DePuy Synthes01.31.19
DePuy Synthes** is launching the Femoral Neck System, a new implant solution for the repair of femoral neck fractures. Approximately 1.6 million hip fractures, specifically fractures that take place in the upper part of the femur or long thigh bone, occur worldwide annually, and femoral neck fractures represent approximately 50 percent of all reported hip fracture cases.1 These types of fractures are often associated with high mortality rates, along with significant loss of function in more than 50 percent of patients.2,3
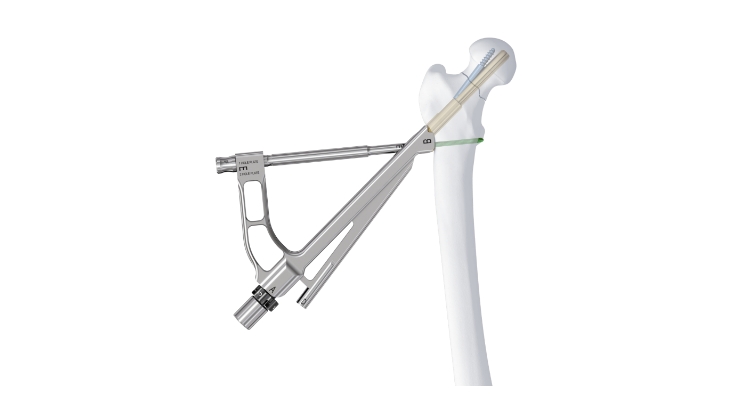
The Femoral Neck System is designed to address the need for improved treatment options for these types of fractures by combining stable fixation in a compact design with a minimally invasive approach, with the intent to reduce complications associated with current devices on the market.4,5 The product is designed to offer a minimized footprint on the bone and a reduced length of incision, which reduces the surface area where a surgical site infection can develop compared to current existing solutions.6
"The DePuy Synthes Femoral Neck System has an articulated blade and screw design that limits rotational movement on the femoral head and is protective of an anatomic provisional reduction, which is a critical step in the treatment of femoral neck fractures. It may additionally optimize the mechanical advantages of a sliding hip screw while mitigating the negative effects of rotational displacement seen with traditional sliding hip screws,” said Eben A. Carroll, M.D., orthopedic trauma surgeon.*** "The System builds nicely on DePuy Synthes’ existing comprehensive portfolio of implants for hip and femur fractures and offers a dedicated alternative to traditional fixation systems used to treat these difficult fractures."
This new system combines enhanced fixation in a compact design and strengthens the company’s current hip and femur fracture portfolio, which includes TFN-ADVANCED Proximal Femoral Nailing System (TFNA), TRAUMACEM V+ Injectable Bone Cement and the Femoral Recon Nail. With the addition of the Femoral Neck System to its growing portfolio, the company now offers options with specialized solutions to address unmet clinical needs for all types of hip and femur fractures.
The Femoral Neck System offers a streamlined procedure and repeatable approach using a targeted insertion handle to complete all steps of the procedure after placement of one central guidewire into the femoral head.6 The system’s single, dedicated set for the fixation of femoral neck fractures may increase hospital standardization and reduce product overlap, ultimately leading to higher set availability, better hospital logistics and potential cost savings.
“DePuy Synthes has a history of providing solutions in the hip and femur fixation market with a broad portfolio of implants and services that address a variety of patient needs,” said I.V. Hall, worldwide president, Trauma, Craniomaxillofacial & Animal Health, DePuy Synthes.**** “With the launch of the Femoral Neck System, we are proud to continue to innovate in this space and bring new options to the market for patients while creating value for our customers.”
DePuy Synthes, part of the Johnson & Johnson Medical Devices Companies, provides an orthopedics portfolio in specialties including joint reconstruction, trauma, craniomaxillofacial, spinal surgery and sports medicine.
**DePuy Synthes represents the products and services of DePuy Synthes, Inc. and its affiliates.
*** Consultant to DePuy Synthes Trauma
**** Employed by Medical Device Business Services Inc.
† Benchtop testing may not be indicative of clinical performance.
References
1 Filipov O. Epidemiology and social burden of the femoral neck fractures. Journal of IMAB. 2014; 20(4):516-518.
2 Sernbo I, Johnell O. Consequences of a hip fracture: a prospective study over 1 year. Osteoporosis International. 1993; 3(3):148-153.
