Michael Barbella, Managing Editor05.18.21
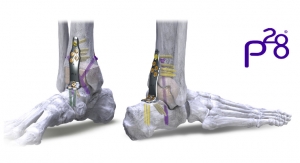
Biologica Technologies is touting clinical data that shows positive fusion rates for its ProteiOS in foot and ankle arthrodesis (fusion) procedures. Results from a retrospective, multicenter trial indicate that fusion rates are higher than or comparable to fusion rates in other published studies of any other bone graft material, including but not limited to; autograft, cellular bone matrices, and PDGF-BB.
ProteiOS has thus far been evaluated in 79 patients undergoing 121 foot and ankle fusion procedures with the primary endpoint deemed successful fusion based on high resolution CT scans and radiographs assessed by a blinded, independent radiologist. Based on radiographic evaluation the data demonstrated a fusion rate of 75.2 percent at six months and 97.1 percent fusion at 12 months. Biologica Technologies plans to submit the full data for publication in a peer-reviewed journal in the coming months as additional patients results are analyzed.
"These study results mirror the anecdotal evidence we have been hearing about ProteiOS from surgeons and sales representatives in the field," stated Jason Mattiello, vice president of Sales. "It is exciting to have this data."
ProteiOS has been shown to include more than 600 naturally occurring growth factors including but not limited to BMP-2, PDGF, TGF-β1, and VEGF. The product is provided in a lyophilized-format that supports the use of virtually any scaffold during implantation, providing flexibility for the surgeon and hospital.
ProteiOS possesses an array of osteoinductive, chemotactic, angiogenic and mitogenic growth factors that can be added to enhance a surgeon's scaffold of choice.
Biologica Technologies’ proprietary processing methods harnesses the relevant intracellular growth factors found within the endosteum layer of allograft bone. This layer is found lining the trabecular space and is comprised of mesenchymal stem cells (MSCs), osteoprogenitors, osteoblasts and pericytes.
ProteiOS has thus far been evaluated in 79 patients undergoing 121 foot and ankle fusion procedures with the primary endpoint deemed successful fusion based on high resolution CT scans and radiographs assessed by a blinded, independent radiologist. Based on radiographic evaluation the data demonstrated a fusion rate of 75.2 percent at six months and 97.1 percent fusion at 12 months. Biologica Technologies plans to submit the full data for publication in a peer-reviewed journal in the coming months as additional patients results are analyzed.
"These study results mirror the anecdotal evidence we have been hearing about ProteiOS from surgeons and sales representatives in the field," stated Jason Mattiello, vice president of Sales. "It is exciting to have this data."
ProteiOS has been shown to include more than 600 naturally occurring growth factors including but not limited to BMP-2, PDGF, TGF-β1, and VEGF. The product is provided in a lyophilized-format that supports the use of virtually any scaffold during implantation, providing flexibility for the surgeon and hospital.
ProteiOS possesses an array of osteoinductive, chemotactic, angiogenic and mitogenic growth factors that can be added to enhance a surgeon's scaffold of choice.
Biologica Technologies’ proprietary processing methods harnesses the relevant intracellular growth factors found within the endosteum layer of allograft bone. This layer is found lining the trabecular space and is comprised of mesenchymal stem cells (MSCs), osteoprogenitors, osteoblasts and pericytes.