Sean Fenske, Michael Barbella, and Sam Brusco08.04.23
With any industry, the top revenue producing companies typically receive the most attention and headlines. Other firms will often look to them as a model in order to attempt to replicate their success. These organizations, however, can also share a tale of how not to proceed in an industry and valuable lessons can be learned from their struggles. As a result, entities emerge that challenge what’s been accomplished before to make their own way with novel products and services. This annual feature seeks to provide consideration for those companies attempting to address market issues from a slightly different angle than those aforementioned industry leaders.
At the heart of all good storytelling lies the suspension of disbelief.
First broached by Aristotle but formally introduced by English poet/philosopher Samuel Taylor Coleridge, the suspension of disbelief (a.k.a., willing suspension of disbelief) calls upon creative arts patrons to reject all logic in order to enjoy fictional works. Such cognitive estrangement is critical to the success of fictitious tales, as the stories are not real.
The concept has been employed fairly regularly through the ages in various literary genres, from Shakespeare’s “Henry V” and Charles Dickens “A Christmas Carol” to Mary Shelley’s “Frankenstein” and J.R.R. Tolkien’s “The Lord of the Rings.”
Suspension of disbelief is particularly vital to science-fiction storytelling, where literally anything is possible: time travel, dinosaur cloning, flying superheroes, Martian aliens, lunar outposts, interplanetary wars, humanoid robots, vampires, supernatural automobiles, and magical cemeteries.
Perfect health and immortality are routine among science-fiction protagonists, thanks to medical breakthroughs like anti-agathic drugs (“Cities in Flight”), Gholas (“Dune Messiah”), Artiforgs (“Ubik”), and Kundrenaline (“The Hands of Aten”), to name a few. Ironically, cryogenic options are available, too.
Oddly, however, there are few solutions for orthopedic-related conditions. The only treatment/potential cure is Gobathian (“Time is the Simplest Thing”), an alien drug that can miraculously repair broken bones and grow new tissue.
Unfortunately, no such miracle cures exist in the non-fiction realm. The only products that bear even a remote resemblance to Gobathian (but are far more inferior) are bone graft substitutes, bioresorbable nanocomposites, immunomodulation, tissue-engineered scaffolds, bone morphogenetic protein, and “dancing molecules.”
The sci-fi-sounding latter solution shows promise in fixing spinal cord injuries, which traditionally have been virtually impossible to reverse. An injectable therapy developed by Northwestern University researchers uses specially engineered molecules to trigger a healing response in spinal cells. In studies, scientists discovered the molecules’ motion within nanofibers could be controlled by changing their chemical structure. Injected as a liquid, the molecular therapy immediately gels into a complex network of nanofibers that mimic the spinal cord’s extracellular matrix. By matching the matrix’s structure, mimicking the motion of biological molecules and incorporating signals for receptors, the synthetic materials can communicate with cells, prompting them to repair damaged neurons.
The treatment has already proven itself in mice: Four weeks after receiving an injection of the therapy, paralyzed mice regained their ability to walk.
“The key innovation in our research, which has never been done before, is to control the collective motion of more than 100,000 molecules within our nanofibers,” said Samuel Stupp, Ph.D., Board of Trustees Professor of Materials Science and Engineering, Chemistry, Medicine, and Biomedical Engineering, and director of the Simpson Querrey Institute for BioNanotechnology, who led the study. “This research will have broad impacts for biomedical therapies…”
Broad impacts indeed: Northwestern’s research could be key to treating other irreversible neurogenerative and degenerative diseases like Alzheimer’s, stroke, or DDD (degenerative disc disease). A fairly common age-related condition, DDD affects roughly 266 million people annually (3.6% of the world’s population).
Depending on its severity, treatment for DDD ranges from pain medication to discectomies or invasive spinal fusions. But the shift to minimally invasive procedures and natural remedies are inspiring new DDD solutions, giving competition to those dancing molecules.
Step aside, Gobathian.
