Michael Barbella, Managing Editor06.05.17
Consider it a natural evolution, of sorts.
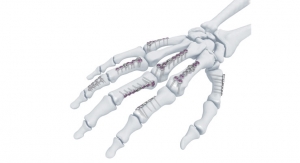
The planet’s extremities market, long ignored by major orthopedic device manufacturers, is finally commanding respect as companies attempt to counterbalance sluggish growth in the saturated, maturing hip and knee replacement sectors. Fueled by an aging world population, more youth-related sports injuries, and rising rates of obesity, osteoarthritis, and osteoporosis, the global extremities market is projected to be worth $15.9 billion by 2024, according to Research and Markets data. Internal fixation devices such as plates and screws have dominated the sector in recent years due to the early functionality, abbreviated hospital stays, minimal scarring, and reduced non-union risk associated with these products. Acumed LLC has particularly benefited from the high demand for internal fixation, having sold 500,000 wrist fixation implants (Acu-Loc and Acu-Loc 2) in just two years.
Such promise, of course, is garnering interest among major orthopedic companies that traditional have overlooked the sector in favor of the more lucrative large-joint sector. ODT’s May/June feature story “Going to Extremes” examines the trends and challenges driving the extremities market as well as some of the latest technologies available to patients. Dave Neal, president and founder of Morris Plains, N.J.-based STAT Design LLC, was among the experts interviewed for the feature; his full input is provided in the following Q&A.
Michael Barbella: Please discuss the trends in the extremities market. What trends have you noticed this year and how do they differ (if at all) from past trends?
Dave Neal: It seems there are more and more small players entering the extremities market, particularly in the foot and ankle realm. In the past, the big orthopedic companies tended to ignore this market, opting to concentrate on the larger hip and knee markets. Now they are adding this segment to their portfolio either through in-house product development of through an acquisition.
Barbella: What challenges face companies in the small joint (extremities) sector, and how are these challenges different than those faced by companies manufacturing large joints - i.e., hips and knees? How can companies in the extremities sector overcome these challenges?
Neal: As it is more of a niche market, the smaller extremities players are finding it difficult to gain traction in hospitals where buying groups like to have the large and small joints bundled for a full portfolio. Small companies have the best chance of success by making their extremity devices so attractive that the larger companies are seeking them out for an acquisition.
Barbella: What new technologies are on the horizon? What kinds of implants hold the most potential for patients and for market growth?
Neal: There are all sorts of new technologies emerging in the orthopedic medical device world. Additive manufacturing is changing the way parts are made. Special CAD software, such as that by nTopology, adds porous structure to designs assisting with the bone in-growth featured in implants. Materials such as PEEK are being used more, along with the addition of titanium coatings applied on this material. Foot and ankle implants are taking advantage of these new technologies and driving the market growth.
Barbella: Where does innovation come from in the extremities market? How can (do) companies stay innovative?
Neal: Innovation is being driven by understanding and addressing the doctor’s clinical needs. Observing surgery and identifying where improvements can be made to benefit the patient, surgeon, staff, hospital, and insurance companies alike are also helping to grow the extremity market.
Barbella: Aside from the implants, how does the extremities market differ from large joints, or spine? What makes extremities implants a design challenge?
Neal: For gaining FDA release through the 510k process, it can be difficult to find equivalent predicate extremity devices for comparison. This challenge has companies rethinking the decision to enter these markets, based on the regulatory path. Many times they will forego the project if it requires a pre-market approval (PMA) based on the return on investment and resources involved.
Barbella: How do the technological/physiological demands of small joints differ from other orthopedic markets?
Neal: From the design side, high stresses resulting from large forces acting on these joints and small contact areas make implant development difficult in the extremities market. The surgical procedures themselves can be more technically demanding and proper patient selection and patient activity compliance is essential for good clinical outcomes.
Barbella: Where do the extremities and trauma markets overlap? How do they differ?
Neal: The extremities and trauma markets can overlap in how they are used. For instance, ankle replacements could be used in one case to repair degenerated cartilage from arthritis and in the next it could be used as a replacement resulting from trauma in a motor vehicle accident.
Barbella: Where do you see the extremities market five years from now?