3 Todd CJ, Freeman CJ, Camilleri-Ferrante C, Palmer CR, Hyder A, Laxton CE, et al. Differences in mortality after fracture of hip: the east Anglian audit. BMJ. 1995; 310:904-908.
4 Stoffel K, Zderic I, Gras F, et al. Biomechanical Evaluation of the Femoral Neck System in Unstable Pauwels III Femoral Neck Fractures: A Comparison with the Dynamic Hip Screw and Cannulated Screws Orthop Trauma. 2017. (31): 131-137.
5 DePuy Synthes Report: Static Cut Through Rotation Test in Bone Foam. 2016. Ref: 0000165855.†
6 DePuy Synthes Report FNS Design & Procedure Comparison. 2018. Ref: 0000274963
The Femoral Neck System is designed to address the need for improved treatment options for these types of fractures by combining stable fixation in a compact design with a minimally invasive approach, with the intent to reduce complications associated with current devices on the market.4,5 The product is designed to offer a minimized footprint on the bone and a reduced length of incision, which reduces the surface area where a surgical site infection can develop compared to current existing solutions.6
"The DePuy Synthes Femoral Neck System has an articulated blade and screw design that limits rotational movement on the femoral head and is protective of an anatomic provisional reduction, which is a critical step in the treatment of femoral neck fractures. It may additionally optimize the mechanical advantages of a sliding hip screw while mitigating the negative effects of rotational displacement seen with traditional sliding hip screws,” said Eben A. Carroll, M.D., orthopedic trauma surgeon.*** "The System builds nicely on DePuy Synthes’ existing comprehensive portfolio of implants for hip and femur fractures and offers a dedicated alternative to traditional fixation systems used to treat these difficult fractures."
This new system combines enhanced fixation in a compact design and strengthens the company’s current hip and femur fracture portfolio, which includes TFN-ADVANCED Proximal Femoral Nailing System (TFNA), TRAUMACEM V+ Injectable Bone Cement and the Femoral Recon Nail. With the addition of the Femoral Neck System to its growing portfolio, the company now offers options with specialized solutions to address unmet clinical needs for all types of hip and femur fractures.
The Femoral Neck System offers a streamlined procedure and repeatable approach using a targeted insertion handle to complete all steps of the procedure after placement of one central guidewire into the femoral head.6 The system’s single, dedicated set for the fixation of femoral neck fractures may increase hospital standardization and reduce product overlap, ultimately leading to higher set availability, better hospital logistics and potential cost savings.
“DePuy Synthes has a history of providing solutions in the hip and femur fixation market with a broad portfolio of implants and services that address a variety of patient needs,” said I.V. Hall, worldwide president, Trauma, Craniomaxillofacial & Animal Health, DePuy Synthes.**** “With the launch of the Femoral Neck System, we are proud to continue to innovate in this space and bring new options to the market for patients while creating value for our customers.”
DePuy Synthes, part of the Johnson & Johnson Medical Devices Companies, provides an orthopedics portfolio in specialties including joint reconstruction, trauma, craniomaxillofacial, spinal surgery and sports medicine.
**DePuy Synthes represents the products and services of DePuy Synthes, Inc. and its affiliates.
*** Consultant to DePuy Synthes Trauma
**** Employed by Medical Device Business Services Inc.
† Benchtop testing may not be indicative of clinical performance.
References
1 Filipov O. Epidemiology and social burden of the femoral neck fractures. Journal of IMAB. 2014; 20(4):516-518.
2 Sernbo I, Johnell O. Consequences of a hip fracture: a prospective study over 1 year. Osteoporosis International. 1993; 3(3):148-153.
3 Todd CJ, Freeman CJ, Camilleri-Ferrante C, Palmer CR, Hyder A, Laxton CE, et al. Differences in mortality after fracture of hip: the east Anglian audit. BMJ. 1995; 310:904-908.
4 Stoffel K, Zderic I, Gras F, et al. Biomechanical Evaluation of the Femoral Neck System in Unstable Pauwels III Femoral Neck Fractures: A Comparison with the Dynamic Hip Screw and Cannulated Screws Orthop Trauma. 2017. (31): 131-137.
5 DePuy Synthes Report: Static Cut Through Rotation Test in Bone Foam. 2016. Ref: 0000165855.†
6 DePuy Synthes Report FNS Design & Procedure Comparison. 2018. Ref: 0000274963