Spinal Stabilization Technologies Ltd. (SST) offers a fast, less invasive alternative to both TDR and spinal fusion via its PerQdisc prosthesis. Touted as the world’s only commercially available lumbar nucleus replacement system, the PerQdisc prosthesis replaces the nucleus pulposus of the intervertebral disc in the L1-S1 spinal region in patients with single-level discogenic pain.
PerQdisc is a silicone-based prosthesis formed in-situ to mimic the natural function of the native nucleus pulposus to provide a motion-preserving surgical solution. It earned the CE mark and U.S. Food and Drug Administration breakthrough device status in May 2021, and has been the subject of clinical studies outside the United States.
The custom-fit implant procedure aims to preserve anatomy, including bones, muscles, and soft tissues around the disc. A small part of the central part of the disc is removed, then perQdisc is inserted in the center of the disc, where it cures within 10 minutes, according to SST. No screws, rods, or fixation devices are placed during surgery, and patients are typically discharged earlier than those receiving a spinal fusion or TDR, the Irish firm claims.
“The results I’m getting with the PerQdisc are excellent,” Dr. Javier Duarte stated at the start of SST’s LOPAIN2 clinical trial last fall. Duarte is principal investigator at Hospital Americano in Asuncion, Paraguay, the largest enroller in SST’s clinical trial program. “I have been able to refine the technique to a point where I’m comfortable routinely offering the option to my DDD patients.”
SST expects to begin a U.S. pivotal trial for PerQdisc in Q2 2024, pursuant to an IDE being designed by spinal surgeon key opinion leaders.
Founded in 2010 by two interventional radiologists, SST and BlueRiver Acquisition Corp. are now working on a merger deal. Upon closing, the combined entity will operate as Spinal Stabilization Technologies and have an estimated $302 million post-transaction enterprise value, given a proposed future $40 million equity raise and 100% redemptions by BlueRiver public shareholders. The deal is expected to close late this year or in early 2024.
The pedicle screws help form a solid bridge between adjacent vertebrae as a form of stabilization. Although the pedicle screw construct is a 30-year-old technology, it still leads the posterior fusion instrumentation market.
ZygoFix Ltd. has reimagined this decades-old architecture with a simpler, less invasive implant that relies on the spinal column’s natural bone structure for stability. The Israeli company has developed the screw-less zLOCK implant, a miniature facet fusion cage that is inserted inside the joint and anchors into both bones to immobilize motion.
Made from rigid titanium, zLOCK adjusts its shape during implantation to accommodate the challenging synovial joint anatomy and maintains its strength to resist the joint’s applied load. With zLOCK, the surgeon leverages the vertebrae’s natural construction to provide spinal stability, and thereby eliminate the need for pedicle screws.
“When we’re looking at our anatomy, it seems that most of that bridge exists naturally in our boney construct,” ZgoFix CEO Ofer Levy explained last spring during an interview with CTech, Israel’s tech news site. “The only motion remaining is in the facet joints, or, actually, zygapophyseal, which is the source for the name of our company. What we set out to do is develop an implant which provides the stability by inserting into this [facet] joint and stabilizing it from within. By that, we can provide a much simpler procedure, much less invasive, and one that is capable of being performed as an outpatient.”
ZygoFix obtained CE mark for its minimally invasive screwless spinal fusion system in June 2021. The zLOCK system has been in clinical use for more than three years in Hungary and Israel with high levels of patient and physician satisfaction.
“We’ve been in a clinical study for over four years focused in Europe and a small one in Israel as well,” Levy stated in the CTech interview. “We’ve been advancing, seeing very good outcomes in terms of long-term pain reduction for these patients.”
There are a variety of trends driving the orthopedic device industry. Digital capabilities and single-use products are just two having an impact on the way in which treatments are being handled today. As with anything new and unique, however, adoption is not typically swift.
In the face of what can sometimes be a slower response from the industry, it’s important to bring attention to potentially superior products that represent an alternative to the traditional products with which many are comfortable. The following two companies have embraced their respective trend and developed solutions for the orthopedic industry with these at the top of their minds.
