GreenBone Ortho srl02.13.20
GreenBone Ortho srl has received CE mark approval for its nature-inspired biomimetic GreenBone Bone Substitute.
Lorenzo Pradella, CEO of GreenBone, stated, "The CE mark certifies that GreenBone has successfully completed the development of the product and of its Quality System, which guarantees that all product design and production processes are in line with the ISO13485: 2016 and MDD 93/42 requirements. We believe this achievement will boost GreenBone bone substitute to become a most useful, cost-effective bone reconstructive solution.”
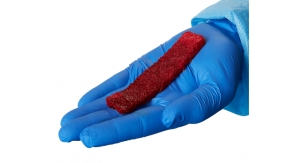
The GreenBone Bone Substitute is a nature-inspired biomimetic graft mimicking the chemical composition (not-sintered calcium phosphate phases) and the 3D porous architecture of natural bone. Derived from biomorphic transformation of rattan wood, it is endowed with osseointegration, osteoconduction, and osteoinduction properties as needed for an effective and physiological healing of large bone damages. The bone graft can be shaped by surgeons to achieve the best fit of the osseous defect.
Accoring to GreenBone, the goal is to provide orthopedics and trauma surgeons with the most innovative nature-inspired solution to offer patients the opportunity to have their own natural bone regenerated.
Every year, millions of patients undergo orthopedic surgeries where bone grafts are used to reconstruct different skeleton defects. All available solutions, including bone transplant (autograft), which is still the gold standard, have well known limitations. The GreenBone Bone Substitute will offer physicians and patients a new solution to avoid these surgical and postoperative complications. Future development will be in the spinal surgery and other skeleton defects and diseases affecting many patients. GreenBone is expected to enable patients a faster return to normal life with their own bone completely regenerated with a significant reduction of health care and social costs.
Lorenzo Pradella, CEO of GreenBone, stated, "The CE mark certifies that GreenBone has successfully completed the development of the product and of its Quality System, which guarantees that all product design and production processes are in line with the ISO13485: 2016 and MDD 93/42 requirements. We believe this achievement will boost GreenBone bone substitute to become a most useful, cost-effective bone reconstructive solution.”
The GreenBone Bone Substitute is a nature-inspired biomimetic graft mimicking the chemical composition (not-sintered calcium phosphate phases) and the 3D porous architecture of natural bone. Derived from biomorphic transformation of rattan wood, it is endowed with osseointegration, osteoconduction, and osteoinduction properties as needed for an effective and physiological healing of large bone damages. The bone graft can be shaped by surgeons to achieve the best fit of the osseous defect.
Accoring to GreenBone, the goal is to provide orthopedics and trauma surgeons with the most innovative nature-inspired solution to offer patients the opportunity to have their own natural bone regenerated.
Every year, millions of patients undergo orthopedic surgeries where bone grafts are used to reconstruct different skeleton defects. All available solutions, including bone transplant (autograft), which is still the gold standard, have well known limitations. The GreenBone Bone Substitute will offer physicians and patients a new solution to avoid these surgical and postoperative complications. Future development will be in the spinal surgery and other skeleton defects and diseases affecting many patients. GreenBone is expected to enable patients a faster return to normal life with their own bone completely regenerated with a significant reduction of health care and social costs.