ApiFix Ltd.08.27.19
ApiFix Ltd., a leading motion-preservation scoliosis correction company, has received approval from the U.S. Food and Drug Administration (FDA) via a Humanitarian Device Exemption (HDE) to market the Minimally Invasive Deformity Correction (MID-C) system for the treatment of progressive adolescent idiopathic scoliosis (AIS).
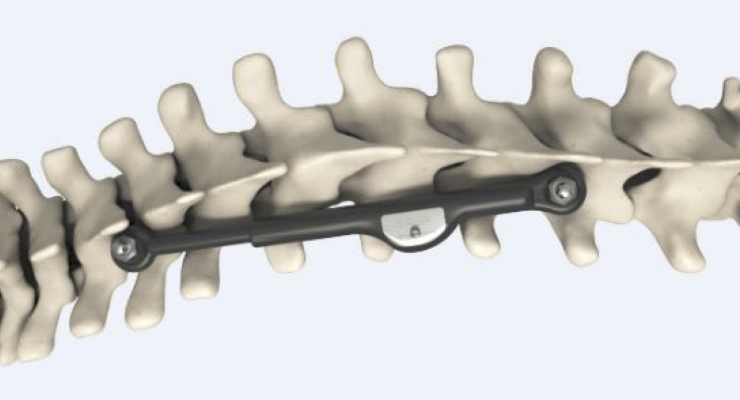
ApiFix’s MID-C technology is a posterior dynamic deformity correction (PDDC) system that enables surgeons to perform a unique treatment providing permanent curve correction while retaining spine flexibility, all via a less invasive surgical procedure compared to spinal fusion. The MID-C system acts as an “internal brace” with a patented unidirectional, self-adjusting rod mechanism and motion-preserving polyaxial joints, allowing additional post-operative correction over time.
AIS is the most common type of scoliosis with no identifiable cause and affects 2-3% of children between ages 10 to 21. Generally, AIS curves progress during an adolescent’s rapid growth period and, while most curves slow their progression significantly at the time of skeletal maturity, some deformities, especially curves greater than 50 degrees, continue to progress during adulthood.
“FDA approval of ApiFix’s MID-C system is a significant achievement for our company,” said Paul Mraz, CEO for ApiFix Ltd. “But more importantly, it makes a notable treatment advancement available for patients and their families who want FDA approved alternatives to permanent spinal fusion. Our MID-C System addresses a significant unmet clinical need for a motion-preserving alternative to spinal fusion and is a viable treatment option for progressive scoliosis in a select group of AIS patients.”
“The FDA rigorously evaluated data from patients treated with ApiFix’s MID-C system since 2012 to demonstrate the safety and probable clinical benefits of our less invasive, non-fusion deformity correction system,” said ApiFix co-founder and chief medical officer, Yizhar Floman, MD. “We are grateful to the FDA for this approval and we are excited to be in a position to offer a game-changing alternative to young patients in the United States,” continued Uri Arnin, ApiFix co-founder and chief technology officer. “Since forming the company in 2011, it has been our mission to positively change the lives of scoliosis patients forever. This HDE approval brings us much closer to achieving that goal.”
The FDA also announced its approval of ApiFix’s MID-C System via social media, stating that the approval reflects part of its “…continuing efforts to facilitate more options to pediatric patients previously limited to fusion surgeries,” and that it is “…the first posterior non-fusion device to treat adolescents with idiopathic scoliosis.”
“This FDA approval will enable surgeons to offer the first posterior dynamic deformity correction technology for treating Lenke Type 1 and Type 5 curves in patients with progressive AIS,” commented Baron Lonner, MD, chief of minimally invasive scoliosis surgery at Mount Sinai Hospital in New York City, NY and professor of orthopedic surgery at the Icahn School of Medicine. “MID-C is an alternative that fills the significant treatment gap for patients between non-operative therapies and irreversible spinal fusion.”
ApiFix’s MID-C technology is a posterior dynamic deformity correction (PDDC) system that enables surgeons to perform a unique treatment providing permanent curve correction while retaining spine flexibility, all via a less invasive surgical procedure compared to spinal fusion. The MID-C system acts as an “internal brace” with a patented unidirectional, self-adjusting rod mechanism and motion-preserving polyaxial joints, allowing additional post-operative correction over time.
AIS is the most common type of scoliosis with no identifiable cause and affects 2-3% of children between ages 10 to 21. Generally, AIS curves progress during an adolescent’s rapid growth period and, while most curves slow their progression significantly at the time of skeletal maturity, some deformities, especially curves greater than 50 degrees, continue to progress during adulthood.
“FDA approval of ApiFix’s MID-C system is a significant achievement for our company,” said Paul Mraz, CEO for ApiFix Ltd. “But more importantly, it makes a notable treatment advancement available for patients and their families who want FDA approved alternatives to permanent spinal fusion. Our MID-C System addresses a significant unmet clinical need for a motion-preserving alternative to spinal fusion and is a viable treatment option for progressive scoliosis in a select group of AIS patients.”
“The FDA rigorously evaluated data from patients treated with ApiFix’s MID-C system since 2012 to demonstrate the safety and probable clinical benefits of our less invasive, non-fusion deformity correction system,” said ApiFix co-founder and chief medical officer, Yizhar Floman, MD. “We are grateful to the FDA for this approval and we are excited to be in a position to offer a game-changing alternative to young patients in the United States,” continued Uri Arnin, ApiFix co-founder and chief technology officer. “Since forming the company in 2011, it has been our mission to positively change the lives of scoliosis patients forever. This HDE approval brings us much closer to achieving that goal.”
The FDA also announced its approval of ApiFix’s MID-C System via social media, stating that the approval reflects part of its “…continuing efforts to facilitate more options to pediatric patients previously limited to fusion surgeries,” and that it is “…the first posterior non-fusion device to treat adolescents with idiopathic scoliosis.”
“This FDA approval will enable surgeons to offer the first posterior dynamic deformity correction technology for treating Lenke Type 1 and Type 5 curves in patients with progressive AIS,” commented Baron Lonner, MD, chief of minimally invasive scoliosis surgery at Mount Sinai Hospital in New York City, NY and professor of orthopedic surgery at the Icahn School of Medicine. “MID-C is an alternative that fills the significant treatment gap for patients between non-operative therapies and irreversible spinal fusion.”