Truth be told, the company provides solutions for areas beyond ODT’s typical scope, as it focuses more on the care environment than the orthopedic device technology sector. However, it does maintain at least one aspect of its business that could be very much of interest to OEMs supporting this segment.
In the fall of 2021, GHX announced it was acquiring Explorer Surgical Corp., a supplier of a digital and remote case support platform that connects device manufacturers with healthcare provider teams. This aspect of GHX’s business is capable of putting key orthopedic product representatives virtually into the OR with surgeons and other medical professionals. Such access provides an array of advantages to both the healthcare team and the device firms, eliminating the need for travel and reducing the number of people within an OR (more of a concern since COVID).
The Explorer product is billed as a commercialization acceleration tool, as it synchronizes case support and data collection with comprehensive guides for each role in the OR. Simply put, it enables effective communication between technology supplier, surgeon, and the clinical support team to ensure an optimized procedure. A checklist of tasks helps ensure all are prepared to proceed with a surgery, while an orthopedic device OEM representative can “sit in” and offer clarification to the medical team.
While this type of interaction helps with product adoption and training, there’s an aspect of Explorer that offers advantages not as apparent as those providing clinical support. A medical device firm can leverage the Explorer platform as part of a product development strategy. As human factors considerations have become significantly more important during the design phase of a new innovation, getting real-time feedback from a surgeon can be challenging. Through the Explorer platform, a firm can have an entire design team observe a cooperating surgeon using their device, enabling the exchange of questions and comments as the technology is being used. This can only aid in the enhancement of a new product to ensure adoption, effectiveness, and efficiency through critical feedback from the experts who will use it.
Unfortunately, reprocessing of these devices has proved challenging at times and, as a result, patient safety has been compromised. With this in mind, some healthcare professionals have sought scopes that have disposable components or a significant portion of the instrument is single-use.
Pristine Surgical’s Summit is a 4K, single-use arthroscope designed to eliminate concerns associated with sterility in the form of a plug-and-play solution that results in more efficient procedures. In addition, the company’s systems all come pre-installed with Pristine Connect, which is cloud-based software for automated inventory management, image/video sharing functionality, and staff educational capabilities.
In addition, the organization announced the first-in-human procedure had taken place in April 2023. Dr. Mark Getelman performed the procedure at the Southern California Orthopedic Institute (SCOI) and offered praise for the technology.
“The Summit arthroscope performed exceptionally well, and the procedure went according to plan,” said Dr. Getelman, who specializes in arthroscopic and minimally invasive reconstructive surgery of the shoulder and knee. “The ability to get a new 4K scope for every procedure while improving our workflow makes Summit an exciting product for our operating room. We’re proud to have performed this procedure at SCOI where surgical innovation has been a hallmark of our practice since our founding.”
It’s tough to break into the large joint replacement industry. The knee, hip, and shoulder market is chock full of clinically proven implants that have been on the market for quite some time.
The only ways to break in are to develop joint replacement implants that solve a clinical need the mainstays haven’t addressed yet, create a successful joint replacement alternative, or find a way to monetize on improving the mainstay implants. The two companies I’ve chosen took the latter two of those strategies.
The first iteration was KineSpring, which is implanted in subcutaneous tissue alongside the knee joint. The partial load absorber reduces the knee joint’s load by up to 13 kg. KineSpring made its debut in Europe and reached 500 implantations by October 2013.
Shortly after a $33 million equity raise, the Atlas Knee earned EU approval in November 2015. The next-gen knee joint unloader also provides 13 kg of unloading and features a surgical technique based on patient anatomy. The first U.S. knee OA patients were treated with Atlas a year later.
The company was fairly quiet—if you call conducting various clinical trials quiet—until August 2022. Moximed raised another $40 million in Series C equity to obtain approvals and build commercial infrastructure for the next iteration of its knee ISA. Her name—MISHA.