Neal: Niche implants, such as hand, fingers, wrist and associated instrumentation being developed by small specialized companies and associated M&A’s to round out the large orthopedic company’s portfolio and be on the cutting edge of medicine.
The planet’s extremities market, long ignored by major orthopedic device manufacturers, is finally commanding respect as companies attempt to counterbalance sluggish growth in the saturated, maturing hip and knee replacement sectors. Fueled by an aging world population, more youth-related sports injuries, and rising rates of obesity, osteoarthritis, and osteoporosis, the global extremities market is projected to be worth $15.9 billion by 2024, according to Research and Markets data. Internal fixation devices such as plates and screws have dominated the sector in recent years due to the early functionality, abbreviated hospital stays, minimal scarring, and reduced non-union risk associated with these products. Acumed LLC has particularly benefited from the high demand for internal fixation, having sold 500,000 wrist fixation implants (Acu-Loc and Acu-Loc 2) in just two years.
Such promise, of course, is garnering interest among major orthopedic companies that traditional have overlooked the sector in favor of the more lucrative large-joint sector. ODT’s May/June feature story “Going to Extremes” examines the trends and challenges driving the extremities market as well as some of the latest technologies available to patients. Dave Neal, president and founder of Morris Plains, N.J.-based STAT Design LLC, was among the experts interviewed for the feature; his full input is provided in the following Q&A.
Michael Barbella: Please discuss the trends in the extremities market. What trends have you noticed this year and how do they differ (if at all) from past trends?
Dave Neal: It seems there are more and more small players entering the extremities market, particularly in the foot and ankle realm. In the past, the big orthopedic companies tended to ignore this market, opting to concentrate on the larger hip and knee markets. Now they are adding this segment to their portfolio either through in-house product development of through an acquisition.
Barbella: What challenges face companies in the small joint (extremities) sector, and how are these challenges different than those faced by companies manufacturing large joints - i.e., hips and knees? How can companies in the extremities sector overcome these challenges?
Neal: As it is more of a niche market, the smaller extremities players are finding it difficult to gain traction in hospitals where buying groups like to have the large and small joints bundled for a full portfolio. Small companies have the best chance of success by making their extremity devices so attractive that the larger companies are seeking them out for an acquisition.
Barbella: What new technologies are on the horizon? What kinds of implants hold the most potential for patients and for market growth?
Neal: There are all sorts of new technologies emerging in the orthopedic medical device world. Additive manufacturing is changing the way parts are made. Special CAD software, such as that by nTopology, adds porous structure to designs assisting with the bone in-growth featured in implants. Materials such as PEEK are being used more, along with the addition of titanium coatings applied on this material. Foot and ankle implants are taking advantage of these new technologies and driving the market growth.
Barbella: Where does innovation come from in the extremities market? How can (do) companies stay innovative?
Neal: Innovation is being driven by understanding and addressing the doctor’s clinical needs. Observing surgery and identifying where improvements can be made to benefit the patient, surgeon, staff, hospital, and insurance companies alike are also helping to grow the extremity market.
Barbella: Aside from the implants, how does the extremities market differ from large joints, or spine? What makes extremities implants a design challenge?
Neal: For gaining FDA release through the 510k process, it can be difficult to find equivalent predicate extremity devices for comparison. This challenge has companies rethinking the decision to enter these markets, based on the regulatory path. Many times they will forego the project if it requires a pre-market approval (PMA) based on the return on investment and resources involved.
Barbella: How do the technological/physiological demands of small joints differ from other orthopedic markets?
Neal: From the design side, high stresses resulting from large forces acting on these joints and small contact areas make implant development difficult in the extremities market. The surgical procedures themselves can be more technically demanding and proper patient selection and patient activity compliance is essential for good clinical outcomes.
Barbella: Where do the extremities and trauma markets overlap? How do they differ?
Neal: The extremities and trauma markets can overlap in how they are used. For instance, ankle replacements could be used in one case to repair degenerated cartilage from arthritis and in the next it could be used as a replacement resulting from trauma in a motor vehicle accident.
Barbella: Where do you see the extremities market five years from now?
Neal: Niche implants, such as hand, fingers, wrist and associated instrumentation being developed by small specialized companies and associated M&A’s to round out the large orthopedic company’s portfolio and be on the cutting edge of medicine.