Globe Newswire08.13.20
Safe Orthopaedics, a company specializing in the design and marketing of ready-to-use technologies for spinal surgeries, delivering the safest treatment of spinal fractures urgently treated, has received 510k approval from the U.S. Food and Drug Administration (FDA), for the sale the second generation of SteriSpine PS in the United States.
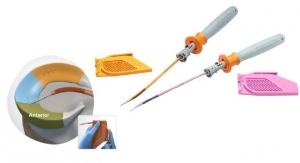
Developed in 2010, SteriSpine PS is the first technology of pedicle screws combined to a ready-to-use instrumentation (single use and sterile) worldwide. As more than 10,000 surgeries have been performed, Safe orthopaedics has been announcing in 2019 the launch of its 2nd generation. Significantly thinner, more rigid and still radiolucent, SteriSpine PS 2nd generation enables the surgeon to perform the surgery in an even more mini-invasive approach, and can be used on a wider number of spine pathologies.
Safe Orthopaedics has been enhancing business development of SteriSpine PS 2nd generation since the beginning of 2020. In the first quarter, technology was delivered to Japan, and in the second quarter, it was available to several Europeans countries and in Asia Pacific area. At the end of the year, all direct French, English and German direct customers will have been converted.
Following this 510k approval from the FDA, Safe Orthopaedics is getting ready for its commercial launch.
"Obtaining the FDA approval for the 2nd Generation SteriSpine PS in the United States is an important milestone in entering our second decade, as we observe a real adoption of ready-to-use technologies on a global scale and growing competition. Thanks to the feedback from our European users, we have designed this thinner, more rigid and totally radio-lucent evolution for an easier minimally invasive surgery, now meeting the expectations of American surgeons," sid Pierre Dumouchel, Safe Orthopaedics CEO. "Following our first experience in the United States, we are very attentive to developments happening in the world's leading market, and are organizing ourselves to make the SteriSpine PS second generation available to American surgeons. The acceleration of outpatient surgeries, in dedicated centers called Ambulatory Surgery Centers (ASC), represents a new business opportunity for Safe Orthopedics. Combined with a very short production cycle made possible by the acquisition of LCI Medical, in 2021 we will launch a new distribution method, totally digital and perfectly fitted to the ASC."
Finally, Safe Orthopedics, which already holds around a hundred patents, is also announcing today the acquisition of the Japanese patent for its Oak screw, an implant from the SteriSpine PS range offering an unprecedented minimally invasive surgical technique for treating high-energy fractures.
Developed in 2010, SteriSpine PS is the first technology of pedicle screws combined to a ready-to-use instrumentation (single use and sterile) worldwide. As more than 10,000 surgeries have been performed, Safe orthopaedics has been announcing in 2019 the launch of its 2nd generation. Significantly thinner, more rigid and still radiolucent, SteriSpine PS 2nd generation enables the surgeon to perform the surgery in an even more mini-invasive approach, and can be used on a wider number of spine pathologies.
Safe Orthopaedics has been enhancing business development of SteriSpine PS 2nd generation since the beginning of 2020. In the first quarter, technology was delivered to Japan, and in the second quarter, it was available to several Europeans countries and in Asia Pacific area. At the end of the year, all direct French, English and German direct customers will have been converted.
Following this 510k approval from the FDA, Safe Orthopaedics is getting ready for its commercial launch.
"Obtaining the FDA approval for the 2nd Generation SteriSpine PS in the United States is an important milestone in entering our second decade, as we observe a real adoption of ready-to-use technologies on a global scale and growing competition. Thanks to the feedback from our European users, we have designed this thinner, more rigid and totally radio-lucent evolution for an easier minimally invasive surgery, now meeting the expectations of American surgeons," sid Pierre Dumouchel, Safe Orthopaedics CEO. "Following our first experience in the United States, we are very attentive to developments happening in the world's leading market, and are organizing ourselves to make the SteriSpine PS second generation available to American surgeons. The acceleration of outpatient surgeries, in dedicated centers called Ambulatory Surgery Centers (ASC), represents a new business opportunity for Safe Orthopedics. Combined with a very short production cycle made possible by the acquisition of LCI Medical, in 2021 we will launch a new distribution method, totally digital and perfectly fitted to the ASC."
Finally, Safe Orthopedics, which already holds around a hundred patents, is also announcing today the acquisition of the Japanese patent for its Oak screw, an implant from the SteriSpine PS range offering an unprecedented minimally invasive surgical technique for treating high-energy fractures.