The first data on MISHA was presented at last year’s Orthopaedic Summit in Boston. Calypso, an 81-subject, pivotal investigational device exemption (IDE) study, compared MISHA against the gold-standard high tibial osteotomy (HTO) to treat OA in the knee’s medial compartment. Calypso revealed that MISHA’s 85.6% composite success rate far outshined HTO’s 65.5%, and that MISHA reduces knee load by over 30%—more than double its predecessors. March 2023’s publication of two-year pivotal trial data yielded further encouragements: 95.8% of MISHA subjects had clinically meaningful pain relief, with 76% pain reduction and 74% functional improvement.
That was enough to satiate the FDA; MISHA grabbed U.S. marketing authorization in April 2023. Now, knee OA sufferers ineligible for joint replacement who were unable to find pain relief from all other treatments have another option to improve their quality of life.
“This is a milestone event for knee OA sufferers, and it’s the result of unwavering clinical research and development that spans more than 10 years. We offer special thanks to our study patients and surgeon investigators who helped advance the understanding of this new treatment for OA,” Moximed CEO Anton Clifford, Ph.D. proclaimed upon MISHA’s FDA nod.
The idea arose from the number of joint replacements that fail because of adverse immune responses. When small CoCr particles leach into the blood—which can happen when a hip prosthesis’ hollow CoCr femoral head articulates against its CoCr acetabular component—immune response can bring about pain and joint failure.
“Essentially, the immune system attacks the implant in a process similar to how a patient rejects an organ transplant,” David Langton, Ph.D., director of ExplantLab, explained to Genetic Engineering & Biotechnology News. “How quickly this happens is variable and unpredictable, but it appears to be dependent on the type of material, the amount of wear debris released, and other patient-specific factors.”
Dr. Langton found the culprit was genetic. Specifically, a variation in the HLA class II genotype that influences a patient’s susceptibility to delayed-type hypersensitivity (DTH) responses to CoCr implants. After a 606-patient study to refine an algorithm to predict DTH in hip replacement, Orthotype was born.
The test can be used pre- or post-op. Pre-op, blood or saliva samples can be taken that then undergo genetic sequencing to predict likelihood of a metal sensitivity reaction developing after surgery. (The saliva test can be done either at home or in the clinic). Post-op, ExplantLab provides testing kits for two blood samples that are genetically sequenced with ICP-MS to determine blood metal concentrations.
This could begin a new era where genetic testing prior to receiving medical implants becomes routine. Orthotype can help identify patients more likely to react following joint replacement made of CoCr components, helping surgeons choose an implant based on a manufactured material more suited to the patient.
“This represents a significant advance in orthopedic care for patients,” said Dr. Langton, “with potentially significant financial repercussions for global healthcare systems, through the avoidance of repeat surgery.”
In only the U.K. (for now), the MyOrthotype saliva-based home test kit is available for pre-op joint replacement patients to order online via the ExplantLab website, and more detailed, blood-based versions can be found in pathology lab services in hospitals.
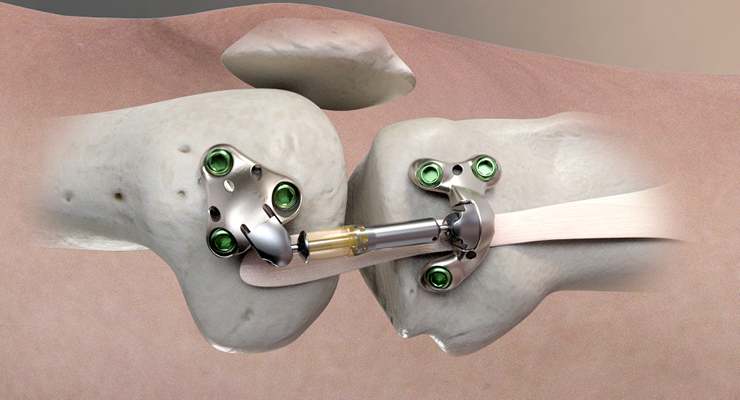
Spine Stabilization Technologies & ZygoFix
Michael Barbella • Managing EditorAt the heart of all good storytelling lies the suspension of disbelief.
First broached by Aristotle but formally introduced by English poet/philosopher Samuel Taylor Coleridge, the suspension of disbelief (a.k.a., willing suspension of disbelief) calls upon creative arts patrons to reject all logic in order to enjoy fictional works. Such cognitive estrangement is critical to the success of fictitious tales, as the stories are not real.
