayed after 10 weeks of daily use of the Cervigard for a 20-minute duration. This is a permanent correction. At right, the original X-ray showing the patient’s alignment.
Curious about the technology and the processes taken to develop the device, Orthopedic Design and Technology spoke with Dr. Dellanno regarding his collar.
Sean Fenske: What has been the response from users/patients?
Dr. Ronald P. Dellanno: The response has been universally positive. Patients experience a reduction in pain (on a subjective measurement scale) and the clinicians see improvement in neck posture as shown on x-ray. Feedback by patients includes a video testimonial by an ob/gyn physician who was willing to endorse the product.
Fenske: How was it developed/manufactured? Was it done “roughly," such as in a home workshop, or was a contract manufacturing firm used?
Dr. Dellanno: The product has gone through a series of prototype phases using design engineering and manufacturing firms. The most recent phase was by a large-volume manufacturer who completed a pilot production run for product verification. The product is now in the full production phase.
Fenske: Did the final version come about as a result of several rounds of prototypes? If so, what were some of the changes to the design along the way?
Dr. Dellanno: Yes, the final version is the result of an iterative process, as noted previously. The most noticeable change was how the ratchet mechanism works. This took a few iterations to get it so that it had the proper length of travel and the gears matched properly.
Fenske: What was the inspiration for the development of the device?
Dr. Dellanno: The challenges of forward head posture—also called “text neck”—are well understood among chiropractors and physiatrists. Current products and methods fall short of providing relief from pain and correction of the curvature (or lordosis) of the cervical spine. It was the experiences of patients in my clinic that led me to develop the device, in collaboration with a designer who has done other work in orthotic devices and a physiatrist who runs a clinic to address orthopedic injury.
Fenske: To your knowledge, are there any current, non-surgical technologies on the market that address this issue?
Dr. Dellanno: There are other devices (i.e., a variety of neck braces) that seek to address the problem of “text neck.” None of them, however, actively position the head over the shoulders in a way that provides relief to the pain that patients suffer.
Fenske: Is development still ongoing or is the current version envisioned as the final design?
Dr. Dellanno: The current version of the collar is envisioned as the final design for the present time. It has been used extensively for patients suffering from neck pain and design iterations have resulted in a product that is well-tested and effective. That said, we will conduct comparison studies of other devices and methods to demonstrate the broader effectiveness of the Cervigard collar and actively monitor feedback on the use of the device.
As we gain experience with an ever-wider range of patients, there may be versions that would be better suited for larger patients or patients with anatomical variations who could benefit from other form factors in the device. At the present time, we believe the current version will serve the vast majority of patients but we want to be able to serve all patients who suffer from neck pain.
Fenske: Has the device gone through any types of clinical trials?
Dr. Dellanno: While no clinical trials are needed for regulatory or reimbursement purposes, there has been broad use of the product in a variety of patients who suffer from cervical neck pain. Data from these patients have been captured and show effectiveness (qualitative results). Quantitative studies to demonstrate effectiveness of the product are planned.
Fenske: Have you secured or have you applied for a patent for the device?
Dr. Dellanno: U.S. Patent US 8,038,635 has been granted for the Forward Head Position Correction Collar and PCT patents have been applied.

The Cervigard can be placed and adjusted by the patient.
Dr. Dellanno: The collar can be placed and adjusted by the patient. Initial settings are made by and with a clinician. Once these fine adjustments have been made in the practice office, the collar is taken by the patient to use at home for a prescribed period of time.
Fenske: What’s your next step for the device? Going to market? Pitching to companies for purchase or distribution?
Dr. Dellanno: The product is scheduled to be available for general use in the spring. An important part of our business is regulatory compliance and we are registered in good standing with the FDA. Initial sales are planned through selected practices of chiropractors and physiatrists. Some training on the use of all features of the collar for practitioners is needed and training certification is in place. The product will be available through the company’s web site (cervigard.com) as well as by phone.
The company ARMAC is the first distributor we have engaged; they are a sports medicine distributor of orthotic and other devices, as well as experts in fitting devices, supporting practitioners, and conducting medical billing. Several on-going discussions are addressing the availability of the collar through networks of rehabilitation clinics.
Reference
1 https://realspinesurgery.com/text-neck/