Promimic08.25.20
Promimic AB, an innovator in nano surface modification, announced the recent Innovasis Inc. special 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the first spinal interbody devices modified with HAnano Surface. This was the first special 510(k) FDA approval of a device with HAnano Surface, meaning that since the regulatory pathway is established, an expedited regulatory process can occur. This also marks the first approval of a 3D printed porous titanium implant modified with HAnano Surface.
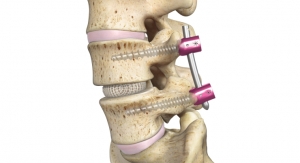
The newly approved Innovasis micro porous 3D printed titanium devices include the AxTiHA System, a standalone IBF device for Anterior Lumbar Interbody Fusion (ALIF) with up to 66 percent porosity, and the TxTiHA System, an IBF device for Transforaminal Lumbar Interbody Fusion (TLIF) with up to 61 percent porosity. Through modification with HAnano Surface, bio-enhanced AxTiHA and TxTiHA fully maintain their micro porous properties, allows osseointegration, and are super hydrophilic, which can stimulate an osteoconductive integration process to promote faster and more direct bone on-growth to the implant.
“The product development efforts with Innovasis have been extremely positive and collaborative. Promimic is delighted that they chose our HAnano Surface technology to further differentiate their product portfolio. With them and our other spine, orthopedic, and dental corporate partners, we are excited to further our mission of making HAnano Surface the gold standard for osseointegration of surgical implants,” said Magnus Larsson, Promimic CEO.
HAnano Surface has been evaluated in more than 30 pre-clinical studies testing a variety of implant materials and designs in different models. It was proven to increase anchoring strength in surface modified titanium implants by up to 35 percent in just 3 weeks. The results also show that HAnano Surface up-regulates important bone marker proteins and improves new bone formation on titanium implant surface by 67 percent at 4 weeks.
The newly approved Innovasis micro porous 3D printed titanium devices include the AxTiHA System, a standalone IBF device for Anterior Lumbar Interbody Fusion (ALIF) with up to 66 percent porosity, and the TxTiHA System, an IBF device for Transforaminal Lumbar Interbody Fusion (TLIF) with up to 61 percent porosity. Through modification with HAnano Surface, bio-enhanced AxTiHA and TxTiHA fully maintain their micro porous properties, allows osseointegration, and are super hydrophilic, which can stimulate an osteoconductive integration process to promote faster and more direct bone on-growth to the implant.
“The product development efforts with Innovasis have been extremely positive and collaborative. Promimic is delighted that they chose our HAnano Surface technology to further differentiate their product portfolio. With them and our other spine, orthopedic, and dental corporate partners, we are excited to further our mission of making HAnano Surface the gold standard for osseointegration of surgical implants,” said Magnus Larsson, Promimic CEO.
HAnano Surface has been evaluated in more than 30 pre-clinical studies testing a variety of implant materials and designs in different models. It was proven to increase anchoring strength in surface modified titanium implants by up to 35 percent in just 3 weeks. The results also show that HAnano Surface up-regulates important bone marker proteins and improves new bone formation on titanium implant surface by 67 percent at 4 weeks.