The concept has been employed fairly regularly through the ages in various literary genres, from Shakespeare’s “Henry V” and Charles Dickens “A Christmas Carol” to Mary Shelley’s “Frankenstein” and J.R.R. Tolkien’s “The Lord of the Rings.”
Suspension of disbelief is particularly vital to science-fiction storytelling, where literally anything is possible: time travel, dinosaur cloning, flying superheroes, Martian aliens, lunar outposts, interplanetary wars, humanoid robots, vampires, supernatural automobiles, and magical cemeteries.
Perfect health and immortality are routine among science-fiction protagonists, thanks to medical breakthroughs like anti-agathic drugs (“Cities in Flight”), Gholas (“Dune Messiah”), Artiforgs (“Ubik”), and Kundrenaline (“The Hands of Aten”), to name a few. Ironically, cryogenic options are available, too.
Oddly, however, there are few solutions for orthopedic-related conditions. The only treatment/potential cure is Gobathian (“Time is the Simplest Thing”), an alien drug that can miraculously repair broken bones and grow new tissue.
Unfortunately, no such miracle cures exist in the non-fiction realm. The only products that bear even a remote resemblance to Gobathian (but are far more inferior) are bone graft substitutes, bioresorbable nanocomposites, immunomodulation, tissue-engineered scaffolds, bone morphogenetic protein, and “dancing molecules.”
The sci-fi-sounding latter solution shows promise in fixing spinal cord injuries, which traditionally have been virtually impossible to reverse. An injectable therapy developed by Northwestern University researchers uses specially engineered molecules to trigger a healing response in spinal cells. In studies, scientists discovered the molecules’ motion within nanofibers could be controlled by changing their chemical structure. Injected as a liquid, the molecular therapy immediately gels into a complex network of nanofibers that mimic the spinal cord’s extracellular matrix. By matching the matrix’s structure, mimicking the motion of biological molecules and incorporating signals for receptors, the synthetic materials can communicate with cells, prompting them to repair damaged neurons.
The treatment has already proven itself in mice: Four weeks after receiving an injection of the therapy, paralyzed mice regained their ability to walk.
“The key innovation in our research, which has never been done before, is to control the collective motion of more than 100,000 molecules within our nanofibers,” said Samuel Stupp, Ph.D., Board of Trustees Professor of Materials Science and Engineering, Chemistry, Medicine, and Biomedical Engineering, and director of the Simpson Querrey Institute for BioNanotechnology, who led the study. “This research will have broad impacts for biomedical therapies…”
Broad impacts indeed: Northwestern’s research could be key to treating other irreversible neurogenerative and degenerative diseases like Alzheimer’s, stroke, or DDD (degenerative disc disease). A fairly common age-related condition, DDD affects roughly 266 million people annually (3.6% of the world’s population).
Depending on its severity, treatment for DDD ranges from pain medication to discectomies or invasive spinal fusions. But the shift to minimally invasive procedures and natural remedies are inspiring new DDD solutions, giving competition to those dancing molecules.
Step aside, Gobathian.
Core Correction
Although DDD-related pain can be managed with physical therapy, medication, or steroid injections, some (severe) cases require more invasive treatments such as decompression surgery, spinal fusion, or total disc replacement (TDR). Spinal fusion proficiently relieves pain but does so by sacrificing motion, thus altering the spinal column’s flexibility. TDR, on the other hand, completely removes the diseased disc and replaces it with an artificial one, but limited implant sizes and access to the affected area can prove challenging.Spinal Stabilization Technologies Ltd. (SST) offers a fast, less invasive alternative to both TDR and spinal fusion via its PerQdisc prosthesis. Touted as the world’s only commercially available lumbar nucleus replacement system, the PerQdisc prosthesis replaces the nucleus pulposus of the intervertebral disc in the L1-S1 spinal region in patients with single-level discogenic pain.
PerQdisc is a silicone-based prosthesis formed in-situ to mimic the natural function of the native nucleus pulposus to provide a motion-preserving surgical solution. It earned the CE mark and U.S. Food and Drug Administration breakthrough device status in May 2021, and has been the subject of clinical studies outside the United States.
The custom-fit implant procedure aims to preserve anatomy, including bones, muscles, and soft tissues around the disc. A small part of the central part of the disc is removed, then perQdisc is inserted in the center of the disc, where it cures within 10 minutes, according to SST. No screws, rods, or fixation devices are placed during surgery, and patients are typically discharged earlier than those receiving a spinal fusion or TDR, the Irish firm claims.
“The results I’m getting with the PerQdisc are excellent,” Dr. Javier Duarte stated at the start of SST’s LOPAIN2 clinical trial last fall. Duarte is principal investigator at Hospital Americano in Asuncion, Paraguay, the largest enroller in SST’s clinical trial program. “I have been able to refine the technique to a point where I’m comfortable routinely offering the option to my DDD patients.”
SST expects to begin a U.S. pivotal trial for PerQdisc in Q2 2024, pursuant to an IDE being designed by spinal surgeon key opinion leaders.
Founded in 2010 by two interventional radiologists, SST and BlueRiver Acquisition Corp. are now working on a merger deal. Upon closing, the combined entity will operate as Spinal Stabilization Technologies and have an estimated $302 million post-transaction enterprise value, given a proposed future $40 million equity raise and 100% redemptions by BlueRiver public shareholders. The deal is expected to close late this year or in early 2024.
LOCKing the Motion
Spinal fusion procedures for treating DDD, spondylolisthesis (slipped vertebra), and narrowing of the spinal canal (stenosis) typically entail placing pedicle screws and a short rod in the affected segment for better stability.The pedicle screws help form a solid bridge between adjacent vertebrae as a form of stabilization. Although the pedicle screw construct is a 30-year-old technology, it still leads the posterior fusion instrumentation market.
ZygoFix Ltd. has reimagined this decades-old architecture with a simpler, less invasive implant that relies on the spinal column’s natural bone structure for stability. The Israeli company has developed the screw-less zLOCK implant, a miniature facet fusion cage that is inserted inside the joint and anchors into both bones to immobilize motion.
Made from rigid titanium, zLOCK adjusts its shape during implantation to accommodate the challenging synovial joint anatomy and maintains its strength to resist the joint’s applied load. With zLOCK, the surgeon leverages the vertebrae’s natural construction to provide spinal stability, and thereby eliminate the need for pedicle screws.
“When we’re looking at our anatomy, it seems that most of that bridge exists naturally in our boney construct,” ZgoFix CEO Ofer Levy explained last spring during an interview with CTech, Israel’s tech news site. “The only motion remaining is in the facet joints, or, actually, zygapophyseal, which is the source for the name of our company. What we set out to do is develop an implant which provides the stability by inserting into this [facet] joint and stabilizing it from within. By that, we can provide a much simpler procedure, much less invasive, and one that is capable of being performed as an outpatient.”
ZygoFix obtained CE mark for its minimally invasive screwless spinal fusion system in June 2021. The zLOCK system has been in clinical use for more than three years in Hungary and Israel with high levels of patient and physician satisfaction.
“We’ve been in a clinical study for over four years focused in Europe and a small one in Israel as well,” Levy stated in the CTech interview. “We’ve been advancing, seeing very good outcomes in terms of long-term pain reduction for these patients.”
GHX Explorer Surgical & Pristine Surgical
Sean Fenske • Editor-in-ChiefThere are a variety of trends driving the orthopedic device industry. Digital capabilities and single-use products are just two having an impact on the way in which treatments are being handled today. As with anything new and unique, however, adoption is not typically swift.
In the face of what can sometimes be a slower response from the industry, it’s important to bring attention to potentially superior products that represent an alternative to the traditional products with which many are comfortable. The following two companies have embraced their respective trend and developed solutions for the orthopedic industry with these at the top of their minds.
A Virtual Visit to the OR
GHX describes itself as having established “healthcare’s largest cloud-based supply chain network, which today connects tens of thousands of healthcare organizations across the globe.” The organization’s stated goal is “to simplify the patient-centered business of healthcare to improve outcomes.”Truth be told, the company provides solutions for areas beyond ODT’s typical scope, as it focuses more on the care environment than the orthopedic device technology sector. However, it does maintain at least one aspect of its business that could be very much of interest to OEMs supporting this segment.
In the fall of 2021, GHX announced it was acquiring Explorer Surgical Corp., a supplier of a digital and remote case support platform that connects device manufacturers with healthcare provider teams. This aspect of GHX’s business is capable of putting key orthopedic product representatives virtually into the OR with surgeons and other medical professionals. Such access provides an array of advantages to both the healthcare team and the device firms, eliminating the need for travel and reducing the number of people within an OR (more of a concern since COVID).
The Explorer product is billed as a commercialization acceleration tool, as it synchronizes case support and data collection with comprehensive guides for each role in the OR. Simply put, it enables effective communication between technology supplier, surgeon, and the clinical support team to ensure an optimized procedure. A checklist of tasks helps ensure all are prepared to proceed with a surgery, while an orthopedic device OEM representative can “sit in” and offer clarification to the medical team.
While this type of interaction helps with product adoption and training, there’s an aspect of Explorer that offers advantages not as apparent as those providing clinical support. A medical device firm can leverage the Explorer platform as part of a product development strategy. As human factors considerations have become significantly more important during the design phase of a new innovation, getting real-time feedback from a surgeon can be challenging. Through the Explorer platform, a firm can have an entire design team observe a cooperating surgeon using their device, enabling the exchange of questions and comments as the technology is being used. This can only aid in the enhancement of a new product to ensure adoption, effectiveness, and efficiency through critical feedback from the experts who will use it.
Gaining a One-Time View
Minimally invasive procedures have been a rapidly growing trend in healthcare. They facilitate more rapid recoveries, less patient discomfort, and reduced scarring. A major component enabling these techniques is an endoscope, which provides the visualization required within a patient’s body to properly perform the procedure. Endoscopes can be used for other medical applications outside of surgery as well, enabling a view within the body without an incision.Unfortunately, reprocessing of these devices has proved challenging at times and, as a result, patient safety has been compromised. With this in mind, some healthcare professionals have sought scopes that have disposable components or a significant portion of the instrument is single-use.
Pristine Surgical’s Summit is a 4K, single-use arthroscope designed to eliminate concerns associated with sterility in the form of a plug-and-play solution that results in more efficient procedures. In addition, the company’s systems all come pre-installed with Pristine Connect, which is cloud-based software for automated inventory management, image/video sharing functionality, and staff educational capabilities.
In addition, the organization announced the first-in-human procedure had taken place in April 2023. Dr. Mark Getelman performed the procedure at the Southern California Orthopedic Institute (SCOI) and offered praise for the technology.
“The Summit arthroscope performed exceptionally well, and the procedure went according to plan,” said Dr. Getelman, who specializes in arthroscopic and minimally invasive reconstructive surgery of the shoulder and knee. “The ability to get a new 4K scope for every procedure while improving our workflow makes Summit an exciting product for our operating room. We’re proud to have performed this procedure at SCOI where surgical innovation has been a hallmark of our practice since our founding.”
Moximed & ExplantLab
Sam Brusco • Associate EditorIt’s tough to break into the large joint replacement industry. The knee, hip, and shoulder market is chock full of clinically proven implants that have been on the market for quite some time.
The only ways to break in are to develop joint replacement implants that solve a clinical need the mainstays haven’t addressed yet, create a successful joint replacement alternative, or find a way to monetize on improving the mainstay implants. The two companies I’ve chosen took the latter two of those strategies.
Reducing Load on the Knee
Moximed, formed in 2008 and based in Fremont, Calif., recognized the important role of load management as a strategy against knee osteoarthritis (OA). The company formed a vision to design an implantable shock absorber (ISA) to lighten the load on an arthritic knee.The first iteration was KineSpring, which is implanted in subcutaneous tissue alongside the knee joint. The partial load absorber reduces the knee joint’s load by up to 13 kg. KineSpring made its debut in Europe and reached 500 implantations by October 2013.
Shortly after a $33 million equity raise, the Atlas Knee earned EU approval in November 2015. The next-gen knee joint unloader also provides 13 kg of unloading and features a surgical technique based on patient anatomy. The first U.S. knee OA patients were treated with Atlas a year later.
The company was fairly quiet—if you call conducting various clinical trials quiet—until August 2022. Moximed raised another $40 million in Series C equity to obtain approvals and build commercial infrastructure for the next iteration of its knee ISA. Her name—MISHA.
The first data on MISHA was presented at last year’s Orthopaedic Summit in Boston. Calypso, an 81-subject, pivotal investigational device exemption (IDE) study, compared MISHA against the gold-standard high tibial osteotomy (HTO) to treat OA in the knee’s medial compartment. Calypso revealed that MISHA’s 85.6% composite success rate far outshined HTO’s 65.5%, and that MISHA reduces knee load by over 30%—more than double its predecessors. March 2023’s publication of two-year pivotal trial data yielded further encouragements: 95.8% of MISHA subjects had clinically meaningful pain relief, with 76% pain reduction and 74% functional improvement.
That was enough to satiate the FDA; MISHA grabbed U.S. marketing authorization in April 2023. Now, knee OA sufferers ineligible for joint replacement who were unable to find pain relief from all other treatments have another option to improve their quality of life.
“This is a milestone event for knee OA sufferers, and it’s the result of unwavering clinical research and development that spans more than 10 years. We offer special thanks to our study patients and surgeon investigators who helped advance the understanding of this new treatment for OA,” Moximed CEO Anton Clifford, Ph.D. proclaimed upon MISHA’s FDA nod.
Genetic Testing for Better Joint Replacement
Established in 2011, Newcastle, U.K.-based ExplantLab claims to be the largest independent retrieval unit in Europe. And thanks to bioengineers, medical staff, and patients from collaborating institutions, the company developed Orthotype, a test that uses the patient’s genotype to build a risk profile of developing cobalt-chromium (CoCr, used in hip replacement implants) hypersensitivity.The idea arose from the number of joint replacements that fail because of adverse immune responses. When small CoCr particles leach into the blood—which can happen when a hip prosthesis’ hollow CoCr femoral head articulates against its CoCr acetabular component—immune response can bring about pain and joint failure.
“Essentially, the immune system attacks the implant in a process similar to how a patient rejects an organ transplant,” David Langton, Ph.D., director of ExplantLab, explained to Genetic Engineering & Biotechnology News. “How quickly this happens is variable and unpredictable, but it appears to be dependent on the type of material, the amount of wear debris released, and other patient-specific factors.”
Dr. Langton found the culprit was genetic. Specifically, a variation in the HLA class II genotype that influences a patient’s susceptibility to delayed-type hypersensitivity (DTH) responses to CoCr implants. After a 606-patient study to refine an algorithm to predict DTH in hip replacement, Orthotype was born.
The test can be used pre- or post-op. Pre-op, blood or saliva samples can be taken that then undergo genetic sequencing to predict likelihood of a metal sensitivity reaction developing after surgery. (The saliva test can be done either at home or in the clinic). Post-op, ExplantLab provides testing kits for two blood samples that are genetically sequenced with ICP-MS to determine blood metal concentrations.
This could begin a new era where genetic testing prior to receiving medical implants becomes routine. Orthotype can help identify patients more likely to react following joint replacement made of CoCr components, helping surgeons choose an implant based on a manufactured material more suited to the patient.
“This represents a significant advance in orthopedic care for patients,” said Dr. Langton, “with potentially significant financial repercussions for global healthcare systems, through the avoidance of repeat surgery.”
In only the U.K. (for now), the MyOrthotype saliva-based home test kit is available for pre-op joint replacement patients to order online via the ExplantLab website, and more detailed, blood-based versions can be found in pathology lab services in hospitals.